Abstract
Introduction
Schizophrenia (SCH) is one of the most common psychiatric disorders, which involves impairments in motivation and cognition. The pathological mechanisms underlying SCH are still unknown, and no effective therapies can prevent or treat perfectly the cognitive impairments and deficit symptoms caused by SCH.
Objectives
We aimed to find the lipid expression change in plasma that underlie SCH onset and antipsychotics treatment.
Methods
We performed a data independent acquisition-based untargeted lipidomic approach on a quadrupole-time of flight liquid chromatography coupled to mass spectrometry platform. The plasma lipidomic profiles of SCH patients (n = 20) pre- and post-antipsychotics treatment were acquired as well as healthy controls (n = 29). Grouped or paired t-test were used to analyze the data.
Results
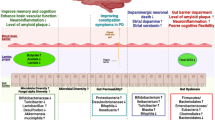
Over 1000 features were detected by our lipidomic analysis, of which 445 lipids belonging to 17 lipid species were reliably identified by tandem mass spectrometry. After statistical analysis, 47 lipids belonging to 9 lipid species were found to be dysregulated between naive SCH patients and healthy controls, and 50 lipids belonging to 9 lipid species were found to be dysregulated after antipsychotics treatment. These findings include several new SCH-relevant lipid species such as sphingomyelin, acylcarnitine and ceramide. Four types of lipid expression regulative patterns can be concluded from the above mentioned findings, revealing information about mechanism, side-effect and potential target of antipsychotics.
Conclusions
The work presented here have revealed several new lipid species which are significantly dysregulated in SCH disease development or antipsychotics treatment. These lipids provide new evidence for the pathological studies of SCH and new antipsychotics development, or can be considered as potentially candidate biomarkers for further validation.




Similar content being viewed by others
Abbreviations
- SCH:
-
Schizophrenia
- LC–MS:
-
Liquid chromatography coupled to mass spectrometry
- MS/MS:
-
Tandem mass spectrometry
- TOF:
-
Time of flight
- QC:
-
Quality control
- CE:
-
Cholesteryl ester
- DG:
-
Diacylglycerol
- TG:
-
Triacylglycerol
- FA:
-
Fatty acid
- LysoPC:
-
Lysophosphatidylcholine
- LysoPE:
-
Lysophosphatidylethanolamine
- PC:
-
Phosphatidylcholine
- PE:
-
Phosphatidylethanolamine
- p-PC:
-
Plasmenyl-PC
- p-PE:
-
Plasmenyl-PE
- PI:
-
Phosphatidylinositol
- SM:
-
Sphingomyelin
- GlcCer:
-
Glucosylceramide
- Cer:
-
Ceramide
References
Cannon, T. D., Thompson, P. M., van Erp, T. G., Toga, A. W., Poutanen, V. P., Huttunen, M., Lonnqvist, J., Standerskjold-Nordenstam, C. G., Narr, K. L., Khaledy, M., Zoumalan, C. I., Dail, R., & Kaprio, J. (2002). Cortex mapping reveals regionally specific patterns of genetic and disease-specific gray-matter deficits in twins discordant for schizophrenia. Proceedings of the National Academy of Sciences of USA, 99, 3228–3233.
Divac, N., Prostran, M., Jakovcevski, I., & Cerovac, N. (2014). Second-generation antipsychotics and extrapyramidal adverse effects. Biomedical Research International. https://doi.org/10.1155/2014/656370.
English, J. A., Pennington, K., Dunn, M. J., & Cotter, D. R. (2011). The neuroproteomics of schizophrenia. Biol Psychiatry, 69, 163–172.
Franke, B., Stein, J. L., Ripke, S., Anttila, V., Hibar, D. P., van Hulzen, K. J. E., Arias-Vasquez, A., Smoller, J. W., Nichols, T. E., Neale, M. C., McIntosh, A. M., Lee, P., McMahon, F. J., Meyer-Lindenberg, A., Mattheisen, M., Andreassen, O. A., Gruber, O., Sachdev, P. S., Roiz-Santianez, R., Saykin, A. J., Ehrlich, S., Mather, K. A., Turner, J. A., Schwarz, E., Thalamuthu, A., Shugart, Y. Y., Ho, Y. Y., Martin, N. G., Wright, M. J., C. Schizophrenia Working Group of the Psychiatric Genomics, E. Consortium, O’Donovan, M. C., Thompson, P. M., Neale, B. M., Medland, S. E., & Sullivan, P. F. (2016). Genetic influences on schizophrenia and subcortical brain volumes: Large-scale proof of concept. Nature Neuroscience, 19, 420–431.
Gauthier, J., Champagne, N., Lafreniere, R. G., Xiong, L., Spiegelman, D., Brustein, E., Lapointe, M., Peng, H., Cote, M., Noreau, A., Hamdan, F. F., Addington, A. M., Rapoport, J. L., Delisi, L. E., Krebs, M. O., Joober, R., Fathalli, F., Mouaffak, F., Haghighi, A. P., Neri, C., Dube, M. P., Samuels, M. E., Marineau, C., Stone, E. A., Awadalla, P., Barker, P. A., Carbonetto, S., Drapeau, P., Rouleau, G. A., & S. D. Team (2010). De novo mutations in the gene encoding the synaptic scaffolding protein SHANK3 in patients ascertained for schizophrenia. Proceedings of the National Academy of Sciences of USA, 107, 7863–7868.
German, J. B., Hammock, B. D., & Watkins, S. M. (2005). Metabolomics: Building on a century of biochemistry to guide human health. Metabolomics, 1, 3–9.
Glausier, J. R., & Lewis, D. A. (2013). Dendritic spine pathology in schizophrenia. Neuroscience, 251, 90–107.
Kaddurah-Daouk, R., McEvoy, J., Baillie, R. A., Lee, D., Yao, J. K., Doraiswamy, P. M., & Krishnan, K. R. (2007). Metabolomic mapping of atypical antipsychotic effects in schizophrenia. Molecular Psychiatry, 12, 934–945.
Kay, S. R., Opler, L. A., & Lindenmayer, J. P. (1989). The Positive and Negative Syndrome Scale (PANSS): Rationale and standardisation. The British Journal of Psychiatry Supplement, 59–67.
Martins-de-Souza, D., Harris, L. W., Guest, P. C., & Bahn, S. (2011). The role of energy metabolism dysfunction and oxidative stress in schizophrenia revealed by proteomics. Antioxidants and Redox Signaling, 15, 2067–2079.
McEvoy, J., Baillie, R. A., Zhu, H., Buckley, P., Keshavan, M. S., Nasrallah, H. A., Dougherty, G. G., Yao, J. K., & Kaddurah-Daouk, R. (2013). Lipidomics reveals early metabolic changes in subjects with schizophrenia: Effects of atypical antipsychotics. PLoS ONE, 8, e68717.
Mirnics, K., Middleton, F. A., Stanwood, G. D., Lewis, D. A., & Levitt, P. (2001). Disease-specific changes in regulator of G-protein signaling 4 (RGS4) expression in schizophrenia. Molecular Psychiatry, 6, 293–301.
Morris, J. A. (2003). DISC1 (Disrupted-In-Schizophrenia 1) is a centrosome-associated protein that interacts with MAP1A, MIPT3, ATF4/5 and NUDEL: Regulation and loss of interaction with mutation. Human Molecular Genetics, 12, 1591–1608.
Naz, S., Gallart-Ayala, H., Reinke, S. N., Mathon, C., Blankley, R., Chaleckis, R., & Wheelock, C. E. (2017). Development of a liquid chromatography–high resolution mass spectrometry metabolomics method with high specificity for metabolite identification using all ion fragmentation acquisition. Analytical Chemistry, 89, 7933–7942.
Oresic, M., Seppanen-Laakso, T., Sun, D., Tang, J., Therman, S., Viehman, R., Mustonen, U., van Erp, T. G., Hyotylainen, T., Thompson, P., Toga, A. W., Huttunen, M. O., Suvisaari, J., Kaprio, J., Lonnqvist, J., & Cannon, T. D. (2012). Phospholipids and insulin resistance in psychosis: A lipidomics study of twin pairs discordant for schizophrenia. Genome Medicine, 4, 1.
Oresic, M., Tang, J., Seppanen-Laakso, T., Mattila, I., Saarni, S. E., Saarni, S. I., Lonnqvist, J., Sysi-Aho, M., Hyotylainen, T., Perala, J., & Suvisaari, J. (2011). Metabolome in schizophrenia and other psychotic disorders: A general population-based study. Genome Medicine, 3, 19.
Patti, G. J., Yanes, O., & Siuzdak, G. (2012). Innovation: Metabolomics: The apogee of the omics trilogy. Nature Reviews Molecular Cell Biology, 13, 263–269.
Plumb, R. S., Johnson, K. A., Rainville, P., Smith, B. W., Wilson, I. D., Castro-Perez, J. M., & Nicholson, J. K. (2006). UPLC/MS(E); a new approach for generating molecular fragment information for biomarker structure elucidation. Rapid Communications in Mass Spectrometry, 20, 1989–1994.
Poo, S. X., & Agius, M. (2015). Atypical antipsychotics for schizophrenia and/or bipolar disorder in pregnancy: Current recommendations and updates in the NICE guidelines. Psychiatria Danubina, 27(Suppl 1), S255–S260.
Ripke, S., O’Dushlaine, C., Chambert, K., Moran, J. L., Kahler, A. K., Akterin, S., Bergen, S. E., Collins, A. L., Crowley, J. J., Fromer, M., Kim, Y., Lee, S. H., Magnusson, P. K., Sanchez, N., Stahl, E. A., Williams, S., Wray, N. R., Xia, K., Bettella, F., Borglum, A. D., Bulik-Sullivan, B. K., Cormican, P., Craddock, N., de Leeuw, C., Durmishi, N., Gill, M., Golimbet, V., Hamshere, M. L., Holmans, P., Hougaard, D. M., Kendler, K. S., Lin, K., Morris, D. W., Mors, O., Mortensen, P. B., Neale, B. M., O’Neill, F. A., Owen, M. J., Milovancevic, M. P., Posthuma, D., Powell, J., Richards, A. L., Riley, B. P., Ruderfer, D., Rujescu, D., Sigurdsson, E., Silagadze, T., Smit, A. B., Stefansson, H., Steinberg, S., Suvisaari, J., Tosato, S., Verhage, M., Walters, J. T., C. Multicenter Genetic Studies of Schizophrenia, Levinson, D. F., Gejman, P. V., Kendler, K. S., Laurent, C., Mowry, B. J., O’Donovan, M. C., Owen, M. J., Pulver, A. E., Riley, B. P., Schwab, S. G., Wildenauer, D. B., Dudbridge, F., Holmans, P., Shi, J., Albus, M., Alexander, M., Campion, D., Cohen, D., Dikeos, D., Duan, J., Eichhammer, P., Godard, S., Hansen, M., Lerer, F. B., Liang, K. Y., Maier, W., Mallet, J., Nertney, D. A., Nestadt, G., Norton, N., O’Neill, F. A., Papadimitriou, G. N., Ribble, R., Sanders, A. R., Silverman, J. M., Walsh, D., Williams, N. M., Wormley, B., C. Psychosis Endophenotypes International, Arranz, M. J., Bakker, S., Bender, S., Bramon, E., Collier, D., Crespo-Facorro, B., Hall, J., Iyegbe, C., Jablensky, A., Kahn, R. S., Kalaydjieva, L., Lawrie, S., Lewis, C. M., Lin, K., Linszen, D. H., Mata, I., McIntosh, A., Murray, R. M., Ophoff, R. A., Powell, J., Rujescu, D., Van Os, J., Walshe, M., Weisbrod, M., Wiersma, D., C. Wellcome Trust Case Control, Donnelly, P., Barroso, I., Blackwell, J. M., Bramon, E., Brown, M. A., Casas, J. P., Corvin, A. P., Deloukas, P., Duncanson, A., Jankowski, J., Markus, H. S., Mathew, C. G., Palmer, C. N., Plomin, R., Rautanen, A., Sawcer, S. J., Trembath, R. C., Viswanathan, A. C., Wood, N. W., Spencer, C. C., Band, G., Bellenguez, C., Freeman, C., Hellenthal, G., Giannoulatou, E., Pirinen, M., Pearson, R. D., Strange, A., Su, Z., Vukcevic, D., Donnelly, P., Langford, C., Hunt, S. E., Edkins, S., Gwilliam, R., Blackburn, H., Bumpstead, S. J., Dronov, S., Gillman, M., Gray, E., Hammond, N., Jayakumar, A., McCann, O. T., Liddle, J., Potter, S. C., Ravindrarajah, R., Ricketts, M., Tashakkori-Ghanbaria, A., Waller, M. J., Weston, P., Widaa, S., Whittaker, P., Barroso, I., Deloukas, P., Mathew, C. G., Blackwell, J. M., Brown, M. A., Corvin, A. P., McCarthy, M. I., Spencer, C. C., Bramon, E., Corvin, A. P., O’Donovan, M. C., Stefansson, K., Scolnick, E., Purcell, S., McCarroll, S. A., Sklar, P., Hultman, C. M., & Sullivan, P. F. (2013). Genome-wide association analysis identifies 13 new risk loci for schizophrenia. Nature Genetics, 45, 1150–1159.
Saito, K., & Matsuda, F. (2010). Metabolomics for functional genomics, systems biology, and biotechnology. Annual Review of Plant Biology, 61, 463–489.
Scalbert, A., Brennan, L., Fiehn, O., Hankemeier, T., Kristal, B. S., van Ommen, B., Pujos-Guillot, E., Verheij, E., Wishart, D., & Wopereis, S. (2009). Mass-spectrometry-based metabolomics: Limitations and recommendations for future progress with particular focus on nutrition research. Metabolomics, 5, 435–458.
Sekar, A., Bialas, A. R., de Rivera, H., Davis, A., Hammond, T. R., Kamitaki, N., Tooley, K., Presumey, J., Baum, M., Van Doren, V., Genovese, G., Rose, S. A., Handsaker, R. E., C. Schizophrenia Working Group of the Psychiatric Genomics, Daly, M. J., Carroll, M. C., Stevens, B., & McCarroll, S. A. (2016). Schizophrenia risk from complex variation of complement component 4. Nature, 530, 177–183.
Tautenhahn, R., Cho, K., Uritboonthai, W., Zhu, Z., Patti, G. J., & Siuzdak, G. (2012). An accelerated workflow for untargeted metabolomics using the METLIN database. Nature Biotechnology, 30, 826–828.
Tsugawa, H., Cajka, T., Kind, T., Ma, Y., Higgins, B., Ikeda, K., Kanazawa, M., VanderGheynst, J., Fiehn, O., & Arita, M. (2015). MS-DIAL: Data-independent MS/MS deconvolution for comprehensive metabolome analysis. Nature Methods, 12, 523–526.
Velasquez, E., Nogueira, F. C. S., Velasquez, I., Schmitt, A., Falkai, P., Domont, G. B., & Martins-de-Souza, D. (2017). Synaptosomal proteome of the orbitofrontal cortex from schizophrenia patients using quantitative label-free and iTRAQ-based shotgun proteomics. J Proteome Res. https://doi.org/10.1021/acs.jproteome.7b00422.
Wood, P. L., Unfried, G., Whitehead, W., Phillipps, A., & Wood, J. A. (2015). Dysfunctional plasmalogen dynamics in the plasma and platelets of patients with schizophrenia. Schizophrenia Research, 161, 506–510.
Xia, J., Broadhurst, D. I., Wilson, M., & Wishart, D. S. (2013). Translational biomarker discovery in clinical metabolomics: An introductory tutorial. Metabolomics, 9, 280–299.
Xia, J., Sinelnikov, I. V., Han, B., & Wishart, D. S. (2015). MetaboAnalyst 3.0—Making metabolomics more meaningful. Nucleic Acids Research, 43, W251–W257.
Zhou, J., Li, Y., Chen, X., Zhong, L., & Yin, Y. (2017). Development of data-independent acquisition workflows for metabolomic analysis on a quadrupole-orbitrap platform. Talanta, 164, 128–136.
Acknowledgements
Research reported in this publication was supported by the Fund for Fostering Young Scholars of Peking University Health Science Center (Grant No. BMU2018PY006), the Interdisciplinary Medicine Seed Fund of Peking University (Grant No. BMU2017MX006), the 111 Project (Grant B07001) and the Lam Chung Nin Foundation for Systems Biomedicine.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interests
The authors declare that they have no competing interests.
Ethics approval
This study was conducted in accordance with the guidelines of the Declaration of Helsinki and the Principles of Good Clinical Practice. The Ethical Review Board of the Peking University Health Science Center approved the study.
Informed consent
Informed consent was obtained from all patients who were asked to donate blood.
Additional information
Lailai Yan and Juntuo Zhou are equal contributors.
Rights and permissions
About this article
Cite this article
Yan, L., Zhou, J., Wang, D. et al. Unbiased lipidomic profiling reveals metabolomic changes during the onset and antipsychotics treatment of schizophrenia disease. Metabolomics 14, 80 (2018). https://doi.org/10.1007/s11306-018-1375-3
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11306-018-1375-3