Abstract
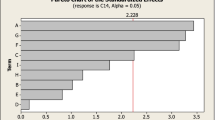
Mango peel is one of the major wastes from fruit processing industries, which poses considerable disposal problems and ultimately leads to environmental pollution. The objective of the current research was to determine the significant parameters on the production of polygalacturonase from mango peel which is a major industrial waste. Solid state culture conditions for polygalacturonase production by Fusarium moniliforme from dried mango peel powder were optimized by Taguchi’s L-18 orthogonal array experimental design methodology. Eight fungal metabolic influencing variables, viz. temperature, mango peel, inoculum, peptone, ammonium nitrate (NH4NO3), magnesium sulphate (MgSO4), zinc sulphate (ZnSO4) and potassium dihydrogen phosphate (KH2PO4) were selected to optimize polygalacturonase production. The optimized parameters composed of temperature (30°C), mango peel (6.5%, g, w/v), inoculum (8%, ml, v/v), peptone (1%, g, w/v), NH4NO3 (0.60%, g, w/v), MgSO4 (0.05%, g, w/v), ZnSO4 (0.06%, g, w/v) and KH2PO4 (0.4%, g, w/v). Based on the influence of interaction of fermentation components of fermentation, these could be classified as the least significant and the most significant at individual and interaction levels. The temperature, inoculum level, mango peel substrate and KH2PO4 showed maximum production impact at optimized conditions. From the optimized conditions the polygalacturonase activity was maximized to 43.2 U g−1.
Similar content being viewed by others
References
Acuna-Arguelles ME, Gutierrez-Rojas M, Viniegra-Gonzalez G, Favela-Torres E (1995) Production and properties of three pectinolytic activities produced by Aspergillus niger in submerged and solid state fermentation. Appl Microbiol Biotechnol 43:808–814. doi:10.1007/BF02431912
Aguilar G, Huitron C (1987) Stimulation of the production of extracellular pectinolytic activities of Aspergillus species by galacturonic acid and glucose addition. Enzyme Microb Technol 9:690–696. doi:10.1016/0141-0229(87)90129-3
Annis SL, Goodwin PH (1997) Recent advances in the molecular genetics of plant cell wall degradation enzymes produced by plant pathogenic fungi. Euro J Plant Pathol 103:1–14. doi:10.1023/A:1008656013255
Berardini N, Knodler M, Schieber A, Carle R (2005) Utilization of mango peels as a source of pectin and polyphenolics. Innov Food Sci Emerg Technol 6:442–452. doi:10.1016/j.ifset.2005.06.004
Chang MY, Tsai GJ, Houng YY (2006) Optimization of the medium composition for the submerged culture of Ganoderma lucidum by taguchi array design and sleepest ascent method. Enzyme Microb Technol 38:407–414. doi:10.1016/j.enzmictec.2005.06.011
Condemine G, Robert-Baudouy J (1995) Synthesis and secretion of Erwinia chrysanthemi virulence factors are coregulated. Mol Plant-Microbe Interact 8:632–636. doi:10.1094/MPMI-8-0632
Cooper RM, Wood RKS (1975) Regulation of synthesis of cell wall degrading enzymes by Verticilium albo-altrum and Fusarium oxysporium f.sp. lycopersicii. Physiol Plant Pathol 5:135–156
De Lorenzo G, Salvi G, Degra L, D’ Ovidio R, Cervone F (1987) Induction of extracellular polygalacturonase and its mRNA in the phytopathogenic fungus Fusarium moniliforme. J Gen Microbiol 133:3365–3373. doi:10.1099/00221287-133-12-3365
Diaz-Godfnez G, Soriano-Santos J, Augur C, Viniegra-Gonzalez G (2001) Exopectinases produced by Aspergillus niger in solid state and submerged fermentation: a comparative study. J Ind Microbiol Biotechnol 26:271–275. doi:10.1038/sj.jim.7000113
FAOSTAT (2007) FAO statistics, food and agriculture organization of the United Nations, Rome, Italy. http://faostat.fao.org/
Friedrich J, Cimerman A, Steiner W (1989) Submerged production pectinase enzymes Aspergillus niger. Effect of different aeration/agitation regimes. Appl Microbiol Biotechnol 31:490–494. doi:10.1007/BF00270782
Giridhari L, Siddappa GS, Tandon GL (1998) Preservation of fruits and vegetables. Indian Council of Agricultural Research, New Delhi, pp 265–272
Guimaraes CM, Giao MS, Martinez SS, Pintado AI, Pintado ME, Bento LS, Malcata FX (2007) Antioxidant activity of sugar molasses, including protective effect against DNA oxidative damage. J Food Sci 72:C39–C43. doi:10.1111/j.1750-3841.2006.00231.x
Gupta R, Paresh G, Mohapatra H, Goswami VK, Chauhan B (2003) Microbial α-amylases: a biotechnological perspective. Process Biochem 38:1599–1616. doi:10.1016/S0032-9592(03)00053-0
Hours RA, Voget CE, Ertola RJ (1988) Some factors affecting pectinase production from apple pomace in solid-state-cultures. Biol Wastes 24:147–157. doi:10.1016/0269-7483(88)90057-2
Joshi VK, Mukesh P, Neerja SR (2006) Pectin esterase production from apple pomace in solid-state and submerged fermentations. Food Technol Biotechnol 44:253–256
Kapoor M, Kuhad RC (2002) Improved polygalacturonase production from Bacillus sp. MG-cp-2 under submerged (SmF) and solid state (SSF) fermentation. Lett Appl Microbiol 34:317–322. doi:10.1046/j.1472-765X.2002.01107.x
Kashyap DR, Soni SK, Tewari R (2003) Enhanced production of pectinase by Bacillus sp. DT7 using solid state fermentation. Bioresour Technol 88:251–254. doi:10.1016/S0960-8524(02)00206-7
Kester HCM, Visser J (1990) Purification and characterisation of polygalacturonases produced by the hyphal fungus Aspergillus niger. Biotechnol Appl Biochem 12:150–160
Kumar SS, Palanivelu P (1998) Short note: production and properties of pectinolytic enzymes from the thermophilic fungus, Thermomyces lanuginosus. World J Microbiol Biotechnol 14:781–782. doi:10.1023/A:1008803123817
Kumar YS, Prakasam RS, Reddy OVS (2009) Optimization of fermentation conditions for mango (Mangifera indica L.) wine production by employing response surface methodology. Int J Food Sci Technol 44:2320–2327. doi:10.1111/j.1365-2621.2009.02076.x
Lang C, Dornenburg H (2000) Perspectives in the biological function and the technological application of polygalacturonases. Appl Microbiol Biotechnol 53:366–375. doi:10.1007/s002530051628
Lei SP, Lin HC, Heffernan L, Wilcox G (1985) Evidence that polygalacturonase is a virulence determinant in Erwinia carotova. J Bacteriol 164:831–835
Leuchtenberger A, Mayer G (1992) Changed pectinase synthesis by aggregated mycelium of some Aspergillus niger mutantes. Enzyme Microb Technol 14:18–22. doi:10.1016/0141-0229(92)90020-O
Macedo AC, Malcata FX, Oliveira JC (1997) Effect of production factors and ripening conditions on the characteristics of Serra cheese. Int J Food Sci Technol 32:501–511. doi:10.1111/j.1365-2621.1997.tb02124.x
Maldonado MC, Strasser de Saad AM (1998) Production of pectinesterase and polygalacturonase by Aspergillus niger in submerged and solid state systems. J Ind Microbiol Biotechnol 20:34–38. doi:10.1038/sj.jim.2900470
Martinez MJ, Alconada MT, Guillen F, Vazquez C, Reyes F (1991) Pectic activities from Fusarium oxysporum f. sp. Menolis: purification and characterization of exopolygalacturonase. FEMS Lett 81:145–149. doi:10.1111/j.1574-6968.1991.tb04737.x
Martins ES, Silva D, Da Silva R, Gomes E (2002) Solid state production of thermostable pectinases from thermophilic Thermoascus aurantiacus. Process Biochem 37:949–954. doi:10.1016/S0032-9592(01)00300-4
Mason RL, Gunst RF, Hess JL (1989) Statistical design and analysis of experiments with application to engineering and science. Wiley, Newyork
Miller GL (1959) Use of dinitrosalicylic acid reagent for determination of reducing sugar. Anal Chem 31:426–429
Nair SR, Rakshit SK, Panda T (2004) Effect of carbon sources on the synthesis of pectinase by Aspergillus. Bioprocess Biosys Eng 13:37–40. doi:10.1007/BF00368763
Pandey A, Nigam P, Soccol CR, Soccol VT, Singh D, Mohan R (2000) Advances in microbial amylases. Biotechnol Appl Biochem 31:135–152
Pandey MK, Singh MK, Singh RB (2002) Mycotoxic potential of some higher plants. Plant Dis Res 17:51–56
Patil RS, Dayanand A (2006) Optimization of process for the production of fungal pectinase from deseeded sunflower head in submerged and solid state conditions. Bioresour Technol 97:2340–2344. doi:10.1016/j.biortech.2005.10.025
Pereira SS, Torres FE, Gonzalez GV, Rojas MG (1993) Effect of different carbon sources on the synthesis of pectinase by Aspergillus niger on submerged and solid state fermentation. Appl Microbiol Biotechnol 39:36–41. doi:10.1007/BF00166845
Pilnik W, Voragen AGJ (1993) Pectic enzymes in fruit juice and vegetable juice manufacture. In: Reeds G (ed) Food and science technology. Enzymes in food processing. Academic Press, New York, pp 363–399
Prakasham RS, SubbaRao Ch, Sarma PN (2006) Green gram husk as innovative and inexpensive support for alkaline protease production by isolated Bacillus sp. under solid-state fermentation. Bioresour Technol 97:1449–1454. doi:10.1016/j.biortech.2005.07.015
Rao Narsimha M (1996) Pectinases from molds: their production, purification, characterizations and applications. Ph.D. Thesis University of Pune, India
Riou C, Freyssinet G, Fevre M (1992) Purification and characterization of extracellular pectinolytic enzyme produced by Sclerotinia sclerotiorum. Appl Environ Microbiol 58:578–583
Roy KR (1990) A primer on the Taguchi method. Van Nostrand Reinholds, New York
Sanzo AV, Hasan SDM, Costa JAV, Bertolin TE (2001) Enhanced glucoamylase production in semi-continuous solid state cultivation of Aspergillus niger NRRL 3122. Cienc Engenharia 10:59–62
Siessere V, Said S (1989) Pectic enzymes production in solid-state fermentation using citrus pulp pellets by Talaromyces flavus, Tubercularia vulgaris and Penicillium charlessi. Biotechnol Lett 11:343–344. doi:10.1007/BF01024515
Sreenivas Rao R, Prakasham RS, Krishna Prasad K, Rajesham S, Sarma PN, Venkateswara Rao L (2004) Xylitol production by Candida sp.: parameter optimization using Taguchi approach. Process Biochem 39:951–956. doi:10.1016/S0032-9592(03)00207-3
Srirangarajan AN, Shrikhande AJ (1976) Characterization of mango peel pectin. J Food Sci 42:279–280. doi:10.1111/j.1365-2621.1977.tb01273.x
Stowe RA, Mayer RP (1999) Efficient screening of process variables. Ind Eng Chem 56:36–40. doi:10.1021/ie50674a007
Strand LL, Corden ME, MacDonald DL (1976) Characterization of two endopolygalacturonase isoenzymes produced by Fusarium oxysporum f. sp. Lycopersici. Biochem Biophys Acta 429:870–883
Yakoby N, Kobiler I, Dinoor A, Prusky D (2000) pH regulation of pectate lyase secretion modulated the attack of Colletotrichum gloeosporiodes on avocado fruits. Appl Environ Microbiol 66:1026–1030
Acknowledgments
The authors express their thanks to CSIR, New Delhi, India, for the financial assistance. We also wish to thank Dr. S. C Basappa, former Deputy Director and Scientist, Central Food Technological Research Institute (CFTRI), Mysore, for his encouragement and critical comments on the manuscript.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Sudheer Kumar, Y., Varakumar, S. & Reddy, O.V.S. Production and optimization of polygalacturonase from mango (Mangifera indica L.) peel using Fusarium moniliforme in solid state fermentation. World J Microbiol Biotechnol 26, 1973–1980 (2010). https://doi.org/10.1007/s11274-010-0380-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11274-010-0380-0