Abstract
Introduction
Since blood is in contact with all tissues in the body and is considered to dynamically reflect the body’s pathophysiological status, serum metabolomics changes are important and have diagnostic value in early cancer detection.
Objectives
In this prospective study, we investigated the application of metabolomics to differentiate subjects with incident breast cancer (BC) from subjects who remained free of cancer during a mean follow-up period of 7 years with the aim of identifying valuable biomarkers for BC.
Methods
Baseline serum samples from 84 female subjects with incident BC (BC group) and 88 cancer-free female subjects (control group) were used. Metabolic alterations associated with BC were investigated via metabolomics analysis of the baseline serum samples using ultra-performance liquid chromatography-linear-trap quadrupole-Orbitrap mass spectrometry.
Results
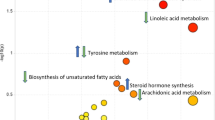
A total of 57 metabolites were identified through the metabolic analysis. Among them, 20 metabolite levels were significantly higher and 22 metabolite levels were significantly lower in the BC group than in the control group at baseline. Ten metabolic pathways, including amino acid metabolism, arachidonic acid (AA) metabolism, fatty acid metabolism, linoleic acid metabolism, and retinol metabolism, showed significant differences between the BC group and the control group. Logistic regression revealed that the incidence of BC was affected by leucine, AA, prostaglandin (PG)J2, PGE2, and γ-linolenic acid (GLA).
Conclusions
This prospective study showed the clinical relevance of dysregulation of various metabolisms on the incidence of BC. Additionally, leucine, AA, PGJ2, PGE2, and GLA were identified as independent variables affecting the incidence of BC.


Similar content being viewed by others
Data availability
The datasets used and/or analyzed during the current study are available from the corresponding author upon reasonable request.
References
Alò, P. L., Visca, P., Trombetta, G., Mangoni, A., Lenti, L., Monaco, S., et al. (1999). Fatty acid synthase (FAS) predictive strength in poorly differentiated early breast carcinomas. Tumori, 85, 35–40.
American Cancer Society. (2015). Cancer facts & figs. 2015. Atlanta, GA: American Cancer Society, Inc.
Ananieva, E. A., & Wilkinson, A. C. (2018). Branched-chain amino acid metabolism in cancer. Current Opinion in Clinical Nutrition and Metabolic Care, 21, 64–70.
Anzano, M. A., Byers, S. W., Smith, J. M., Peer, C. W., Mullen, L. T., Brown, C. C., et al. (1994). Prevention of breast cancer in the rat with 9-cis-retinoic acid as a single agent and in combination with tamoxifen. Cancer Research, 54, 4614–4617.
Asiago, V. M., Alvarado, L. Z., Shanaiah, N., Gowda, G. A., Owusu-Sarfo, K., Ballas, R. A., et al. (2010). Early detection of recurrent breast cancer using metabolite profiling. Cancer Research, 70, 8309–8318.
Bougnoux, P., Giraudeau, B., & Couet, C. (2006). Diet, cancer, and the lipidome. Cancer Epidemiology, Biomarkers & Prevention, 15, 416–421.
Bro, R., Kamstrup-Nielsen, M. H., Engelsen, S. B., Savorani, F., Rasmussen, M. A., Hansen, L., et al. (2015). Forecasting individual breast cancer risk using plasma metabolomics and biocontours. Metabolomics, 11, 1376–1380.
Budczies, J., Denkert, C., Müller, B. M., Brockmöller, S. F., Klauschen, F., Györffy, B., et al. (2012). Remodeling of central metabolism in invasive breast cancer compared to normal breast tissue—a GC-TOFMS based metabolomics study. BMC Genomics, 13, 334.
Chajès, V., Hultén, K., Van Kappel, A. L., Winkvist, A., Kaaks, R., Hallmans, G., et al. (1999). Fatty-acid composition in serum phospholipids and risk of breast cancer: An incident case-control study in Sweden. International Journal of Cancer, 83, 585–590.
Chen, S., Kong, H., Lu, X., Li, Y., Yin, P., Zeng, Z., et al. (2013). Pseudotargeted metabolomics method and its application in serum biomarker discovery for hepatocellular carcinoma based on ultra high-performance liquid chromatography/triple quadrupole mass spectrometry. Analytical Chemistry, 85, 8326–8333.
Chen, T., & Li, H. (2017). Fatty acid metabolism and prospects for targeted therapy of cancer. European Journal of Lipid Science and Technology, 119, 1600366.
Choi, Y., Park, Y., Storkson, J. M., Pariza, M. W., & Ntambi, J. M. (2002). Inhibition of stearoyl-CoA desaturase activity by the cis-9,trans-11 isomer and the trans-10,cis-12 isomer of conjugated linoleic acid in MDA-MB-231 and MCF-7 human breast cancer cells. Biochemical Biophysical Research Communications, 294, 785–790.
Denkert, C., Bucher, E., Hilvo, M., Salek, R., Orešič, M., Griffin, J., et al. (2012). Metabolomics of human breast cancer: New approaches for tumor typing and biomarker discovery. Genome Medicine, 4, 37.
Greter, J., Lindstedt, S., Seeman, H., & Steen, G. (1980). 3-Hydroxydecanedioic acid and related homologues: Urinary metabolites in ketoacidosis. Clinical Chemistry, 26, 261–265.
Hadi, N. I., & Jamal, Q. (2015). “OMIC” tumor markers for breast cancer: A review. Pakistan Journal of Medical Sciences, 31, 1256–1262.
Hanaka, H., Pawelzik, S. C., Johnsen, J. I., Rakonjac, M., Terawaki, K., Rasmuson, A., et al. (2009). Microsomal prostaglandin E synthase 1 determines tumor growth in vivo of prostate and lung cancer cells. Proceedings of the National Academy of Science of the United States of America, 106, 18757–18762.
Harizi, H., Corcuff, J. B., & Gualde, N. (2008). Arachidonic-acid-derived eicosanoids: Roles in biology and immunopathology. Trends in Molecular Medicine, 14, 461–469.
Hayden, L. J., & Satre, M. A. (2002). Alterations in cellular retinol metabolism contribute to differential retinoid responsiveness in normal human mammary epithelial cells versus breast cancer cells. Breast Cancer Research and Treatment, 72, 95–105.
Horia, E., & Watkins, B. A. (2005). Comparison of stearidonic acid and alpha-linolenic acid on PGE2 production and COX-2 protein levels in MDA-MB-231 breast cancer cell cultures. The Journal of Nutritional Biochemistry, 16, 184–192.
Hwang, D., Scollard, D., Byrne, J., & Levine, E. (1998). Expression of cyclooxygenase-1 and cyclooxygenase-2 in human breast cancer. Journal of the National Cancer Institute, 90, 455–460.
Jee, S. H., Batty, G. D., Jang, Y., Oh, D. J., Oh, B. H., Lee, S. H., et al. (2014). The Korean Heart Study: Rationale, objectives, protocol, and preliminary results for a new prospective cohort study of 430,920 men and women. European Journal of Preventive Cardiology, 21, 1484–1492.
Jee, S. H., Kim, M., Kim, M., Kang, M., Seo, Y. W., Jung, K. J., et al. (2016). Clinical relevance of glycerophospholipid, sphingomyelin and glutathione metabolism in the pathogenesis of pharyngolaryngeal cancer in smokers: The Korean Cancer Prevention Study-II. Metabolomics, 12, 164.
Jin, S. J., & Tserng, K. Y. (1989). Identification of isomeric unsaturated medium-chain dicarboxylic acids in human urine. Journal of Lipid Research, 30, 1611–1619.
Kim, M., Jung, S., Kim, S. Y., Lee, S. H., & Lee, J. H. (2014). Prehypertension-associated elevation in circulating lysophosphatidlycholines, Lp-PLA2 activity, and oxidative stress. PLoS ONE, 9, e96735.
Kim, Y. C., & Ntambi, J. M. (1999). Regulation of stearoyl-CoA desaturase genes: Role in cellular metabolism and preadipocyte differentiation. Biochemical Biophysical Research Communications, 266, 1–4.
Kundu, N., Yang, Q., Dorsey, R., & Fulton, A. M. (2001). Increased cyclooxygenase-2 (cox-2) expression and activity in a murine model of metastatic breast cancer. International Journal of Cancer, 93, 681–686.
Kusumoto, K., Kageyama, K., Tanaka, H., Kogawa, H., & Miwa, N. (2004). Enhancement of carcinostatic activity of omega-hydroxy fatty acids by their esterification through increased uptake into tumor cells. Oncology Reports, 11, 857–861.
Lee, P. P., Lee, M. T., Darcy, K. M., Shudo, K., & Ip, M. M. (1995). Modulation of normal mammary epithelial cell proliferation, morphogenesis, and functional differentiation by retinoids: A comparison of the retinobenzoic acid derivative RE80 with retinoic acid. Endocrinology, 136, 1707–1717.
Liu, S., Umezu-Goto, M., Murph, M., Lu, Y., Liu, W., Zhang, F., et al. (2009). Expression of autotaxin and lysophosphatidic acid receptors increases mammary tumorigenesis, invasion, and metastases. Cancer Cell, 15, 539–550.
Liu, X., Cheng, X., Liu, X., He, L., Zhang, W., Wang, Y., et al. (2018). Investigation of the urinary metabolic variations and the application in bladder cancer biomarker discovery. International Journal of Cancer. https://doi.org/10.1002/ijc.31323.
Lu, J., Pei, H., Kaeck, M., & Thompson, H. J. (1997). Gene expression changes associated with chemically induced rat mammary carcinogenesis. Molecular Carcinogenesis, 20, 204–215.
Lu, S., & Archer, M. C. (2005). Fatty acid synthase is a potential molecular target for the chemoprevention of breast cancer. Carcinogenesis, 26, 153–157.
Lu, X., Nie, H., Li, Y., Zhan, C., Liu, X., Shi, X., et al. (2015). Comprehensive characterization and evaluation of hepatocellular carcinoma by LC–MS based serum metabolomics. Metabolomics, 11, 1381–1393.
Maurya, P., Meleady, P., Dowling, P., & Clynes, M. (2007). Proteomic approaches for serum biomarker discovery in cancer. Anticancer Research, 27, 1247–1255.
Menendez, J. A., Decker, J. P., & Lupu, R. (2005). In support of fatty acid synthase (FAS) as a metabolic oncogene: Extracellular acidosis acts in an epigenetic fashion activating FAS gene expression in cancer cells. Journal of Cellular Biochemistry, 94, 1–4.
Moon, R. C. (1994). Vitamin A, retinoids and breast cancer. Advances in Experimental Medicine and Biology, 364, 101–107.
Moon, R. C., & Constantinou, A. I. (1997). Dietary retinoids and carotenoids in rodent models of mammary tumorigenesis. Breast Cancer Research and Treatment, 46, 181–189.
Nagata, C., Wada, K., Tsuji, M., Hayashi, M., Takeda, N., & Yasuda, K. (2014). Plasma amino acid profiles are associated with biomarkers of breast cancer risk in premenopausal Japanese women. Cancer Causes & Control, 25, 143–149.
Nakamura, I., Kimijima, I., Zhang, G. J., Onogi, H., Endo, Y., Suzuki, S., et al. (1999). Fatty acid synthase expression in Japanese breast carcinoma patients. International Journal of Molecular Medicine, 4, 381–387.
Ntambi, J. M. (1999). Regulation of stearoyl-CoA desaturase by polyunsaturated fatty acids and cholesterol. Journal of Lipid Research, 40, 1549–1558.
Oakman, C., Tenori, L., Biganzoli, L., Santarpia, L., Cappadona, S., Luchinat, C., et al. (2011). Uncovering the metabolomic fingerprint of breast cancer. The International Journal of Biochemistry & Cell Biology, 43, 1010–1020.
Pala, V., Krogh, V., Muti, P., Chajès, V., Riboli, E., Micheli, A., et al. (2001). Erythrocyte membrane fatty acids and subsequent breast cancer: A prospective Italian study. Journal of the National Cancer Institute, 93, 1088–1095.
Parrett, M., Harris, R., Joarder, F., Ross, M., Clausen, K., & Robertson, F. (1997). Cyclooxygenase-2 gene expression in human breast cancer. International Journal of Oncology, 10, 503–507.
Peng, F., Liu, Y., He, C., Kong, Y., Ouyang, Q., Xie, X., et al. (2018). Prediction of platinum-based chemotherapy efficacy in lung cancer based on LC-MS metabolomics approach. Journal of Pharmaceutical and Biomedical Analysis, https://doi.org/10.1016/j.jpba.2018.02.051.
Pignatelli, M., Sánchez-Rodríguez, J., Santos, A., & Perez-Castillo, A. (2005). 15-deoxy-Delta-12,14-prostaglandin J2 induces programmed cell death of breast cancer cells by a pleiotropic mechanism. Carcinogenesis, 26, 81–92.
Poschke, I., Mao, Y., Kiessling, R., & de Boniface, J. (2013). Tumor-dependent increase of serum amino acid levels in breast cancer patients has diagnostic potential and correlates with molecular tumor subtypes. Journal of Translational Medicine, 11, 290.
Qiu, Y., Zhou, B., Su, M., Baxter, S., Zheng, X., Zhao, X., et al. (2013). Mass spectrometry-based quantitative metabolomics revealed a distinct lipid profile in breast cancer patients. International Journal of Molecular Sciences, 14, 8047–8061.
Ristimäki, A., Sivula, A., Lundin, J., Lundin, M., Salminen, T., Haglund, C., et al. (2002). Prognostic significance of elevated cyclooxygenase-2 expression in breast cancer. Cancer Research, 62, 632–635.
Rolland, P. H., Martin, P. M., Jacquemier, J., Rolland, A. M., & Toga, M. (1980). Prostaglandin in human breast cancer: Evidence suggesting that an elevated prostaglandin production is a marker of high metastatic potential for neoplastic cells. Journal of the National Cancer Institute, 64, 1061–1070.
Schumacher, M. C., Laven, B., Petersson, F., Cederholm, T., Onelöv, E., Ekman, P., et al. (2013). comparative study of tissue ω-6 and ω-3 polyunsaturated fatty acids (PUFA) in benign and malignant pathologic stage pT2a radical prostatectomy specimens. Urologic Oncology, 31, 318–324.
Schumacher, M. C., Laven, B., Wolk, A., Brendler, C., & Ekman, P. (2007). Do omega-3 dietary fatty acids lower prostate cancer risk? A review of the literature. Current Urology, 1, 2–10.
Seewaldt, V. L., Dietze, E. C., Johnson, B. S., Collins, S. J., & Parker, M. B. (1999). Retinoic acid-mediated G1-S-phase arrest of normal human mammary epithelial cells is independent of the level of p53 protein expression. Cell Growth & Differentiation, 10, 49–59.
Seewaldt, V. L., Kim, J. H., Caldwell, L. E., Johnson, B. S., Swisshelm, K., & Collins, S. J. (1997). All-trans-retinoic acid mediates G1 arrest but not apoptosis of normal human mammary epithelial cells. Cell Growth & Differentiation, 8, 631–641.
Simińska, E., & Koba, M. (2016). Amino acid profiling as a method of discovering biomarkers for early diagnosis of cancer. Amino Acids, 48, 1339–1345.
Skaane, P. (2009). Studies comparing screen-film mammography and full-field digital mammography in breast cancer screening: Updated review. Acta Radiologica, 50, 3–14.
Spratlin, J. L., Serkova, N. J., & Eckhardt, S. G. (2009). Clinical applications of metabolomics in oncology: A review. Clinical Cancer Research, 15, 431–440.
Statistics Korea. (2018). Cancer incident cases and incidence rates by site (24 items) and sex. Retrieved February 20, 2018, from http://kosis.kr/eng/statisticsList/statisticsList_01List.jsp?vwcd=MT_ETITLE&parmT abId=M_01_01#SubCont.
Vassalle, C., Petrozzi, L., Botto, N., Andreassi, M. G., & Zucchelli, G. C. (2004). Oxidative stress and its association with coronary artery disease and different atherogenic risk factors. Journal of Internal Medicine, 256, 308–315.
World Health Organization. (2011). International statistical classification of diseases and related health problems 2010 edition. 10th revision. Volume 2 instruction manual. Geneva: WHO Press.
Xie, Y., & Meier, K. E. (2004). Lysophospholipase D and its role in LPA production. Cellular Signalling, 16, 975–981.
Zhang, J., Zhang, L., Ye, X., Chen, L., Zhang, L., Gao, Y., et al. (2013). Characteristics of fatty acid distribution is associated with colorectal cancer prognosis. Prostaglandins, Leukotrienes and Essential Fatty Acids, 88, 355–360.
Funding
This study was funded by grants from the Korean Health Technology R&D Project, the Ministry of Health & Welfare, Republic of Korea (HI14C2686010116 and HI14C2686); and the Bio-Synergy Research Project of the Ministry of Science ICT through the National Research Foundation, Republic of Korea (NRF-2012M3A9C4048762).
Author information
Authors and Affiliations
Contributions
YHJ analyzed and interpreted the data and wrote the manuscript. KM and KM analyzed and interpreted the data. KM conducted experiments. JKJ, HS, and JSH contributed to acquisition of the data. JSH and LJH contributed to the conception and designed the study. LJH interpreted the data and wrote the manuscript. All authors critically revised, read, and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
All authors declare that they have no conflicts of interest.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Yoo, H.J., Kim, M., Kim, M. et al. Analysis of metabolites and metabolic pathways in breast cancer in a Korean prospective cohort: the Korean Cancer Prevention Study-II. Metabolomics 14, 85 (2018). https://doi.org/10.1007/s11306-018-1382-4
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11306-018-1382-4