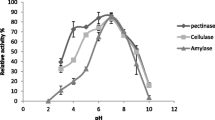
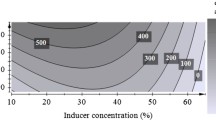
Abstract
Enzymes are biocatalysts that are widely used in different industries and generate billions of dollars annually. With the advancement of biotechnology, new enzymatic sources are being evaluated, especially microbial ones, in order to find efficient producers. Endophytic fungi are promising sources of biomolecules; however, Amazonian species are still poorly studied as to their enzymatic production potential. In this sense, the production of hydrolases (amylases, lipases, cellulases and pectinases) was evaluated in endophytic fungi isolated from the leaves, roots and stems of açai palms (Euterpe precatoria). A qualitative test was carried out to detect the enzymatic synthesis in each isolate, and the most promising ones were cultivated using submerged fermentation. The enzyme extracts were quantified to determine those with the greatest activity. Cellulolytic and amylolytic extracts showed the highest enzymatic activities and were partially characterized. Among 50 isolates, 82.9% produced pectinase, 58.5% produced cellulase, 31.7% produced amylase, and 12.2% produced lipase. Penicillium sp. L3 was the best producer of amylase and Colletotrichum sp. S1 was the best producer of cellulase in liquid medium cultivation. The amylolytic extract showed the highest enzymatic activity at pH 8.0 and 45 °C, and the cellulolytic extract at pH 5.0 and 35 °C. The cellulase and amylase produced by the endophytes had their molecular masses estimated between 38 and 76 kDa. These results indicate that endophytic fungi from the açai palm can be used as a new source of hydrolytic enzymes, which can be applied in numerous biotechnological processes.






Similar content being viewed by others
Data availability
Not applicable.
Code availability
Not applicable.
References
Abdel-Azeem AM, Abdel-Azeem MA, Abdul-Hadi SY, Darwish AG (2019) Aspergillus: biodiversity, ecological significances, and industrial applications. In: Mishra S, Singh S, Gupta A, Yadav A (eds) Recent advancement in white biotechnology through fungi. Fungal Biology. Springer, Cham. https://doi.org/10.1007/978-3-030-10480-1_4
Ahmed S, Bashir A, Saleem H, Saadia M, Jamil A (2009) Production and purification of cellulose-degrading enzymes from a filamentous fungus Trichoderma harzianum. Pak J Bot 41:1411–1419
Almeida MN, Falkoski DL, Guimarães VM, Ramos HJO, Visser EM, Maitan-Alfenas GP, Rezende ST (2013) Characteristics of free endoglucanase and glicosydases multienzyme complex from Fusarium verticillioides. Bioresour Technol 143:413–422. https://doi.org/10.1016/j.biortech.2013.06.021
Anand T, Bhaskaran R, Raguchander T, Karthikeyan G, Rajesh M, Senthilraja G (2008) Production of cell wall degrading enzymes and toxins by Colletotrichum capsici and Alternaria alternata causing fruit rot of chillies. J Plant Prot Res 48:437–451. https://doi.org/10.2478/v10045-008-0053-2
Baltas N, Dincer B, Ekinci AP, Kolayli S, Adiguzel A (2016) Purification and characterization of extracellular α-amylase from a thermophilic Anoxybacillus thermarum A4 strain. Braz Arch Biol Technol 59:1–14. https://doi.org/10.1590/1678-4324-2016160346
Batista BN, Silva IR, Rapôso NVM (2018) Isolation and antimicrobial activity evaluation of endophytic fungi of açaizeiro. Rev Fitos 12:161–174. https://doi.org/10.5935/2446-4775.20180015
Benoliel B, Torres FA, Moraes LM (2013) A novel promising Trichoderma harzianum strain for the production of a cellulolytic complex using sugarcane bagasse in natura. Springerplus 2:656. https://doi.org/10.1186/2193-1801-2-656475
Bezerra JDP, Santos MGS, Svedese VM, Lima DMM, Fernandes MJS, Paiva LM, Souza-Motta CM (2012) Richness of endophytic fungi isolated from Opuntia fícus-indica Mill. (Cactaceae) and preliminar screening for enzyme production. World J Microbiol Biotechnol 28:1989–1995. https://doi.org/10.1007/s11274-011-1001-2
Bilal M, Iqbal HMN (2019) Chemical, physical, and biological coordination: an interplay between materials and enzymes as potential for immobilization. Coordin Chem Rev 388:1–23. https://doi.org/10.1016/j.ccr.2019.02.024
Bon EPS, Ferrara MA, Corvo ML, Vermelho AB, Paiva CLA, Alencastro RB, Coelho RRR (2008) Enzimas em biotecnologia: produção, aplicações e mercado. Interciência, Rio de Janeiro
Boratyński F, Szczepańska E, Grudniewska A, Gniłka R, Olejniczak T (2018) Improving of hydrolase biosythesis by solid-state fermentation of Penicillium camemberti on rapeseed cake. Sci Rep 8:10157. https://doi.org/10.1038/s41598-018-28412-y
Carrasco M, Alcaíno J, Cifuentes V, Baeza M (2017) Purification and characterization of a novel cold adapted fungal glucoamylase. Microb Cell Fact 16:75. https://doi.org/10.1186/s12934-017-0693-x
Choi JM, Han SS, Kim HS (2015) Industrial applications of enzyme biocatalysis: current status and future aspect. Biotechnol Adv 33:1443–1454. https://doi.org/10.1016/j.biotechadv.2015.02.014
Cortez DV, Castro HF, Andrade GSS (2017) Potential catalytic of mycelium-bound lipase of filamentous fungi in biotransformation processes. Quim Nova 40:85–96. https://doi.org/10.21577/0100-4042.20160163
Cunha JRB, Santos FCP, Assis FGV, Leal PL (2016) Cultivation of Penicillium spp. in soybean crop residues for production of cellulase, protease and amylase. Rev Ceres 63:597–504. https://doi.org/10.1590/0034-737x201663050002
Cysneiros CSS, Ferreira RN, Oliveira MA, Favoretto AO, Arnhold E, Ulhoa CJ (2013) Production, characterization and evaluation of fibrolytic enzymes on digestibility of forage maize. Cienc Anim Brasileira 14:426–435. https://doi.org/10.5216/cab.v14I4.19491
Damodaran S, Parkin KL, Fanema OR (2008) Química de alimentos de Fennema, 4th edn. Artmed, Porto Alegre
Dar GH, Kamili AN, Nazir R, Bandh SA, Jan TR, Christi MZ (2015) Enhanced production of α-amylase by Penicillium chrysogenum in liquid culture by modifying the process parameters. Microb Pathogenesis 88:10–15. https://doi.org/10.1016/j.micpath.2015.07.016
Delatorre AB, Ladeira SA, Andrade MVV, Barbosa JB, Martins MLL (2010) Micro-organismos termofílicos e enzimas termoestáveis de importância comercial. Perspectivas 4:132–145
El-Gendy MMAA (2012) Production of glucoamylase by marine endophytic Aspergillus sp. JAN-25 under optimized solid-state fermentation conditions on agro residues. Aust J Basic Appl Sci 6:4154
Espinel E, López E (2009) Purification and characterization of a-amylase from Penicillium commune produced by solid state fermentation. Rev Colomb Qui 38:191–208
Fadiji AE, Babalola OO (2020) Elucidating mechanisms of endophytes used in plant protection and other bioactivities with multifunctional prospects. Front Bioeng Biotechnol 8:467. https://doi.org/10.3389/fbioe.2020.00467
Gopinath SCB, Anbu P, Arshad MKM, Lakshmipriya T, Voon CH, Hashim U, Chinni S (2017) Biotechnological process in microbial amylase production. BioMed Res Int 2017:1272193. https://doi.org/10.1155/2017/1272193
Grand View Research (2020) Enzyme’s market size, share & trends analysis report by application (industrial enzymes, specialty enzymes), by product (carbohydrase, proteases, lipases), by source, by region, and segment forecasts, 2020–2027. https://www.grandviewresearch.com/industry-analysis/ enzymes-industry. Accessed 15 April 2021
Gulhane PA, Gomashe AV, Patne M (2016) Endophytic fungi: a source of novel enzymic antioxidants and biologically active secondary metabolites. Int J Recent Sci Res 7:8226–8231
Hankin L, Anagnostakis SL (1975) The use of solid media for detection of enzyme production by fungi. Mycologia 67:597–607. https://doi.org/10.2307/3758395
Hegde SV, Ramesha A, Srinvas C (2011) Optimization of amylase production from an endophytic fungi Discosia sp. isolated from Calophyllum inophyllum. J Agric Technol 7:805–813
Jahan N, Shahid F, Aman A, Mujahid TY, Qader SAU (2017) Utilization of agro waste pectin for the production of industrially important polygalacturonase. Heliyon 3:e00330. https://doi.org/10.1016/j.heliyon.2017.e00330
Janec̆ek S̆ (1997) α-amylase family: molecular biology and evolution. Prog Biophys Mol Biol 67:67–97. https://doi.org/10.1016/S0079-6107(97)00015-1
Kanti A, Sudiana IM (2017) Ethanol production using cellulolytic, xylanolytic and fermentative yeast on cassava waste. In: 1st SATREPS Conference Proceedia, p 39–52
Li N, Zong M-H (2010) Lipases from the genus Penicillium: production, purification, characterization and applications. J Mol Catal B 66:43–54. https://doi.org/10.1016/j.molcatb.2010.05.004
Lopez AMQ, Pereira DST (2010) Interaction between Colletotrichum gloeosporioides and ecotypes of sugar apple. Bragantia 69:105–114. https://doi.org/10.1590/S0006-87052010000100015
Marchi CE, Dornelas FC, Araújo GLR, Rodrigues FL, Freitas BM, Trentin RA, Jerba VF (2009) Pectinolytic activity of Colletotrichum gloeosporioides and its relationship with aggressiveness to Stylosanthes spp. Bragantia 68:423–433. https://doi.org/10.1590/S0006-87052009000200017
Marques NP, Pereira JC, Gomes E, Da Silva R, Araújo AR, Ferreira H, Rodrigues A, Dussán KJ, Bocchini DA (2018) Cellulases and xylanases production by endophytic fungi by solid state fermentation using lignocellulosic substrates and enzymatic saccharification of pretreated sugarcane bagasse. Ind Crop Prod 122:66–75. https://doi.org/10.1016/j.indcrop.2018.05.022
Matias RR, Sepúlveda AMG, Batista BN, De Lucena JMVM, Albuquerque PM (2021) Degradation of Staphylococcus aureus biofilm using hydrolytic enzymes produced by Amazonian endophytic fungi. Appl Biochem Biotech. https://doi.org/10.1007/s12010-021-03542-8
Mendes MMGS, Pereira SA, Oliveira RL, Silva LAO, Duvoisin Junior S, Albuquerque PM (2015) Screening of Amazon fungi for the production of hydrolytic enzymes. Afr J Microbiol Res 9:741–748. https://doi.org/10.5897/AJMR2014.7158
Miller GL (1959) Use of dinitrosalicyllic acid for determination of reducing sugar. Anal Chem 11:426–428. https://doi.org/10.1021/ac60147a030
Moran LA, Horton HR, Scrimgeour KG, Perr MD (2013) Principles of biochemistry, 5th edn. Pearson, New York
Narra M, Dixit G, Divecha J, Kumar K, Madamwar D, Shah AR (2014) Production, purification and characterization of a novel GH 12 family endoglucanase from Aspergillus terreus and its application in enzymatic degradation of delignified rice straw. Int Biodeterior Biodegrad 88:150–161. https://doi.org/10.1016/j.ibiod.2013.12.016
Nascimento CS, Dos Santos VL, Andrade MHC (2015) Análise da produção de protease e lipase por fungos filamentosos isolados do fruto da macaúba (Acrocomia aculeata (Jacq) Lood. ex Mart). Blucher Chem Eng Proc 1:336–343. https://doi.org/10.5151/chemeng-cobeq2014-0222-26465-171722
Norouzian D, Akbarzadeh A, Scharer JM, Moo Young M (2006) Fungal glucoamylases. Biotechnol Adv 24:80–85. https://doi.org/10.1016/j.biotechadv.2005.06.003
Oliveira AN, Flor NS, Oliveira LA (2010) Influence of pH and temperature on amylolytic activity of rhizobia isolated from Amazonian soils. Acta Amazon 40:401–404. https://doi.org/10.1590/S0044-59672010000200019
Onofre SB, Steilmnn P, Bertolini J, Rotta D, Sartori A, Kagimura FY, Groff AS, Mazzali L (2011) Amylolytic enzymes produced by the fungus Colletotrichum gloeosporioides in rice semi-solid fermentation. J Yeast Fungal Res 2:28–32. https://doi.org/10.5897/JYFR11.003
Orlandelli RC, Almeida TT, Alberto RN, Polonio JC, Azevedo JL, Pamphile JA (2015) Antifungal and proteolytic activities of endophytic fungi isolated from Piper hispidum Sw. Braz J Microbiol 46:359–366. https://doi.org/10.1590/S1517-838246220131042
Peeran MF, Kuppusami P, Thiruvengadam R (2014) Pathogenesis of Colletotrichum lindemuthianum the incitant of anthracnose disease in beans mediated by macerating enzymes. The Bioscan 9:295–300
Robinson PK (2015) Enzymes: principles and biotechnological applications. Essays Biochem 59:1–41. https://doi.org/10.1042/bse0590001
Rodrigues MLF, Silva EA, Borba CE, Oliveira ACD, Kruger C, Raimundo RW, Silva LP, Rodriques MF, Stuani BT (2015) Hydrolytic enzymes production by the endophytic fungus Penicillium sp. isolated from the sheets of Ricinus communis L. Rev Bras Energias Renováveis 4:129–145. https://doi.org/10.5380/rber.v4i2.42839
Romão AS, Sposito MB, Andreote FD, Azevedo JL, Araujo WL (2011) Enzymatic differences between the endophyte Guignardia mangiferae (Botryosphaeriacceae) and the citrus pathogen G. citricarpa. Genet Mol Res 10:243–252. https://doi.org/10.4238/vol10-1gmr952
Roosdiana A, Prasetyawan S, Mahdi C, Sutrisno S (2013) Production and characterizarion of Bacillus firmus pectinase. J Pure Appl Chem Res 2:35–41. https://doi.org/10.21776/ub.jpacr.2013.002.01.111
Sagu ST, Nso EJ, Hornann T, Kapseu C, Rawel HM (2015) Extraction and purification of beta-amylase from stems of Abrus precatorius by three phase partitioning. Food Chem 183:144–153. https://doi.org/10.1016/j.foodchem.2015.03.028
Sandri IG, Lorenzoni CMT, Fontana RC, Silveira MM (2013) Use of pectinases produced by a new strain of Aspergillus niger for the enzymatic treatment of apple and blueberry juice. Food Sci Technol 51:469–475. https://doi.org/10.1016/j.lwt.2012.10.015
Schneider WDH, Dos Reis L, Camassola M, Dillon AJP (2014) Morphogenesis and production of enzymes by Penicillium echinulatumin response to different carbon sources. BioMed Res Int 2014:1–10. https://doi.org/10.1155/2014/254863
Shubha J, Srinivas C (2017) Diversity and extracellular enzymes of endophytic fungi associated with Cymbidium aloifolium L. Afr J Biotechnol 16:2248–2258. https://doi.org/10.5897/AJB2017.16261
Silva RLO, Luz JS, Silveira EB, Cavalcante UMT (2006) Endophytic fungi of Annona spp.: isolation, enzymatic characterization of isolates and plant growth promotion in Annona squamosa L. seedlings. Acta Bot Bras 20:649–655. https://doi.org/10.1590/S0102-33062006000300015
Sindhu R, Suprabha GN, Shashidhar S (2011) Purification and characterization of α-amylase from Penicillium janthinellum (NCIM 4960) and its application in detergent industry. Biotechnol Bioinform Bioeng 1:25–32
Singh R, Kumar M, Mittal A, Mehta PK (2016) Microbial enzymes: industrial progress in 21st century. 3 Biotech 6:174. https://doi.org/10.1007/s13205-016-0485-8
Soro RY, Diopoh JK, Willemot RM, Combes D (2007) Enzymatic synthesis of polyglucosylfructosides from sucrose alone α-glucosidase isolated from the digestive juice of Archachatina ventricose (Achatinideae). Enzyme Microb Technol 42:4451. https://doi.org/10.1016/j.enzmictec.2007.07.024
Souza HQ, Oliveira LA, Andrade JS (2008) Screening of basidiomycetes from amazonia for the production of biotechnological interest enzymes. Ciencia Tecnol Alime 28:116–124. https://doi.org/10.1590/S0101-20612008000500019
Vasconcelos NM, Pinto GAS, Aragão FAS (2013) Determination of reducing sugars by 3,5-dinitrosalicylic acid: historical development of the method and establishment of a protocol to the Laboratory of Bioprocess. Boletim de Pesquisa e Desenvolvimento 88. Embrapa Agroindústria Tropical, Fortaleza
Zhou H-Y, Zhou J-B, Yi X-N, Wang Y-M, Xue Y-P, Chen D-S, Cheng X-P, Li M, Wang H-Y, Chen K-Q, Liu Z-Q, Zheng Y-G (2020) Heterologous expression and biochemical characterization of a thermostable endo-β-1,4-glucanase from Colletotrichum orchidophilum. Bioproc Biosyst Eng 44:67–79. https://doi.org/10.1007/s00449-020-02420-7
Zimbardi AL, Sehan C, Meleiro LP, Souza FHM, Masui DC, Nozawa MSF, Guimarães LHS, Jorge JA, Furriel RPM (2013) Optimization of β-glucosidase, β-xylosidase and xylanase production by Colletotrichum graminicolaunder solid-state fermentation and application in raw sugarcane trash saccharification. Int J Mol Sci 14:2875–2902. https://doi.org/10.3390/ijms14022875
Acknowledgements
The authors thank the Coordenação de Aperfeiçoamento de Pessoal de Nível Superior—CAPES and the Fundação de Amparo à Pesquisa do Estado do Amazonas—FAPEAM for the financial support of this study, the Chemistry Applied to Technology Research Group for the infra-structure availability, and Mr. Matthew Miller for the revision of the English language.
Funding
This work was supported by a research grant from the Coordenação de Aperfeiçoamento de Pessoal de Nível Superior, CAPES (Pro-Amazônia Program, Project 52, and Financial Code 001), and by the Fundação de Amparo à Pesquisa do Estado do Amazonas—FAPEAM.
Author information
Authors and Affiliations
Contributions
BNB performed the fungal hydrolases production, carried out the enzymatic activity testing, the enzymatic extracts characterization and participated in data analysis and drafted the manuscript. RRM carried out the initial screening of Euterpe precatoria endophytic fungi that produced hydrolases and helped in the hydrolases production in liquid medium. RLO participated in the design of the study, guided the enzymatic extracts characterization, participated in the analysis and interpretation of data and contributed to the review of the manuscript. PMA conceived the study, and participated in its design and coordination, carried out the analysis and interpretation of hydrolases production, and revised the manuscript. All authors read and approved the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no competing interests.
Ethical approval
Not applicable.
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Batista, B.N., Matias, R.R., Oliveira, R.L.e. et al. Hydrolytic enzyme production from açai palm (Euterpe precatoria) endophytic fungi and characterization of the amylolytic and cellulolytic extracts. World J Microbiol Biotechnol 38, 30 (2022). https://doi.org/10.1007/s11274-021-03217-w
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11274-021-03217-w