Abstract
Introduction
While atenolol is an effective antihypertensive agent, its use is also associated with adverse events including hyperglycemia and incident diabetes that may offset the benefits of blood pressure lowering. By combining metabolomic and genomic data acquired from hypertensive individuals treated with atenolol, it may be possible to better understand the pathways that most impact the development of an adverse glycemic state.
Objective
To identify biomarkers that can help predict susceptibility to blood glucose excursions during exposure to atenolol.
Methods
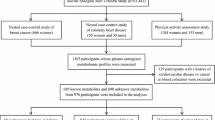
Plasma samples acquired from 234 Caucasian participants treated with atenolol in the Pharmacogenomic Evaluation of Antihypertensive Responses trial were analyzed by gas chromatography Time-Of-Flight Mass Spectroscopy. Metabolomics and genomics data were integrated by first correlating participant’s metabolomic profiles to change in glucose after treatment with atenolol, and then incorporating genotype information from genes involved in metabolite pathways associated with glucose response.
Results
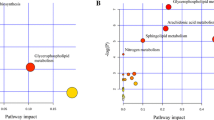
Our findings indicate that the baseline level of β-alanine was associated with glucose change after treatment with atenolol (Q = 0.007, β = 2.97 mg/dL). Analysis of genomic data revealed that carriers of the G allele for SNP rs2669429 in gene DPYS, which codes for dihydropyrimidinase, an enzyme involved in β-alanine formation, had significantly higher glucose levels after treatment with atenolol when compared with non-carriers (Q = 0.05, β = 2.76 mg/dL). This finding was replicated in participants who received atenolol as an add-on therapy (P = 0.04, β = 1.86 mg/dL).
Conclusion
These results suggest that β-alanine and rs2669429 may be predictors of atenolol-induced hyperglycemia in Caucasian individuals and further investigation is warranted.




Similar content being viewed by others
References
Akbar, S., & Alorainy, M. S. (2014). The current status of beta blockers’ use in the management of hypertension. Saudi Medical Journal, 35(11), 1307–1317. Retrieved from Oct 16, 2016. http://www.pubmedcentral.nih.gov/articlerender.fcgi?artid=4362137&tool=pmcentrez&rendertype=abstract.
Blancquaert, L., Everaert, I., & Derave, W. (2015). Beta-alanine supplementation, muscle carnosine and exercise performance. Current Opinion in Clinical Nutrition and Metabolic Care, 18(1), 63–70. doi:10.1097/MCO.0000000000000127.
Cheng, S., Rhee, E. P., Larson, M. G., Lewis, G. D., McCabe, E. L., Shen, D., et al. (2012). Metabolite profiling identifies pathways associated with metabolic risk in humans. Circulation, 125(18), 2222–2231. doi:10.1161/CIRCULATIONAHA.111.067827.
David, B. (2012). TOF-MS takes on complex GC analyses. Laboratory Equipment. Retrieved from http://www.laboratoryequipment.com/article/2012/09/tof-ms-takes-complex-gc-analyses. Accessed on Jul 11, 2016.
Evans, J. R., Kelly, D. L., Morris, K. J., Arvide, E. M., & Harris, A. (2008). RNA interference-mediated inhibition of hepatocyte nuclear factor 1alpha identifies target genes. Biochimica et Biophysica Acta, 1779(5), 341–346. doi:10.1016/j.bbagrm.2008.03.002.
Fathallah, N., Slim, R., Larif, S., Hmouda, H., & Ben Salem, C. (2015). Drug-induced hyperglycaemia and diabetes. Drug Safety. doi:10.1007/s40264-015-0339-z.
Fidlerova, J., Kleiblova, P., Bilek, M., Kormunda, S., Formankova, Z., Novotny, J., & Kleibl, Z. (2010). Contribution of dihydropyrimidinase gene alterations to the development of serious toxicity in fluoropyrimidine-treated cancer patients. Cancer Chemotherapy and Pharmacology, 65(4), 661–669. doi:10.1007/s00280-009-1071-0.
Gress, T. W., Nieto, F. J., Shahar, E., Wofford, M. R., & Brancati, F. L. (2000). Hypertension and antihypertensive therapy as risk factors for type 2 diabetes mellitus. atherosclerosis risk in communities study. The New England Journal of Medicine, 342(13), 905–912. doi:10.1056/NEJM200003303421301.
Gualano, B., Everaert, I., Stegen, S., Artioli, G. G., Taes, Y., Roschel, H., et al. (2012). Reduced muscle carnosine content in type 2, but not in type 1 diabetic patients. Amino Acids, 43(1), 21–24. doi:10.1007/s00726-011-1165-y.
Harris, R. C., Tallon, M. J., Dunnett, M., Boobis, L., Coakley, J., Kim, H. J., et al. (2006). The absorption of orally supplied beta-alanine and its effect on muscle carnosine synthesis in human vastus lateralis. Amino Acids, 30(3), 279–289. doi:10.1007/s00726-006-0299-9.
Heart Outcomes Prevention Evaluation (HOPE) Study Investigators. (2001). The HOPE Study and MICRO-HOPE Substudy: effects of ramipril on cardiovascular and microvascular outcomes in people with diabetes mellitus. The British Journal of Diabetes & Vascular Disease, 1(1), 44–51. doi:10.1177/14746514010010010701.
IMS Institute for Healthcare Informatics. (2012). The use of medicines in the United States: Review of 2011. http://www.imshealth.com/ims/Global/Content/Insights/IMS Institute for Healthcare Informatics/IHII_Medicines_in_U.S_Report_2011.pdf. Accessed 16 October 2015
Johnson, J. A., Boerwinkle, E., Zineh, I., Chapman, A. B., Bailey, K., Cooper-DeHoff, R. M., et al. (2009). Pharmacogenomics of antihypertensive drugs: Rationale and design of the Pharmacogenomic Evaluation of Antihypertensive Responses (PEAR) study. American Heart Journal, 157(3), 442–449.
Kaisaki, P. J., Menzel, S., Lindner, T., Oda, N., Rjasanowski, I., Sahm, J., et al. (1997). Mutations in the hepatocyte nuclear factor-1alpha gene in MODY and early-onset NIDDM: evidence for a mutational hotspot in exon 4. Diabetes, 46(3), 528–35. Retrieved from Feb 22, 2016. http://www.ncbi.nlm.nih.gov/pubmed/9032114.
Kearney, P. M., Whelton, M., Reynolds, K., Muntner, P., Whelton, P. K., & He, J. (2005). Global burden of hypertension—analysis of worldwide data. Lancet, 365(9455), 217–223. doi:10.1016/S0140-6736(05)17741-1.
Lindholm, L. H., Carlberg, B., & Samuelsson, O. (2005). Should beta blockers remain first choice in the treatment of primary hypertension? A meta-analysis. Lancet, 366(9496), 1545–1553. doi:10.1016/S0140-6736(05)67573-3.
Messerli, F. H., Bangalore, S., & Julius, S. (2008). Risk/benefit assessment of -blockers and diuretics precludes their use for first-line therapy in hypertension. Circulation, 117(20), 2706–2715. doi:10.1161/CIRCULATIONAHA.107.695007.
Moore, M. J., Gong, Y., Hou, W., Hall, K., Schmidt, S. O. F., Curry, R. W., et al. (2014). Predictors for glucose change in hypertensive participants following short-term treatment with atenolol or hydrochlorothiazide. Pharmacotherapy, 34(11), 1132–1140. doi:10.1002/phar.1483.
Nichols, G. A., Hillier, T. A., & Brown, J. B. (2008). Normal fasting plasma glucose and risk of type 2 diabetes diagnosis. The American Journal of Medicine, 121(6), 519–524. doi:10.1016/j.amjmed.2008.02.026.
O’Gara, P. T., Kushner, F. G., Ascheim, D. D., Casey, D. E., Chung, M. K., de Lemos, J. A., et al. (2013). 2013 ACCF/AHA guideline for the management of ST-elevation myocardial infarction: a report of the American College of Cardiology Foundation/American Heart Association Task Force on practice guidelines. Journal of the American College of Cardiology, 61(4), e78–e140. doi:10.1016/j.jacc.2012.11.019.
Purcell, S., Neale, B., Todd-Brown, K., Thomas, L., Ferreira, M. A. R., Bender, D., et al. (2007). PLINK: a tool set for whole-genome association and population-based linkage analyses. American Journal of Human Genetics, 81(3), 559–575. doi:10.1086/519795.
Rotroff, D. M., Shahin, M. H., Gurley, S. B., Zhu, H., Motsinger-Reif, A., Meisner, M., et al. (2015). Pharmacometabolomic assessments of atenolol and hydrochlorothiazide treatment reveal novel drug response phenotypes. CPT: Pharmacometrics & Systems Pharmacology, 4(11), 669–679.
Schauer, N., Steinhauser, D., Strelkov, S., Schomburg, D., Allison, G., Moritz, T., et al. (2005). GC-MS libraries for the rapid identification of metabolites in complex biological samples. FEBS Letters, 579(6), 1332–1337.
Srikanthan, P., Singhal, A., Lee, C. C., Nagarajan, R., Wilson, N., Roberts, C. K., et al. (2012). Characterization of Intra-myocellular lipids using 2d localized correlated spectroscopy and abdominal fat using MRI in type 2 diabetes. Magnetic resonance insights, 5, 29–36. doi:10.4137/MRI.S10489.
Stegen, S., Everaert, I., Deldicque, L., Vallova, S., de Courten, B., Ukropcova, B., et al. (2015). Muscle histidine-containing dipeptides are elevated by glucose intolerance in both rodents and men. PLoS One, 10(3), e0121062. doi:10.1371/journal.pone.0121062.
Sung, J., Song, Y.-M., Ebrahim, S., & Lawlor, D. A. (2009). Fasting blood glucose and the risk of stroke and myocardial infarction. Circulation, 119(6), 812–819. doi:10.1161/CIRCULATIONAHA.108.776989.
Thomas, H. R., Ezzeldin, H. H., Guarcello, V., Mattison, L. K., Fridley, B. L., & Diasio, R. B. (2007). Genetic regulation of dihydropyrimidinase and its possible implication in altered uracil catabolism. Pharmacogenetics and Genomics, 17(11), 973–987. doi:10.1097/FPC.0b013e3282f01788.
Trexler, E. T., Smith-Ryan, A. E., Stout, J. R., Hoffman, J. R., Wilborn, C. D., Sale, C., et al. (2015). International society of sports nutrition position stand: Beta-Alanine. Journal of the International Society of Sports Nutrition, 12, 30. doi:10.1186/s12970-015-0090-y.
UK Prospective Diabetes Study (UKPDS) Group. (1998). Intensive blood-glucose control with sulphonylureas or insulin compared with conventional treatment and risk of complications in patients with type 2 diabetes (UKPDS 33). Lancet, 352(9131), 837–853. doi:10.1016/S0140-6736(98)07019-6.
Yancy, C. W., Jessup, M., Bozkurt, B., Butler, J., Casey, D. E., Drazner, M. H., et al. (2013). 2013 ACCF/AHA guideline for the management of heart failure: a report of the American College of Cardiology Foundation/American Heart Association Task Force on Practice Guidelines. Journal of the American College of Cardiology, 62(16), e147–e239. doi:10.1016/j.jacc.2013.05.019.
Yusuf, S., Sleight, P., Pogue, J., Bosch, J., Davies, R., & Dagenais, G. (2000). Effects of an angiotensin-converting-enzyme inhibitor, ramipril, on cardiovascular events in high-risk patients. The Heart Outcomes Prevention Evaluation Study Investigators. The New England Journal of Medicine, 342(3), 145–153. doi:10.1056/NEJM200001203420301.
Zhou, Y., Qiu, L., Xiao, Q., Wang, Y., Meng, X., Xu, R., et al. (2013). Obesity and diabetes related plasma amino acid alterations. Clinical Biochemistry, 46(15), 1447–1452. doi:10.1016/j.clinbiochem.2013.05.045.
Acknowledgments
This study had financial support from the Pharmacogenomics Research Network (PGRN U01 GM074492), the Pharmacometabolomics Research Network (RC2-GM092729), and the National Center for Advancing Translational Sciences Clinical and Translational Science Award (NIH NCATS: UL1 TR000064, UL1TR000454, UL1 TR000135). PEAR efforts at the Mayo Clinic were also supported by funds from the Mayo Foundation.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that they have no conflict of interest.
Ethical Approval
The research outlined in this manuscript stems from human subjects research. As indicated, the research protocol was approved by the Institutional Review Board at all of the enrolling sites, and all patients provided written, voluntary informed consent prior to participation in any research procedures.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
de Oliveira, F.A., Shahin, M.H., Gong, Y. et al. Novel plasma biomarker of atenolol-induced hyperglycemia identified through a metabolomics-genomics integrative approach. Metabolomics 12, 129 (2016). https://doi.org/10.1007/s11306-016-1076-8
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11306-016-1076-8