Abstract
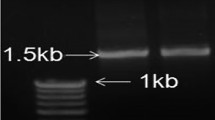
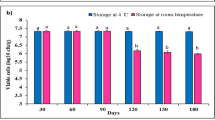
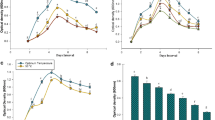
Wilt disease of soybean caused by a very common soil-borne fungus, Fusarium oxysporum is one of the most destructive diseases of the crop. The aim of the present study was to characterize plant growth-promotion activities and induced resistance of a rhizobacterial strain for the soybean plant against F. oxysporum. Rhizobacterium strain SJ-5 exhibited plant growth-promotion characteristics and antagonistic activity against the test pathogen on dual plate assay. It was identified as a Carnobacterium sp. A 950 bp PCR product was amplified from Carnobacterium sp. strain SJ-5, using zwittermicin A self-resistance gene-specific primers (zmaR). The strain produced indole 3-acetic acid (19 μg/ml) in the presence of salt stress and exhibited growth in Dworkin and Foster salt medium amended with 1-aminocyclopropane-1-carboxylate (ACC) through ACC deaminase activity (277 nmol/mg/h) as compared to the control. Strain seeds treated with the strain significantly enhanced the quorum of healthy plants after challenge inoculation at 14 days after seeding. An increase in the activity of stress enzymes after challenge inoculation with the test pathogen is reported. Treatment with the bacterium resulted in an increase in the chlorophyll content in the leaves in comparison with challenge-inoculated plants.


Similar content being viewed by others
References
Allain CC, Poon LC, Chan CSG, Richmond W, Fu PC (1974) Enzymatic determination of total serum cholesterol. Clin Chem 20:470–475
Axelrod B, Cheesbrough TM, Laakso S (1981) Lipoxygenase from soybean. Methods Enzymol 71:441–451
Benhamou N, Kloepper JW, Tuzun S (1998) Induction of resistance against Fusarium wilt of tomato by combination of chitosan with an endophytic bacterial strain: ultrastructure and cytochemistry of the host response. Planta 204:153–168
Broetto F, Marchese JA, Leonardo M, Regina M (2005) Fungal elicitor-mediated changes in polyamine content, phenylalanine ammonia-lyase and peroxidase activities in bean cell culture. Gen Appl Plant Physiol 31:235–246
Choudhary DK (2012) Microbial rescue to plant under habitat-imposed abiotic and biotic stresses. Appl Microbiol Biotechnol 96:1137–1155
Choudhary DK, Sharma KP, Gaur RK (2011) Biotechnological perspectives of microbes in agro-ecosystems. Biotechnol Lett 33:1905–1910
Croft KPC, Voisey CR, Slusarenkl AJ (1990) Mechanism of hypersensitive cell collapse: correlation of increase lipoxygenase activity with membrane damage in leaves of Phaseolus vulgaris cv. Red Maxican inoculated with avirulent race/cells of Pseudomonas syringae pv. Phaseolicola. Physiol Mol Plant Pathol 36:49–62
Dimpka C, Weinand T, Ash F (2009) Plant–rhizobacteria interactions alleviate abiotic stress conditions. Plant Cell Environ 32:1682–1694
El-Abady MI, Seadh SE, Attia AN, El-Saidy Aml EA (2008). Impact of foliar fertilization and its time of application on yield and seed quality of soybean. The 2th field crops conference, FCRI, AV, Giza, Egypt
Fayzalla EA, El-Barougy E, El-Rayes MM (2009) Control of soil-borne pathogenic fungi of soybean by biofumigation with mustard seed meal. J Appl Sci 9:2272–2279
Glick B, Patten C, Holguin G, Penrose D (1999) Biochemical and genetic mechanisms used by plant growth promoting bacteria. Imperial Col, London p 267
Gutierrez-Manero FJ, Ramos-Solano B, Probanza A, Mehouachi J (2001) The plant growth-promoting rhizobacteria Bacillus pumilus and Bacillus licheniformis produce high amounts of physiologically active gibberlins. Physiol Plant 111:206–211
Hammerschmidt R (1999) Induced disease resistance: how do induced plants stop pathogens? Physiol Mol Plant Pathol 55:77–84
Hashem EA, Abdalla HE, Hussein YA, Abd-Elnabi MA (2009) In vitro selection of soybean callus resistant to Fusarium oxysporum metabolites. Res J Agric Biol Sci 5(4):588–596
Hass D, Keel C, Reimann C (2002) Signal transduction in plant beneficial rhizobacteria with biocontrol properties. Ant van Leeuwen 81:385–395
Hiscox JD, Israelstam GF (1979) A method for the extraction of chlorophyll from leaf tissue without maceration. Can J Bot 57:1332–1334
Honna M, Shimomura T (1978) Metabolism of 1-aminocyclopropane-1-carboxylic acid. Agric Biol Chem 42:1825–1831
Liang X, Dron M, Schmid J, Dixon R, Lamb C (1989) Developmental and environmental regulation of a phenylalanine ammonia-lyase-β-glucuronidase gene fusion in transgenic tobacco plants. Proc Natl Acad Sci 86:9284–9288
Mayak S, Tirosh T, Glick B (1999) Effect of wild-type and mutant plant growth-promoting rhizobacteria on the rooting of mung bean cuttings. Plant Growth Regul 18:49–53
Pascholati SF, Nicholson RL, Butler DL (1986) Phenylalanine ammonia-lyase activity and anthocyanin accumulation in wounded maize mesocotyls. J Phytopathol 115:165–172
Patten CL, Glick BR (2002) Role of Pseudomonas putida indoleacetic acid in development of the host plant root system. Appl Environ Microbiol 68:3795–3801
Perret X, Broughton WJ (1998) Rapid identification of Rhizobium strains by targeted PCR fingerprinting. Plant Soil 204:21–34
Podile AR, Lakshmi VDV (1998) Seed bacterization with Bacillus subtilis increases phenylalanine ammonia-lyase and reduces the incidence of fusarium wilt of pigeonpea. J Phytopathol 146:255–259
Ray H, Douches DS, Hammerschmidt R (1998) Transformation of potato with cucumber peroxidase: expression and disease response. Physiol Mol Plant Pathol 53:93–103
Sharma A, Johri BN (2003) Combat of iron-deprivation through a plant growth promoting fluorescent Pseudomonas strain GRP3 in mung bean. Microbiol Res 158:77–81
Sharma A, Johri BN, Sharma AK, Glick BR (2003) Plant growth-promoting bacterium Pseudomonas sp. strain GRP3 influences iron acquisition in mung bean (Vigna radiata L. Wilzeck). Soil Biol Biochem 35:887–894
Sharma A, Jansen R, Nimtez M, Johri BN, Wray V (2007a) Rhamnolipids from the rhizosphere bacterium Pseudomonas sp. GRP3 that reduces damping-off disease in chile and tomato nurseries. Nat Prod 70:941–947
Sharma A, Pathak A, Sahgal M, Meyer JM, Wray V, Johri BN (2007b) Molecular characterization of plant growth-promoting rhizobacteria that enhance peroxidase and phenylalanine ammonia-lyase activities in chile (Capsicum annuum L.) and tomato (Lycopersicon esculentum Mill.). Arch Microbiol 88:483–494
Sharma A, Wray V, Johri BN (2007c) Rhizosphere Pseudomonas sp. strains reduce occurrence of pre- and post-emergence damping-off in chile and tomato in Central Himalayan Region. Arch Microbiol 187:321–335
van Loon LC, Bakker PAHM, Pieterse CMJ (1998) Systemic resistance induced by rhizosphere bacteria. Annu Rev Phytopathol 36:453–483
Weisburg WG, Barns SM, Pelletier DA, Lane DJ (1991) 16S ribosomal DNA amplification for phylogenetic study. J Bacteriol 173:697–703
Yan Z, Reddy MS, Ryu CM, McInroy JA, Wilson M, Kloepper JW (2002) Induced systemic protection against tomato late blight elicited by plant growth-promoting rhizobacteria. Phytopathology 92:1329–1333
Acknowledgments
The research was supported by DBT Grant No. BT/PR1231/AGR/21/340/2011 to DKC. The authors would like to thank Prof. A. R. Podile, DBT TASK-FORCE member for his valuable suggestions and guidance. Some of the research has been supported by and SERB-DST Grant No. SR/FT/LS-129/2012 to DKC.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Jain, S., Vaishnav, A., Kasotia, A. et al. Rhizobacterium-mediated growth promotion and expression of stress enzymes in Glycine max L. Merrill against Fusarium wilt upon challenge inoculation. World J Microbiol Biotechnol 30, 399–406 (2014). https://doi.org/10.1007/s11274-013-1455-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11274-013-1455-5