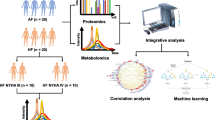
Abstract
Introduction
Atrial fibrillation (AF) is an abnormal heart rhythm characterized by an irregular beating of the atria and is associated with an increased risk of heart failure, dementia, and stroke. Currently, the perturbation of plasma content due to AF disease onset is not well known.
Objectives
To investigate dysregulated molecules in blood plasma of untreated AF patients, with the goal of identifying biomarkers for disease screening and pathological studies.
Methods
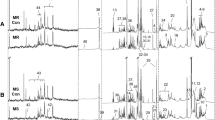
LC-MS based untargeted metabolomics, lipidomics and proteomics analyses were performed to find candidate biomarkers. A targeted quantification assay and an ELISA were performed to validate the results of the omics analyses.
Results
We found that 24 metabolites, 16 lipids and 16 proteins were significantly dysregulated in AF patients. Pathway enrichment analysis showed that the purine metabolic pathway and fatty acid metabolism were perturbed by AF onset. FA 20:2 and FA 22:4 show great linear correlational relationship with the left atrial area and could be considered for AF disease stage monitoring or prognosis evaluation.
Conclusion
we used a comprehensive multiple-omics strategy to systematically investigate the dysregulated molecules in the plasma of AF patients, thereby revealing potential biomarkers for diagnosis and providing information for pathological studies.






Similar content being viewed by others
Abbreviations
- AF:
-
Atrial fibrillation
- ECG:
-
Electrocardiogram
- LC–MS:
-
Liquid chromatography–mass spectrometry
- DDA:
-
Data-dependent acquisition
- PRM:
-
Parallel reaction monitoring
- MS/MS:
-
Tandem mass spectrometry
- QC:
-
Quality control
- PCA:
-
Principle component analysis
- FA:
-
Fatty acid
- ACar:
-
Acylcarnitine
- OxFA:
-
Oxidized fatty acid
- GSS:
-
Glutathione synthetase
References
Aebersold, R., & Mann, M. (2003). Mass spectrometry-based proteomics. Nature, 422, 198–207.
Chamberlain, A. M., Redfield, M. M., Alonso, A., Weston, S. A., & Roger, V. L. (2011). Atrial fibrillation and mortality in heart failure: A community study. Circulation Heart Failure, 4, 740–746.
Christersson, C., et al. (2018). Effect of apixaban compared with warfarin on coagulation markers in atrial fibrillation. Heart, 105(3), 235–242.
Chugh, S. S., et al. (2014). Worldwide epidemiology of atrial fibrillation: A global burden of disease 2010 study. Circulation, 129, 837–847.
Cooke, G., Doust, J., & Sanders, S. (2006). Is pulse palpation helpful in detecting atrial fibrillation? A systematic review. Journal of Family Practice, 55, 130–134.
Cravatt, B. F., Simon, G. M., & Iii, J. R. Y. (2007). The biological impact of mass-spectrometry-based proteomics. Nature, 450, 991–1000.
Dettmer, K., Aronov, P. A., & Hammock, B. D. (2007). Mass spectrometry-based metabolomics. Mass Spectrometry Reviews, 26, 51–78.
Fastner, C., et al. (2018). Interventional left atrial appendage closure affects the metabolism of acylcarnitines. International Journal of Molecular Sciences, 19(2), 500.
Foyer, C. H., et al. (1995). Overexpression of glutathione reductase but not glutathione synthetase leads to increases in antioxidant capacity and resistance to photoinhibition in poplar trees. Plant Physiology, 109, 1047–1057.
Freedman, B., et al. (2017). Screening for atrial fibrillation: A report of the AF-SCREEN international collaboration. Circulation, 135, 1851–1867.
Garcíacañaveras, J. C., Donato, M. T., Castell, J. V., & Lahoz, A. (2012). Targeted profiling of circulating and hepatic bile acids in human, mouse, and rat using a UPLC-MRM-MS-validated method. Journal of Lipid Research, 53, 2231–2241.
Go, A. S., et al. (2001). Prevalence of diagnosed atrial fibrillation in adults: National implications for rhythm management and stroke prevention: The AnTicoagulation and Risk Factors in Atrial Fibrillation (ATRIA) Study. JAMA, 285, 2370–2375.
Gstaiger, M., & Aebersold, R. (2009). Applying mass spectrometry-based proteomics to genetics, genomics and network biology. Nature Reviews Genetics, 10, 617–627.
Harada, M., Melka, J., Sobue, Y., & Nattel, S. (2017). Metabolic considerations in atrial fibrillation—mechanistic insights and therapeutic opportunities. Circulation Journal, 81, 1749–1757.
Hyman, M. C., & Deo, R. (2017). Proteomics of atrial fibrillation: Evolving From a coarse understanding to a fine phenotype. JAMA Cardiology, 2, 474.
Iwasaki, Y. K., Nishida, K., Kato, T., & Nattel, S. (2011). Atrial fibrillation pathophysiology: Implications for management. Circulation, 124, 2264–2274.
January, C. T., et al. (2014). 2014 AHA/ACC/HRS guideline for the management of patients with atrial fibrillation: A report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines and the Heart Rhythm Society. Journal of the American College of Cardiology, 64, e1–76.
Jung, Y., et al. (2018). Lipidomic profiling reveals free fatty acid alterations in plasma from patients with atrial fibrillation. PLoS ONE, 13, e0196709.
Kaarisalo, M. M., et al. (1997). Atrial fibrillation and stroke. Mortality and causes of death after the first acute ischemic stroke. Stroke, 28, 311–315.
Ko, D., et al. (2016). Metabolomic profiling in relation to new-onset atrial fibrillation (from the framingham heart study). American Journal of Cardiology, 118, 1493–1496.
Lau, J., et al. (2013). Performance of an automated iPhone ECG algorithm to diagnose atrial fibrillation in a community AF screening program (SEARCH-AF). Heart Lung and Circulation, 22, S205–S205.
Letsas, K. P., et al. (2010). Uric acid elevation in atrial fibrillation. Hellenic Journal of Cardiology, 51, 209–213.
Li, N., et al. (2016). Adenosine-induced atrial fibrillation. Circulation, 134, 486–498.
Liang, Z. Y., Pilonsmits, E. A., Jouanin, L., & Terry, N. (1999). Overexpression of glutathione synthetase in Indian mustard enhances cadmium accumulation and tolerance. Plant Physiology, 119, 73–79.
Liu, T., Zhang, X., Korantzopoulos, P., Wang, S., & Li, G. (2011). Uric acid levels and atrial fibrillation in hypertensive patients. Internal Medicine, 50, 799–803.
Marazzi, G., et al. (2012). Comparison of microlife BP A200 plus and omron M6 blood pressure monitors to detect atrial fibrillation in hypertensive patients. Advances in Therapy, 29, 64–70.
Miyasaka, Y., et al. (2006). Secular trends in incidence of atrial fibrillation in Olmsted County, Minnesota, 1980 to 2000, and implications on the projections for future prevalence. Circulation, 114, 119–125.
Ong, S. E., & Mann, M. (2005). Mass spectrometry-based proteomics turns quantitative. Nature Chemical Biology, 1, 252–262.
Pandey, A., & Mann, M. (2000). Proteomics to study genes and genomes. Nature, 405, 837–846.
Peterson, A. C., Russell, J. D., Bailey, D. J., Westphall, M. S., & Coon, J. J. (2012). Parallel reaction monitoring for high resolution and high mass accuracy quantitative, targeted proteomics. Molecular and Cellular Proteomics Mcp, 11, 1475.
Rusnak, J., et al. (2018). Interventional left atrial appendage closure may affect metabolism of essential amino acids and bioenergetic efficacy. International Journal of Cardiology, 268, 125–131.
Saito, K., & Matsuda, F. (2010). Metabolomics for functional genomics, systems biology, and biotechnology. Annual Review of Plant Biology, 61, 463–489.
Sattler, K., et al. (2017). Occlusion of left atrial appendage affects metabolomic profile: Focus on glycolysis, tricarboxylic acid and urea metabolism. Metabolomics, 13, 127.
Sievers, B., Kirchberg, S., Addo, M., Bakan, A., Brandts, B., & Trappe, H. J. (2004). Assessment of left atrial volumes in sinus rhythm and atrial fibrillation using the biplane area-length method and cardiovascular magnetic resonance imaging with TrueFISP. Journal of Cardiovascular Magnetic Resonance, 6, 855–863.
Steger, M., et al. (2016). Phosphoproteomics reveals that Parkinson’s disease kinase LRRK2 regulates a subset of Rab GTPases. eLife, 5, e12813.
Suzuki, S., et al. (2012). Gender-specific relationship between plasma uric acid level and atrial fibrillation prevalence. Circulation Journal, 76, 607–611.
Tsugawa, H., et al. (2015). MS-DIAL: Data-independent MS/MS deconvolution for comprehensive metabolome analysis. Nature Methods, 12, 523–526.
Tyanova, S., Temu, T., & Cox, J. (2016). The MaxQuant computational platform for mass spectrometry-based shotgun proteomics. Nature Protocols, 11, 2301.
Vink, R., et al. (2005). Risk of thromboembolism and bleeding after general surgery in patients with atrial fibrillation. American Journal of Cardiology, 96, 822–824.
Watanabe, E. (2012). Uric acid and atrial fibrillation. Circulation Journal, 76, 584–585.
Wishart, D. S. (2016). Emerging applications of metabolomics in drug discovery and precision medicine. Nature Reviews Drug Discovery, 15, 473–484.
Woods, C. E., & Olgin, J. (2014). Atrial fibrillation therapy now and in the future: Drugs, biologicals, and ablation. Circulation Research, 114, 1532–1546.
Zeller, T., et al. (2015). Metabolomics and incidence of atrial fibrillation in African Americans: The atherosclerosis risk in communities (ARIC) study. PLoS ONE, 10, e0142610.
Zhou, J., Liu, H., Liu, Y., Liu, J., Zhao, X., & Yin, Y. (2016). Development and evaluation of a parallel reaction monitoring strategy for large-scale targeted metabolomics quantification. Analytical Chemistry, 88, 4478–4486.
Zhou, J., Liu, C., Si, D., Jia, B., Zhong, L., & Yin, Y. (2017). Workflow development for targeted lipidomic quantification using parallel reaction monitoring on a quadrupole-time of flight mass spectrometry. Analytica Chimica Acta, 972, 62.
Zhou, J., & Yin, Y. (2016). Strategies for large-scale targeted metabolomics quantification by liquid chromatography-mass spectrometry. Analyst, 141, 6362.
Funding
This study was supported by the Fund for Fostering Young Scholars of Peking University Health Science Center (Grant No. BMU2018PY006) and the National Natural Science Foundation of China (Grant No. 81570235).
Author information
Authors and Affiliations
Contributions
JZ, LS, LZ and MC designed the study. LS, LC and SL collected samples and performed clinical related analyses. JZ and LZ performed metabolomics and proteomics experiments. JZ analyzed the data. LC and SL reviewed statistical analyses. JZ and MC wrote the manuscript. All authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Conflict of interests
The authors declare that they have no conflict of interests.
Ethics approval
The study was approved by the Clinical Ethics Committee of Peking University Third Hospital and conforms to the principles in the Declaration of Helsinki. The samples were obtained only from patients who agreed to undergo the exam for the purpose of laboratory research, and informed consent was obtained from all patients who were asked to donate blood. All methods were performed in accordance with the relevant guidelines and regulations.
Informed consent
Awritten informed consent was obtained from all the included patients.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Zhou, J., Sun, L., Chen, L. et al. Comprehensive metabolomic and proteomic analyses reveal candidate biomarkers and related metabolic networks in atrial fibrillation. Metabolomics 15, 96 (2019). https://doi.org/10.1007/s11306-019-1557-7
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11306-019-1557-7