Abstract
Genetic variability of trees influences the chemical composition of tissues. This determines herbivore impact and, consequently, herbivore performance. We evaluated the independent effects of plant genotype and provenance on the tannin content of holm oak (Quercus ilex) and their consequences for herbivory and performance of gypsy moth (Lymantria dispar) larvae. Oak seedlings of 48 open-pollinated families from six populations were grown in a common garden in central Spain. Half the plants were subjected to defoliation by gypsy moth larvae and the other half were destructively sampled for chemical analysis. Tannin content of leaves did not differ significantly among populations but differed significantly among families. Estimates of heritability (h 2) and quantitative genetic differentiation among populations for tannin content (Q ST) were 0.83 and 0.12, respectively. Defoliation was not related to the tannin content of plants nor to spine and trichome densities of leaves, although positive family–mean associations were observed between defoliation and both seed weight and plant height (P < 0.003). Among the oak populations, differential increase in larval weight gain with defoliation was observed. Leaf tannin content in Q. ilex is genetically controlled but does not influence defoliation or predict performance of the larvae. Different efficiencies of food utilisation depending on the oak genotypes indicate that other plant traits are influencing the feeding patterns and fitness of L. dispar and consequent population dynamics.





Similar content being viewed by others
References
Bailey JK, Wooley SC, Lindroth RL, Whitham TG (2006) Importance of species interactions to community heritability: a genetic basis to trophic-level interactions. Ecol Lett 9:78–85
Baldantoni D, Bellino A, Manes F, Alfani A (2013) Ozone fumigation of Quercus ilex L. slows down leaf litter decomposition with no detectable change in leaf composition. Ann For Sci 70:571–578
Barbehenn RV, Constabel CP (2011) Tannins in plant–herbivore interactions. Phytochemistry 72:1551–1565
Barbehenn RV, Jaros A, Lee G, Mozola C, Weir Q, Salminen J-P (2009a) Tree resistance to Lymantria dispar caterpillars: importance and limitations of foliar tannin composition. Oecologia 159:777–788
Barbehenn RV, Jaros A, Lee G, Mozola C, Weir Q, Salminen JP (2009b) Hydrolyzable tannins as “quantitative defenses”: limited impact against Lymantria dispar caterpillars on hybrid poplar. J Insect Physiol 55:297–304
Berenbaum MR (1995) The chemistry of defense—theory and practice. Proc Natl Acad Sci 92:2–8
Bonal R, Hernández M, Ortego J, Muñoz A, Espelta JM (2012) Positive cascade effects of forest fragmentation on acorn weevils mediated by seed size enlargement. Insect Conserv Diver 5:381–388
Büchel K, Fenning T, Gershenzon J, Hilker M, Meiners T (2016) Elm defence against herbivores and pathogens: morphological, chemical and molecular regulation aspects. Phytochem Rev 15:961–983
Busby PE, Lamit LJ, Keith AR, Newcombe G, Gehring CA, Whitham TG, Dirzo R (2015) Genetics-based interactions among plants, pathogens, and herbivores define arthropod community structure. Ecology 96:1974–1984
Caldwell E, Read J, Sanson GD (2016) Which leaf mechanical traits correlate with insect herbivory among feeding guilds? Ann Bot 117:349–361
Caliskan S (2014) Germination and seedling growth of holm oak (Quercus ilex L.): effects of provenance, temperature, and radicle pruning. iForest Biogeosci For 7:103–109
Constabel CP, Yoshida K, Walker V (2014) Diverse ecological roles of plant tannins: plant defense and beyond. Recent Advances in Polyphenol Research 4:115–142
Corcobado T, Cubera E, Juarez E, Moreno G, Solla A (2014) Drought events determine performance of Quercus ilex seedlings and increase their susceptibility to Phytophthora cinnamomi. Agric For Meteorol 192–193:1–8
Corcobado T, Miranda-Torres JJ, Martín-García J, Jung T, Solla A (2016) Early survival of Quercus ilex subspecies from different populations after infections and co-infections by multiple Phytophthora species. Plant Pathol. doi:10.1111/ppa.12627
Díaz M, Møller AP, Pulido FJ (2003) Fruit abortion, developmental selection and developmental stability in Quercus ilex. Oecologia 135:378–385
Estiarte M, De Castro M, Espelta JM (2007) Effects of resource availability on condensed tannins and nitrogen in two Quercus species differing in leaf life span. Ann For Sci 64:439–445
Feeny PP (1976) Plant apparency and chemical defense. In: Wallace JW, Mansell RL (eds) Biochemical interaction between plants and insects. Plenum Press, New York, pp. 1–40
Galván JV, Novo JJJ, Cabrera AG, Ariza D, García-Olmo J, Cerrillo RMN (2012) Population variability based on the morphometry and chemical composition of the acorn in holm oak (Quercus ilex subsp. ballota [Desf.] Samp.). Eur J For Res 131:893–904
Gharzouli K, Khennouf S, Amira S, Gharzouli A (1999) Effects of aqueous extracts from Quercus ilex L. root bark, Punica granatum L. fruit peel and Artemisia herba-alba Asso leaves on ethanol-induced gastric damage in rats. Phytother Res 13:42–45
Gimeno T, Pías B, Lemos-Filho JP, Valladares F (2009) Plasticity and stress tolerance override local adaptation in the responses of Mediterranean holm oak seedlings to drought and cold. Tree Physiol 29:87–98
Guzmán B, Rodríguez-López CM, Forrest A, Cano E, Vargas P (2015) Protected areas of Spain preserve the neutral genetic diversity of Quercus ilex L. irrespective of glacial refugia. Tree Genet Genomes 11:124
Hagerman AE (1987) Radial diffusion method for determination tannin in plant extracts. J Chem Ecol 13:437–449
Hanley ME, Lamont BB, Fairbanks MM, Rafferty CM (2007) Plant structural traits and their role in anti-herbivore defence. Persp Plant Ecol Evol System 8:157–178
Hernández-Serrano A, Verdú M, Santos-del-Blanco L, Climent J, González-Martínez SC, Pausas JG (2014) Heritability and quantitative genetic divergence of serotiny, a fire-persistence plant trait. Ann Bot 114:571–577
Ibáñez-Justicia A, Soto A, Martínez Gonzalvo M, Pérez-Laorga Arias E (2007) Distribución y abundancia de Lymantria dispar (Linnaeus, 1758) (Lepidoptera: Lymantriidae) en las principales masas de carrasca Quercus ilex (L.) subsp. rotundifolia (Lam.) y alcornoque Quercus suber (L.) de la Comunitat Valenciana. Bol San Veg Plagas 33:491–502
Jayaraman K (1999) A statistical manual for forestry research. FORSPA-FAO Publication, Bangkok
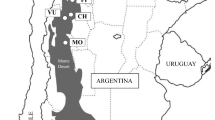
Jiménez-Sancho P, Díaz-Fernández PM, Iglesias-Sauce S, de Tuero M, Gil L (1996) Regiones de procedencia de Quercus ilex L. en España. ICONA, Madrid
Johnson MTJ, Ives AR, Ahern J, Salminen JP (2014) Macroevolution of plant defenses against herbivores in the evening primroses. New Phytol 203:267–279
Jung T, Orlikowski L, Henricot B et al (2016) Widespread Phytophthora infestations in European nurseries put forest, semi-natural and horticultural ecosystems at high risk of Phytophthora diseases. Forest Pathol 46:134–163
Karioti A, Sokovic M, Ciric A, Koukoulitsa C, Bilia AR, Skaltsa H (2011a) Antimicrobial properties of Quercus ilex L. proanthocyanidin dimers and simple phenolics: evaluation of their synergistic activity with conventional antimicrobials and prediction of their pharmacokinetic profile. J Agr Food Chem 59:6412–6422
Karioti A, Tooulakoc G, Bilia AR, Psaras GK, Karabourniotis G, Skaltsa H (2011b) Erinea formation on Quercus ilex leaves: anatomical, physiological and chemical responses of leaf trichomes against mite attack. Phytochemistry 72:230–237
Kite M, Thomson R (2006) Conservation of leather and related materials. Butterworth-Heinemann, Oxford
Leinonen T, McCairns RJS, O’Hara RB, Merilä J (2013) QST–FST comparisons: evolutionary and ecological insights from genomic heterogeneity. Nat Rev Genet 14:179–190
Liebhold A, Elkinton J, Williams D, Muzika RM (2000) What causes outbreaks of the gypsy moth in North America? Popul Ecol 42:257–266
Martín-García J, Solla A, Corcobado T, Siasou E, Woodward S, Belbahri L (2015) Influence of temperature on germination of Quercus ilex in Phytophthora cinnamomi, P. gonapodyides, P. quercina and P. psychrophila infested soils. For Pathol 45(3):215–223
McKown AD, Guy RD, Quamme L et al (2014) Association genetics, geography and ecophysiology link stomatal patterning in Populus trichocarpa with carbon gain and disease resistance trade-offs. Mol Ecol 23:5771–5790
Michaud H, Lumaret R, Romane F (1992) Variation in the genetic structure and reproductive biology of holm oak populations. Vegetatio 99–100:107–113
Michaud H, Toumi L, Lumaret R, Li TX, Romane F, Di Guisto F (1995) Effect of geographical discontinuity on genetic variation in Quercus ilex L. (holm oak): evidence from enzyme polymorphism. Heredity 74:590–606
Milanović S, Lazarević J, Popović Z, Miletić Z, Kostić M, Radulović Z, Karadžić D, Vuleta A (2014) Preference and performance of the larvae of Lymantria dispar (Lepidoptera: Lymantriidae) on three species of European oaks. Eur J Entomol 111:371–378
Milanović S, Lazarević J, Karadžić D, Milenković I, Jankovský L, Vuleta A, Solla A (2015) Belowground infections of the invasive Phytophthora plurivora pathogen enhance the suitability of red oak leaves to the generalist herbivore Lymantria dispar. Ecol Entomol 40:479–482
Morin RS, Liebhold AM (2016) Invasive forest defoliator contributes to the impending downward trend of oak dominance in eastern North America. Forestry 89:284–289
Mrdaković M, Perić-Mataruga V, Ilijin L, Vlahović M, Todorović D, Nenadović V, Lazarević J (2011) The effects of tannic acid on the fitness-related traits of Lymantria dispar L. larvae. Arch Biol Sci 63:1037–1045
Mrdaković M, Perić-Mataruga V, Ilijin L, Vlahović M, Todorović D, Nenadović V, Lazarević J (2013) Effects of tannic acid on trypsin and leucine aminopeptidase activities in gypsy moth larval midgut. Arch Biol Sci 65:1405–1413
Niinemets Ü (2015) Is there a species spectrum within the world-wide leaf economics spectrum? Major variations in leaf functional traits in the Mediterranean sclerophyll Quercus ilex. New Phytol 205:79–96
Nisar M, Ghafoor A, Wadood SF, Iqbal A (2016) Intra and inter specific profiling of Pakistani Quercus species growing in the hilly areas of District Dir Khyber Pakhtunkhwa, Pakistan. Pak J Bot 48:263–270
Ortego J, Bonal R, Muñoz A (2010) Genetic consequences of habitat fragmentation in long-lived tree species: the case of the Mediterranean holm oak (Quercus ilex L.). J Hered 101:717–726
Ortego J, Bonal R, Muñoz A, Aparicio JM (2014) Extensive pollen immigration and no evidence of disrupted mating patterns or reproduction in a highly fragmented holm oak stand. J Plant Ecol 7:384–395
Osier T, Lindroth RL (2001) Effects of genotype, nutrient availability, and defoliation on aspen phytochemistry and insect performance. J Chem Ecol 27:1289–1313
Osier TL, Hwang SY, Lindroth RL (2000) Effects of phytochemical variation in quaking aspen Populus tremuloides clones on gypsy moth Lymantria dispar performance in the field and laboratory. Ecol Entomol 25:197–207
Pearse IS (2011) The role of leaf defensive traits in oaks on the preference and performance of a polyphagous herbivore, Orgyia vetusta. Ecol Entomol 36:635–642
Pollastrini M, Feducci M, Bonal D et al (2016) Physiological significance of forest tree defoliation: results from a survey in a mixed forest in Tuscany (central Italy). Forest Ecol Manag 361:170–178
Ramírez-Valiente JA, Valladares F, Aranda I (2014) Exploring the impact of neutral evolution on intrapopulation genetic differentiation in functional traits in a long-lived plant. Tree Genet Genomes 10:1181–1190
Rubert-Nason KF, Couture JJ, Major IT, Constabel CP, Lindroth RL (2015) Influence of genotype, environment, and gypsy moth herbivory on local and systemic chemical defenses in trembling aspen (Populus tremuloides). J Chem Ecol 41:651–661
Salminen JP, Karonen M (2011) Chemical ecology of tannins and other phenolics: we need a change in approach. Funct Ecol 25:325–338
Santiso X, López L, Gilbert KJ, Barreiro R, Whitlock MC, Retuerto R (2015) Patterns of genetic variation within and among populations in Arbutus unedo and its relation with selection and evolvability. Perspect Plant Ecol 17:185–192
Solla A, Aguín O, Cubera E, Sampedro L, Mansilla JP, Zas R (2011) Survival time analysis of Pinus pinaster inoculated with Armillaria ostoyae: genetic variation and relevance of seed and root traits. Eur J Plant Pathol 130:477–488
Solla A, López-Almansa JC, Martín JA, Gil L (2015) Genetic variation and heritability estimates of Ulmus minor and Ulmus pumila hybrids for budburst, growth and tolerance to Ophiostoma novo-ulmi. iForest Biogeosci For 8:422–430
Soto A, Lorenzo Z, Gil L (2007) Differences in fine-scale genetic structure and dispersal in Quercus ilex L. and Q. suber L.: consequences for regeneration of Mediterranean open woods. Heredity 99:601–607
Spathelf P, van der Maaten E, van der Maaten-Theunissen M, Campioli M, Dobrowolska D (2014) Climate change impacts in European forests: the expert views of local observers. Ann Forest Sci 71:131–137
Stevens MT, Lindroth RL (2005) Induced resistance in the indeterminate growth of aspen (Populus tremuloides). Oecologia 145:297–305
Stong RA, Kolodny E, Kelsey RG, González-Hernández MP, Vivanco JM, Manter DK (2013) Effect of plant sterols and tannins on Phytophthora ramorum growth and sporulation. J Chem Ecol 39:733–743
Tapias R, Moreira AC, Fernández M, Saenz A, Domingos AC, Melo E, Cravador A (2008) Variability in the tolerance/resistance of Quercus suber L. seedlings to Phytophthora cinnamomi Rands: evaluation of survival. In: Vázquez-Piqué J, Pereira H, González-Pérez A (eds) Suberwood: new challenges for the integration of cork oak forests and products. Universidad de Huelva, Huelva, pp. 237–246
Vernesi C, Rocchini D, Pecchioli E, Neteler M, Vendramin GG, Paffetti D (2012) A landscape genetics approach reveals ecological-based differentiation in populations of holm oak (Quercus ilex L.) at the northern limit of its range. Biol J Linn Soc 107:458–467
Vivas M, Zas R, Sampedro L, Solla A (2013) Environmental maternal effects mediate the resistance of maritime pine to biotic stress. PLoS One 8:e70148
Vivas M, Nunes C, Coimbra MA, Solla A (2014) Maternal effects and carbohydrate changes of Pinus pinaster after inoculation with Fusarium circinatum. Trees 28:373–379
Wallis IR, Edwards MJ, Windley H et al (2012) Food for folivores: nutritional explanations linking diets to population density. Oecologia 169:281–291
Züst T, Agrawal AA (2016) Mechanisms and evolution of plant resistance to aphids. Nat Plants 2:15206
Acknowledgments
We thank Adrián Mateos, Andrea Pérez and Marta Company for technical help, Dr. Rafael Zas (Misión Biológica, CSIC) for kindly helping us with the statistics, and Jane McGrath for correcting the manuscript. We thank Dr. Rowland Burdon and one anonymous reviewer for very useful comments on the manuscript. This study was funded by the Spanish Ministry of Agriculture, Food and Environment (Ministerio de Agricultura, Alimentación y Medio Ambiente) (project 956, ‘Determinants of biotic resistance in a model tree species: a new tool for adaptive management in national parks’) and the Ministry of Education, Science and Technological Development of the Republic of Serbia (project 43007, ‘Studying climate change and its influence on the environment: impacts, adaptation and mitigation’).
Data archiving statement
Tannin content, defoliation and plant and larval performance data may be requested from the corresponding author (asolla@unex.es).
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by R. Burdon
Rights and permissions
About this article
Cite this article
Solla, A., Milanović, S., Gallardo, A. et al. Genetic determination of tannins and herbivore resistance in Quercus ilex . Tree Genetics & Genomes 12, 117 (2016). https://doi.org/10.1007/s11295-016-1069-9
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11295-016-1069-9