Abstract
West Nile virus (WNV) was first detected in the western hemisphere during the summer of 1999, reawakening US public awareness of the potential severity of vector-borne pathogens. Since its New World introduction, WNV has caused disease in human, avian, and mammalian communities across the continent. American crows (Corvus brachyrhynchos) are a highly susceptible WNV host and when modeled appropriately, changes in crow abundances can serve as a proxy for the spatio-temporal presence of WNV. We use the dramatic declines in abundance of this avian host to examine spatio-temporal heterogeneity in WNV intensity across the northeastern US, where WNV was first detected. Using data from the Breeding Bird Survey, we identify significant declines in crow abundance after WNV emergence that are associated with lower forest cover, more urban land use, and warmer winter temperatures. Importantly, we document continued declines as WNV was present in an area over consecutive years. Our findings support the urban-pathogen link that human WNV incidence studies have shown. For each 1% increase in urban land cover we expect an additional 5% decline in the log crow abundance beyond the decline attributed to WNV in undeveloped areas. We also demonstrate a significant relationship between above-average winter temperatures and WNV-related declines in crow abundance. The mechanisms behind these patterns remain uncertain and hypotheses requiring further research are suggested. In particular, a strong positive relationship between urban land cover and winter temperatures may confound mechanistic understanding, especially when a temperature-sensitive vector is involved.
Similar content being viewed by others
Introduction
The extent and intensity of human development has exceeded the rate of population growth during the past decades (Liu et al. 2003; Brown et al. 2005) making anthropogenic disturbance ever more prevalent across global ecosystems (Grimm et al. 2008; Sanderson et al. 2002). This has already had profound consequences for ecosystem services, including protection from infectious disease (Millennium Ecosystem Assessment 2005). The risks of zoonotic and vector-borne disease in humans and wild animals have increased in the past decades, especially at sites with high human disturbances (Myers and Patz 2009; Jones et al. 2008; Bradley and Altizer 2007; Wilcox and Gubler 2005; Taylor et al. 2001).
West Nile virus (WNV) was historically seen only in the eastern hemisphere and its detection in North America in 1999 (Lanciotti et al. 1999) reawakened US citizens to potential risks associated with mosquito-borne disease (which had largely disappeared after malaria eradication post-WW II). WNV depends on both arthropod (mosquito) vectors and vertebrate (avian) hosts for persistence and amplification in the environment. Humans are at risk when a competent mosquito feeds from an infectious host and then from a human. WNV zoonotic hosts include a number of diverse avian species, although studies suggest that host competence (ability to both be infected and become infectious) is more variable than the long list of species that have been recorded with WNV infections (i.e., CDC 2009) might suggest (Kilpatrick et al. 2007; Komar et al. 2003). Current understanding of how mosquito community composition influences the timing and intensity of avian enzootics and human epidemics remains uncertain, although studies agree that species from the genus Culex (family Culicidae) are important (Kilpatrick et al. 2005; Turell et al. 2005; Andreadis et al. 2004). The emergence of WNV in New York City in 1999 is most likely an example of pathogen movement due to global travel and trade, similar to the 2003 introduction of monkey pox to the American Midwest and the cross-continent spread of both SARS and H5N1 (Anderson et al. 2004; Kilpatrick et al. 2006a). However, the fact that competent hosts and vectors coincided at necessary densities to facilitate WNV spread and support growing endemism of WNV across the North American continent in under 5 years is remarkable.
The rapid dispersal of WNV across North America has resulted in a complex spatio-temporal pattern of WNV incidence and impact in both the primary avian hosts (Bradley et al. 2008; LaDeau et al. 2008; Gibbs et al. 2006; Kilpatrick et al. 2006b; Komar et al. 2005; Yaremych et al. 2004) and in humans (Sugumaran et al. 2009; Reisen et al. 2009; Liu et al. 2009; Brown et al. 2008; Ruiz et al. 2007, 2006; Brownstein et al. 2003). The spatial variability in WNV prevalence likely reflects the combined influence of stochastic dispersal (Kilpatrick et al. 2007) and habitat heterogeneity at both host- and vector-specific scales (Bradley et al. 2008; Brown et al. 2008; Gibbs et al. 2006). A number of studies have correlated WNV seroprevalence in birds (Bradley et al. 2008; Gibbs et al. 2006), small mammals (Gomez et al. 2008), and proportions of WNV-positive mosquito traps (Andreadis et al. 2004) with measures of urban land cover and/or human density. Higher incidence of human WNV cases has also been associated with higher human densities (Liu et al. 2009) and reduced forest cover (Brown et al. 2008) in the northeastern US, but also with more rural (often agricultural) characteristics in the Northern Great Plains (Sugumaran et al. 2009) and Iowa (DeGroote et al. 2008). Inter-regional differences in the influence of urbanization on WNV risk are perhaps unsurprising given habitat and regional differences in the composition of mosquito communities (Darsie and Ward 2005) and potential WNV vector species (Turell et al. 2005). Proposed mechanisms for intra-regional spatial patterns include heterogeneities in host community competence (Loss et al. 2009; Kilpatrick et al. 2006b) and host diversity (Allan et al. 2009), as well as complex ecological interactions driven by mosquito feeding preferences (Kilpatrick et al. 2006c). Vegetation cover and associated variability in vector habitat were associated with human WNV risk in New York City (Brownstein et al. 2003) and both vegetation cover and age of housing were important predictors of WNV risk in Chicago (Ruiz et al. 2006).
The goal of this paper is to explore the connections between land use and WNV amplification and persistence in the environment by examining spatio-temporal changes in the population dynamics of American crows (Corvus brachyrhynchos), a host species with high WNV sensitivity (Komar et al. 2003). American crows are especially susceptible to WNV infection, with nearly 100% mortality in laboratory challenge experiments (Komar et al. 2003) and very low to nonexistent seroprevalence in the wild (Wilcox et al. 2007, but see Reed et al. 2009). We build on the analysis by LaDeau et al. (2007), which documented clear consequences of WNV emergence for crow and other native avian species. Furthermore, work by LaDeau et al. (2007, 2008) indicated that the spatial and temporal variation in crow declines was strongly correlated with similar patterns in human WNV incidence when these data were available. Because several sources of long-term bird population data are freely available [i.e., USGS Breeding Bird Survey (BBS), Audubon Christmas Bird Count], evaluating changes in crow abundances represents a relatively inexpensive and consistent source of proxy data for WNV intensity. Additionally, because these data are not collected specifically in response to the WNV epidemic in the US, they (1) extend backwards, prior to WNV emergence, and (2) include more spatially even information on impacts to the host community that are not necessarily associated with human epidemics, whereas WNV-specific data are generally focused on locations with known (documented) WNV outbreaks. The current study integrates spatial and temporal structure into the model presented in LaDeau et al. (2007) to examine the spatial heterogeneity in crow declines across the northeastern United States—where WNV has been present longest. We examine the relationship between WNV-related crow declines and land use at local and regional spatial scales to evaluate the hypothesis that WNV transmission is positively associated with human-dominated landscapes.
Methods
Data
Data to estimate American crow abundances were collected by skilled volunteers as part of the North American BBS (USGS 2008). Each BBS route is comprised of a specific 24.5-mile stretch of secondary road that is visited annually in June. All birds seen or heard during 3-min intervals at fifty stops (located every 0.5 miles) along the BBS route are recorded. In this study we used time series data from 1994 to 2004, which captures the arrival of WNV in this region (1999) and does not extend too far beyond our best land-cover data (satellite requisition in 2001, see below for details). We used the total annual count (summed over 50 stops per route) for each of 149 routes located across the northeastern US (Fig. 1) where WNV was present by 2000 and documented in human cases by 2001 (CDC, http://www.cdc.gov/ncidod/dvbid/westnile/Mapsactivity/surv&control01Maps.htm). There were 100 missing counts out of the possible 1,639 observations, which were collected by 195 unique observers. The presence or absence of WNV at each route-by-year node was assigned using mosquito and human detection data for each county in our study from information collected by the ArboNET system of the Centers for Disease Control (data acquired from J. Lehman, Div. Vector-borne Infectious Diseases, CDC). We assumed that annual detection within a county indicated potential WNV exposure along any BBS route intersecting that county during that year. WNV presence was indicated in 43% of sites in 2000 and had increased to 88% by 2004.
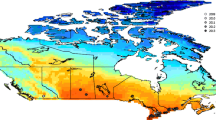
Map of BBS sites (circles are route centers) in the eastern US that were used in this study. Large circles denote sites that were characterized as ‘urban’ (light) and ‘forest’ (black) landscapes for Fig. 3. The asterisk in the inset shows New York City, the site where WNV was first detected
We examined land cover at two spatial scales. In a landscape-scale analysis that captures the full range of habitats where crows seen along the BBS route might feed, breed and roost (Yaremych et al. 2004). We identified the centroid of each route and then buffered the centroid by the length of the route radius (19.7 km, the maximum distance between the starting point and the mid-point of a route). Data from the National Land Cover Database were downloaded at the USGS Land Cover Institute (available online at: http://landcover.usgs.gov/). We tabulated the number of pixels in each land cover class within each buffer around the center of each BBS route. In a separate, fine-scale analysis we used the same land-cover data sources to evaluate land-cover characteristics within a roughly 0.5-km buffer around each BBS route which may better reflect wintering roosts or local mosquito transmission (e.g., Ward et al. 2006). Depending on the shape (straightness) of the actual route, this created discrepancies in total area so we used percentage land-cover classes for our fine-scale analysis. For the purposes of this study (at both spatial scales), land uses classified as deciduous, evergreen, and mixed forest were combined into a single forest class and low- and high-intensity residential, commercial, and urban grasslands were grouped as an urban class. The relative covers were actually fairly consistent across spatial scales; forest cover ranged from 1 to 93% (mean 45%) and urban cover ranged from 0.1 to 44% (mean 9%) using the fine-scale buffers and ranged 10–84% (mean 41%) and 1–41% (mean 12%), respectively, for the landscape-scale buffers.
Mean annual winter temperature (December–February) was estimated from 20 weather stations located throughout the study domain (http://data.giss.nasa.gov/csci/stations/). These temperature observations were used to predict temperature intervals for each year at the specific BBS locations using a Bayesian spatial interpolation model in WinBUGS (http://www.mrc-bsu.cam.ac.uk/bugs/; Lunn et al. 2000) using the spatial.pred and spatial.exp functions (Banerjee et al. 2004). Mean winter temperatures ranged from −6.9 to 7.0°C with mean 0.69°C and standard deviation 2.75°C.
Statistical analysis
We fitted an extended version of the Bayesian Poisson log linear regression model introduced in LaDeau et al. (2007). We let Y it denote the observed crow count at site i and time t where \( i = 1, \ldots ,N \), \( t = 1, \ldots ,T \), and N and T are the number of sites and the number of years observed. The counts (Y it ) are conditionally independent across years and sites with unknown parameters λ it representing the true mean abundance at site i and year t. We assumed that each of these conditional distributions is Poisson with mean λ it :
We then assumed that conditional on an unknown site and year-specific mean parameter μ it and unknown variance parameter σ 2, the natural logarithm of the λ it s are independent and normally distributed with mean μ it and variance σ 2, where σ 2 captures overdispersion with respect to the Poisson distributional assumption. Then, for all \( i = 1, \ldots ,N \) and \( t = 1, \ldots ,T, \)
Here, γ it is a site-specific temporal random effect, β 0 is the intercept, β 1 is the slope parameter corresponding to a linear trend in time, ω k is an observer-specific term, and η i is the site-specific term. X it is a vector of covariates consisting of (1) WNV: an indicator of WNV presence (equals 1 in all years and sites where WNV was detected in humans or mosquitoes and 0 otherwise), (2) \( {\text{WNV}} . {\text{YR:}}\max (0,t - t_{i}^{\text{WNV}} ) \) where \( t_{i}^{\text{WNV}} \)is the year when WNV was first detected at site i, (3) TEMP: site-specific winter temperature for current and previous year, (4) FOR: proportion forest cover, (5) URB: proportion urban/developed land cover. Covariates 3–5 were standardized by subtracting the global mean and dividing by the standard deviation. We also evaluated three interaction terms: (6) WNV.TEMP: WNV[i, t] × TEMP[i, t − 1], (7) WNV.FOR: WNV[i, t] × FOR[i] (or URB[i]), and (8) WNV.YR.FOR: WNV.YR[i, t] × FOR[i]. These interactions were chosen a priori to be of biological interest and relevance to our study goals of assessing land-use influences on WNV in crows. We explicitly examine both immediate changes to an intercept value [WNV] and temporally sustained changes to the trajectory of population changes [WNV.YR]. Because the land-cover variable is constant throughout our time series, we looked at both the interaction between WNV emergence and WNV persistence with forest or urban cover.
The specification of μ it also includes a nonlinear temporal trend component represented by the γ it s which were assumed a priori to follow a first-order autoregressive (AR(1)) model, a flexible structure for capturing smooth temporal trends. The AR(1) model implies that for i = 1, …, N and \( t = 1, \ldots ,T \),
We let
for \( i = 1, \ldots ,N \), and take the prior distribution on the autocorrelation parameter φ to be uniform on the interval from −1 to 1 in order to guarantee stationarity. We assume conditionally conjugate prior distributions for the remaining parameters determining the μ it s, for all p = 1,…,P covariate effects (including the intercept β 0 and slope β 1),
and for \( k = 1, \ldots ,O \), where O is the number of observers,
To account for residual spatial dependence among sites, we included a site-level spatial random effect η that was assumed to follow a multivariate normal distribution with mean zero and covariance matrix (\( \tau_{\eta }^{2} \Upsigma \)), where the (m, n) entry of Σ is an exponential function of the Euclidean distance between the (m, n)th pair of sites,
The parameters \( \tau_{\omega }^{2} \) and \( \tau_{\eta }^{2} \) were given independent vague (proper) inverse gamma prior distributions with parameters (0.01, 0.01). Variance parameters \( \tau_{\gamma }^{2} \) and \( \tau_{\alpha ,\rho }^{2} \) were fixed at 102. We used an informative prior on the rate of decline in correlation with distance, based on the maximum distance between two sites. We estimated model parameters by simulating from the joint posterior distribution of all unknown parameters using a Markov chain Monte Carlo (MCMC) algorithm implemented using the WinBUGS software (Lunn et al. 2000).
We initially fit versions of our model as described above using landscape-scale and route-scale land cover data in turn. We compared the initial models with submodels that removed (1) the temporal autoregressive term γ it , (2) the spatial correlation assumption for random site-level effects, (3) the land-use component and (4) the winter temperature component, sequentially. We compared each of these submodels to each other and to the full model using the deviance information criterion (DIC). DIC is a measure of how expanding or decreasing model structure changes the prediction accuracy of the model, with lower DIC values representing the preferred model (Spiegelhalter et al. 2002). The posterior predictive distribution for abundance at each site-year node was also compared to the raw count data using standard correlation techniques.
Results
We found that the emergence of WNV in the northeastern US has had considerable impact on the trajectory of crow populations, which declined annually between 1999 and 2004. Crows began to decline immediately after 1999 and continued to decline steadily until 2003, when abundance dropped dramatically (Fig. 2). The declines in crow abundances were significantly greater at a subset of urban sites where forest covered <35% of the landscape (1st quartile for forested area across all sites) and urban area was >11% of (3rd quartile for urban area across all sites) than at more rural sites (forest >68% of the landscape and urban area <5% of the landscape) (Fig. 3). This held up across all sites in our full model (Table 1). Crow abundances declined following the emergence of WNV, especially in landscapes that had lower proportions of forest cover and after above-average winter temperatures. Warmer winter temperatures generally had a positive influence on crow populations in the absence of WNV, while parameters describing the influence of total forest or urban cover did not differ from zero (Table 1).
American crow population series [1980–2008] from the posterior predictive distribution of mean annual counts across BBS sites. Bars denote 95% credible intervals for mean across sites. Only the bold-faced years were used to estimate parameters in Table 1
There was no change in the magnitude and significance of parameter estimates between the landscape-level and route-level models, although the DIC value for the landscape-level model was lowest (Table 2). In comparing DIC values between the full model and models that removed climate and land-cover components, we found that mean winter temperature values were more important to the overall model fit than were the land-cover variables (Table 2). Additionally, forest and development measures were not important predictors of crow abundance in either route-level or landscape-level models. Additionally, removing the temporal correlation term substantially increased DIC (reducing model fit), while inclusion of the spatial dependence structure had no apparent effect on DIC (Table 2), nor did it meaningfully change the parameter values.
Even after its initial emergence in an area, WNV continued to cause declines in crow abundances in subsequent years (WNV.YR < 0, Table 1). The negative effects associated with WNV were reduced in landscapes with more forest area. Crow abundances over landscapes with more forest suffered smaller declines in the years after WNV emergence (WNVYR.FOR > 0, Table 1). When the WNV.YR and WNVYR.FOR terms were removed from the full model the WNV estimate became negative, while credible intervals for WNV.FOR still included zero. As removing these parameters increased DIC, we considered the full model to be superior. Replacing FOR with URB in the WNV interaction components had an opposite and negative impact on the population trajectory, although overall model fit declined [posterior mean WNVYR.URB effect = −0.05 (−0.08, −0.04)]. This implies that for a 1% increase in urban land cover there is an additional 5% decline in the expected log abundance beyond the expected linear decline attributed to WNV in undeveloped areas (URB = 0)
Warmer winters were generally associated with increased crow abundances (TEMP > 0), with 5% higher annual abundance with each 1% increase in mean annual winter temperature. After WNV was present, however, warmer winters were associated with bigger crow declines in the following year (WNV.TEMP < 0). Mean winter temperatures and land-use measures were not independent variables (Fig. 4). Sites with more urban land cover were generally warmer than less urban sites within the same latitude (count ~61.0 + 0.27 × urban cover − 1.49 × latitude, R 2 = 0.90, p < 0.001).
a Mean winter temperatures are generally warmer at sites located in lower latitudes (−1.45 ± 0.04, p < 0.001). Average winter temperature across all sites in a given year is shown by the gray line, extending from 1994 on the right to 2004. b After accounting for the broad scale variation associated with latitude, urban land cover is a significant predictor of mean winter temperature (0.27 ± 0.04, p < 0.001)
Discussion
Our work supports the hypothesis that crows, like humans (e.g., Brown et al. 2008), have higher susceptibility to WNV in more urban and less forested landscapes (Table 1; Fig. 3). We also detected a significant influence of winter temperature on the inter-annual variability in crow declines associated with WNV. Crows were most vulnerable to WNV declines 1 year after warmer-than-average winters (Table 1). However, because winter temperature and the proportion of the landscape classified as urban were related (Fig. 4), we cannot disentangle the exact relationship between WNV impact in crows and winter climate or human land use. In our model exercise, the climate component does have a stronger influence on the short-term predictive ability measured by DIC, which could indicate that a temperature mechanism controls both the inter-annual and spatial heterogeneity in WNV incidence but could also be driven by the fact that we have more informative temperature data (which is available on an annual basis) relative to the one-time land cover observations. Still, whether due to the physical nature of anthropogenic habitat modification or to the associated changes in local temperature regimes (e.g., heat island), our results suggest that characteristics of the human-dominated landscape are associated with higher WNV risk in crow populations.
The mechanisms responsible for this association remain uncertain but could involve abiotic and biotic facilitation of WNV amplification (pathogen population growth), higher rates of WNV transmission, and/or greater inter-annual pathogen persistence. The warmer mean annual winter temperatures associated with development may support greater overwinter survival of infected mosquitoes and higher rates of vertical (transgenerational) transmission of WNV (e.g., Turell et al. 2001; Dohm et al. 2002), resulting in a head-start for epizootic amplification each spring and greater exposure of young, fledgling, and adult crows throughout the season. Similarly, residential landscapes may incorporate enough structural support in the form of sewer systems (Ruiz et al. 2007; LaBeaud et al. 2008) and storm water management (Wallace 2007) to facilitate rapid mosquito population growth together with a critical amount of vegetation and food availability to attract avian hosts (e.g., Ruiz et al. 2006; Brownstein et al. 2003).
Wildlife, including those organisms that carry and transmit disease, has adapted to anthropogenic landscapes and many now rely on suburban yards, parks, and water-management systems as habitat. In the United States, the total area covered by cities doubled between 1950 and 1990, although the population growth in urban centers only increased 10% (Livernash and Rodenburg 1998). The northeastern US encompasses the full gradient of rural habitat and anthropogenic disturbances (Woolmer et al. 2008). The sprawl of urban and suburban development is nestled within a regional landscape that includes agricultural fields, forests, and enough habitat diversity to support a wide-range of wildlife within close proximity to the sewers and backyard structures that support potential WNV vectors (Turell et al. 2005; Rochlin et al. 2008). These human-modified landscapes are likely to influence host and vector community ecology in ways that may affect pathogen transmission and even host susceptibility. Similar effects of forest loss and urban expansion have also been evident in the incidence of other zoonotic and vector-borne diseases [e.g., Malaria (Yasuoka and Levins 2007), Lyme disease (Allan et al. 2003), and chronic wasting disease (Farnsworth et al. 2005)].
Devising ways for researchers to detect and quantify pathogens in the wild across large and undefined spatial scales is an important step toward our ability to forecast and manage disease impacts. When the abundance and dispersal of a pathogen depends on wildlife hosts (zoonotic) or transmission by vectors, then understanding the ecology and population history of these species is crucial. Here we have shown that generalized and broad monitoring programs such as the North American Breeding Birds Survey can be valuable additions to disease-specific data protocols. Having these programs in place before pathogen introductions and epidemics occur is vital. Our work is also limited by some important data constraints associated with these broad monitoring/data programs. BBS bird counts are by definition made along secondary roads and there are no sites located at either rural or urban land-use extremes. Similarly, land cover could have changed dramatically between either the beginning or end of our study period and when the remote sensing data were collected (~2000) in ways that we could not capture at the same spatio-temporal scale of our count response variable.
The impacts of development and landscape modification across the globe are a dominant force in the emergence and persistence of many diseases. Historically, many facets of infrastructure development have acted to inhibit the spread and intensity of human disease (e.g., housing, water, and waste-management systems, and others). However, a landscape matrix of increasing fragmentation of forest, interspersed with agriculture, and urban and suburban residences may facilitate transmission dynamics for many of the emerging and reemerging pathogens that we see today. These challenges may be exacerbated by factors such as a changing climate, which has already and will continue to influence the seasonal and spatial dynamics of many pathogens (Paz and Albersheim 2008; Crowl et al. 2008).
References
Allan BF, Keesing F, Ostfeld RS (2003) Effect of forest fragmentation on Lyme disease risk. Conserv Biol 17:267–272
Allan BF, Langerhans RB, Ryberg WA, Landesman WJ, Griffin NW, Katz RS, Oberle BJ, Schutzenhofer MR, Smyth KN, de St Maurice A, Clark L, Crooks KR, Hernandez DE, McLean RG, Ostfeld RS, Chase JM (2009) Ecological correlates of risk and incidence of West Nile virus in the United States. Oecologia 158:699–708
Anderson RM, Fraser C, Ghani AC, Donnelly CA, Riley S, Ferguson NM, Leung GM, Lam TH, Hedley AJ (2004) Epidemiology, transmission dynamics and control of SARS: the 2002–2003 epidemic. Philos Trans R Soc Lond B Biol Sci 359:1091–1105
Andreadis TG, Anderson JF, Vossbrinck CR, Main AJ (2004) Epidemiology of West Nile virus in Connecticut: a five-year analysis of mosquito data 1999–2003. Vector Borne Zoonotic Dis 4:360–378
Banerjee S, Carlin B, AE Gelfand (2004) Hierarchical modeling and analysis for spatial data. Chapman & Hall, New York
Bradley CA, Altizer S (2007) Urbanization and the ecology of wildlife diseases. Trends Ecol Evol 22:95–102
Bradley CA, Gibbs SEJ, Altizer S (2008) Urban land use predicts West Nile virus exposure in songbirds. Ecol Appl 18:1083–1092
Brown DG, Johnson KM, Loveland TR, Theobald DM (2005) Rural land-use trends in the conterminous United States, 1950–2000. Ecol Appl 15:1851–1863
Brown H, Duik-Wasser M, Andreadis T, Fish D (2008) Remotely-sensed vegetation indices identify mosquito clusters of West Nile virus vectors in an urban landscape in the northeastern United States. Vector Borne Zoonotic Dis 8:197–206
Brownstein JS, Rosen H, Purdy D, Miller JR, Merlino M, Mostashari F, Fish D (2003) Spatial analysis of West Nile Virus: rapid risk assessment of an introduced vector-borne zoonosis. Vector Borne Zoonotic Dis 3:155 (vol 2, p 157, 2002)
CDC (2009) Centers for Disease Control and Prevention, West Nile Virus Home, Ecology and Virology, Vertebrate Ecology, 28 April 2009
Crowl TA, Crist TO, Parmenter RR, Belovsky G, Lugo AE (2008) The spread of invasive species and infectious disease as drivers of ecosystem change. Front Ecol Environ 6:238–246
Darsie R, Ward R (2005) Identification and geographical distribution of the mosquitoes of North America north of Mexico. University of Florida Press, Gainesville
DeGroote JP, Sugumaran R, Brend SM, Tucker BJ, Bartholomay LC (2008) Landscape, demographic, entomological, and climatic associations with human disease incidence of West Nile virus in the state of Iowa, USA. Int J Health Geogr 7:19
Dohm DJ, O’Guinn ML, Turell MJ (2002) Effect of environmental temperature on the ability of Culex pipiens (Diptera: Culicidae) to transmit West Nile virus. J Med Entomol 39:221–225
Farnsworth ML, Wolfe LL, Hobbs NT, Burnham KP, Williams ES, Theobald DM, Conner MM, Miller MW (2005) Human land use influences chronic wasting disease prevalence in mule deer. Ecol Appl 15:119–126
Gibbs SEJ, Wimberly MC, Madden M, Masour J, Yabsley MJ, Stallknecht DE (2006) Factors affecting the geographic distribution of West Nile virus in Georgia, USA: 2002–2004. Vector Borne Zoonotic Dis 6:73–82
Gomez A, Kilpatrick AM, Kramer LD, Dupuis AP, Maffei JG, Goetz SJ, Marra PP, Daszak P, Aguirre AA (2008) Land use and West Nile virus seroprevalence in wild mammals. Emerg Infect Dis 14:962–965
Grimm NB, Faeth SH, Golubiewski NE, Redman CL, Wu JG, Bai XM, Briggs JM (2008) Global change and the ecology of cities. Science 319:756–760
Jones KE, Patel NG, Levy MA, Storeygard A, Balk D, Gittleman JL, Daszak P (2008) Global trends in emerging infectious diseases. Nature 451:990–993
Kilpatrick AM, Kramer LD, Campbell SR, Alleyne EO, Dobson AP, Daszak P (2005) West Nile virus risk assessment and the bridge vector paradigm. Emerg Infect Dis 11:425–429
Kilpatrick AM, Chmura AA, Gibbons DW, Fleischer RC, Marra PP, Daszak P (2006a) Predicting the global spread of H5N1 avian influenza. Proc Natl Acad Sci USA 103:19368–19373
Kilpatrick AM, Daszak P, Jones MJ, Marra PP, Kramer LD (2006b) Host heterogeneity dominates West Nile virus transmission. Proc R Soc B Biol Sci 273:2327–2333
Kilpatrick AM, Kramer LD, Jones MJ, Marra PP, Daszak P (2006c) West Nile virus epidemics in North America are driven by shifts in mosquito feeding behavior. PLoS Biol 4:606–610
Kilpatrick AM, LaDeau SL, Marra PP (2007) Ecology of west Nile virus transmission and its impact on birds in the western hemisphere. Auk 124:1121–1136
Komar N, Langevin S, Hinten S, Nemeth N, Edwards E, Hettler D, Davis B, Bowen R, Bunning M (2003) Experimental infection of North American birds with the New York 1999 strain of West Nile virus. Emerg Infect Dis 9:311–322
Komar N, Panella NA, Langevin SA, Brault AC, Amador M, Edwards E, Owen JC (2005) Avian hosts for West Nile virus in St. Tammany Parish, Louisiana, 2002. Am J Trop Med Hyg 73:1031–1037
LaBeaud AD, Gorman AM, Koonce J, Kippes C, McLeod J, Lynch J, Gallagher T, King CH, Mandalakas AM (2008) Rapid GIS-based profiling of West Nile virus transmission: defining environmental factors associated with an urban-suburban outbreak in Northeast Ohio, USA. Geospa Health 2:215–225
LaDeau SL, Kilpatrick AM, Marra PP (2007) West Nile virus emergence and large-scale declines of North American bird populations. Nature 447:710–713
LaDeau SL, Marra PP, Kilpatrick AM, Calder CA (2008) West Nile virus revisited: consequences for North American ecology. Bioscience 58:937–946
Lanciotti RS, Roehrig JT, Deubel V, Smith J, Parker M, Steele K, Crise B, Volpe KE, Crabtree MB, Scherret JH, Hall RA, MacKenzie JS, Cropp CB, Panigrahy B, Ostlund E, Schmitt B, Malkinson M, Banet C, Weissman J, Komar N, Savage HM, Stone W, McNamara T, Gubler DJ (1999) Origin of the West Nile virus responsible for an outbreak of encephalitis in the northeastern United States. Science 286:2333–2337
Liu JG, Daily GC, Ehrlich PR, Luck GW (2003) Effects of household dynamics on resource consumption and biodiversity. Nature 421:530–533
Liu A, Lee V, Galusha D, Slade M, Diuk-Wasser M, Andreadis T, Scotch M, Rabinowitz P (2009) Risk factors for human infection with West Nile virus in Connecticut: a multi-year analysis. Int J Health Geogr 8:67
Livernash R, Rodenburg E (1998) Population change, resources, and the environment. Popul Bull 53(1)
Loss SR, Hamer GL, Walker ED, Ruiz MO, Goldberg TL, Kitron UD, Brawn JD (2009) Avian host community structure and prevalence of West Nile virus in Chicago, Illinois. Oecologia 159:415–424
Lunn DJ, Thomas A, Best N, Spiegelhalter D (2000) WinBUGS—a Bayesian modelling framework: concepts, structure, and extensibility. Stat Comput 10:325–337
Millennium Ecosystem Assessment (2005) Ecosystems and human well-being: synthesis report. Island Press, Washington, DC
Myers SS, Patz JA (2009) Emerging threats to human health from global environmental change. Annu Rev Environ Resour 34:223–252
Paz S, Albersheim I (2008) Influence of warming tendency on Culex pipiens population abundance and on the probability of West Nile Fever outbreaks (Israeli case study: 2001–2005). Ecohealth 5:40–48
Reed LM, Johansson MA, Panella N, McLean R, Creekmore T, Puelle R, Komar N (2009) Declining mortality in American crow (Corvus brachyrhynchos) following natural West Nile virus infection. Avian Dis 53:458–461
Reisen WK, Carroll BD, Takahashi R, Fang Y, Garcia S, Martinez VM, Quiring R (2009) Repeated West Nile Virus Epidemic Transmission in Kern County, California, 2004-2007. J Med Entomol 46:139–157.
Rochlin I, Harding K, Ginsberg HS, Campbell SR (2008) Comparative analysis of distribution and abundance of West Nile and eastern equine encephalomyelitis virus vectors in Suffolk County, New York, using human population density and land use/cover data. J Med Entomol 45:563–571
Ruiz MO, Brown WM, Brawn JD, Hamer GL, Kunkel KE, Loss SR, Walker ED, Kitron UD (2006) Spatiotemporal patterns of precipitation and West Nile virus in Chicago, Illinois, 2002–2005 and implications for surveillance. Am J Trop Med Hyg 75:269–270
Ruiz MO, Walker ED, Foster ES, Haramis LD, Kitron UD (2007) Association of West Nile virus illness and urban landscapes in Chicago and Detroit. Int J Health Geogr 6:10
Sanderson EW, Jaiteh M, Levy MA, Redford KH, Wannebo AV, Woolmer G (2002) The human footprint and the last of the wild. Bioscience 52:891–904
Spiegelhalter DJ, Best NG, Carlin BR, van der Linde A (2002) Bayesian measures of model complexity and fit. J R Stat Soc Series B Stat Methodol 64:583–616
Sugumaran R, Larson SR, DeGroote JP (2009) Spatio-temporal cluster analysis of county-based human West Nile virus incidence in the continental United States. Int J Health Geogr 8:19
Taylor LH, Latham SM, Woolhouse MEJ (2001) Risk factors for human disease emergence. Philos Trans R Soc Lond B Biol Sci 356:983–989
Turell MJ, Sardelis MR, Dohm DJ, O’Guinn ML (2001) Potential North American vectors of West Nile virus. In: West Nile virus: detection, surveillance, and control. New York Acad Sciences, New York, pp 317–324
Turell MJ, Dohm DJ, Sardelis MR, O’Guinn ML, Andreadis TG, Blow JA (2005) An update on the potential of North American mosquitoes (Diptera: Culicidae) to transmit West Nile virus. J Med Entomol 42:57–62
USGS Patuxent Wildlife Research Center (2008) North American Breeding Bird Survey ftp data set, version 2007.2. ftp://ftpext.usgs.gov/pub/er/md/laurel/BBS/datafiles/.
Wallace J (2007) Stormwater management and mosquito ecology: a systems-based approach towards an integrative management strategy. Stormwater 8:20–46
Ward MP, Raim A, Yaremych-Hamer S, Lampman R, Novak RJ (2006) Does the roosting behavior of birds affect transmission dynamics of West Nile virus? Am J Trop Med Hyg 75:350–355
Wilcox BA, Gubler DJ (2005) Disease ecology and the global emergence of zoonotic pathogens. Environ Health Prev Med 10:263–272
Wilcox BR, Yabsley MJ, Ellis AE, Stallknecht DE, Gibbs SEJ (2007) West Nile virus antibody prevalence in American crows (Corvus brachyrhynchos) and fish crows (Corvus ossifragus) in Georgia, USA. Avian Dis 51:125–128
Woolmer G, Trombulak SC, Ray JC, Doran PJ, Anderson MG, Baldwin RF, Morgan A, Sanderson EW (2008) Rescaling the human footprint: a tool for conservation planning at an ecoregional scale. Landsc Urban Plan 87:42–53
Yaremych SA, Novak RJ, Raim AJ, Mankin PC, Warner RE (2004) Home range and habitat use by American crows in relation to transmission of West Nile Virus. Wilson Bull 116:232–239
Yasuoka J, Levins R (2007) Impact of deforestation and agricultural development on anopheline ecology and malaria epidemiology. Am J Trop Med Hyg 76:450–460
Acknowledgments
The authors wish to thank the dedicated volunteers that keep the Breeding Bird Survey active in North America. SLL thanks Dr. Zen Kawabata for the opportunity to present at the 2008 Kyoto symposium on ‘Environmental Change, Pathogens and Human Linkages’. PJD was supported by The Nature Conservancy’s Great Lakes Fund for Partnership in Conservation Science and Economics. SLL was supported by the National Science Foundation (NSF) Program in Bioinformatics for part of this research. This paper is a contribution to the program of the Cary Institute of Ecosystem Studies.
Author information
Authors and Affiliations
Corresponding author
About this article
Cite this article
LaDeau, S.L., Calder, C.A., Doran, P.J. et al. West Nile virus impacts in American crow populations are associated with human land use and climate. Ecol Res 26, 909–916 (2011). https://doi.org/10.1007/s11284-010-0725-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11284-010-0725-z