Abstract
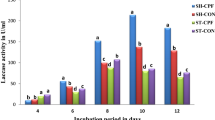
The present work was carried out to determine the optimum culture conditions of Phanerochaete chrysosporium (ATCC 20696) for maximizing ligninolytic enzyme production. Additionally, separation of its lignin peroxidase was conducted. After experiments, an optimized culture medium/condition was constructed (per liter of Kirk’s medium): dextrose 10 g, ammonium tartrate 0.11 g, Tween-80 0.5 g, MnSO4 7 mg, and veratryl alcohol 0.3 g in 10 mM acetic acid buffer pH 4.5. Under the optimized experimental condition, both lignin peroxidase (LiP) and manganese peroxidase (MnP) were detected and reach the highest yield at 30°C on the 8th day culture. Salt precipitation methods was used in the extraction and purification processes. Results show that salt precipitation with 60% (NH4)2SO4 yielded the best result, especially toward LiP. Enzyme separation was conducted and two fractions with LiP activity. LiP1 and LiP2 were produced using three columns sequentially: desalting column, Q FF ion exchange column and Sepharyl S-300 HR gel filtration. LiP1 and LiP2 had been purified by 9.6- and 7.6-fold with a yield of 22.9% and 18.6%, respectively. According to the data of sodium dodecyl sulfate polyacrilamide gel electrophoresis (SDS-PAGE), the molecular weights of the enzymes are 38 kDa and 40 kDa, respectively.








Similar content being viewed by others
References
Arora DS, Gill PK (2001) Effect of various media and supplements on laccase production by some white rot fungi. Bioresour Technol 77:89–91. doi:10.1016/S0960-8524(00)00114-0
Asther M, Corrieu G, Drapron R, Odier E (1987) Effect of Tween 80 and oleic acid on ligninase production by Phanerochaete chrysosporium INA-12. Enzyme Microb Technol 9:245–249. doi:10.1016/0141-0229(87)90024-X
Asther M, Lesage L, Drapron R, Corrieu G, Odier E (1988) Phospholipid and fatty acid enrichment of Phanerochaete chrysosporium INA-12 in relation to ligninase production. Appl Environ Microbiol 27:393–398
Bonnarme P, Jeffries TW (1990) Mn(II) regulation of lignin peroxidases and manganese-dependent peroxidases from lignin-degrading white rot fungi. Appl Environ Microbiol 56:210–217
Börjesson J, Peterson R, Tjerneld F (2006) Enhanced enzymatic conversion of softwood lignocellulose by poly (ethylene glycol) addition. Enzyme Microb 40:754–762. doi:10.1016/j.enzmictec.2006.06.006
Bothast RJ, Saha BC (1997) Ethanol production from agricultural biomass substrates. Adv Appl Microbiol 44:261–286
Brandlyn D, Faison T, Lent K, Roberta LF (1986) Role of veratryl alcohol in regulating ligninase activity in Phanerchaete chrysosporium. Appl Environ Microbiol 52(2):251–254
Breen A, Singleton FL (1999) Fungi in lignocellulose breakdown and biopulping. Curr Opin Biotechnol 10:252–258. doi:10.1016/S0958-1669(99)80044-5
de Boer HA, Zhang YZ, Collins C, Reddy CA (1987) Analysis of nucleotide sequences of two ligninase cDNAs from a white-rot filamentous fungus, Phanerochaete chrysosporium. Gene 60:93–102. doi:10.1016/0378-1119(87)90217-4
Faison BD, Kirk TK (1985) Factors involved in the regulation of a ligninase activity in Phanerochaete chrysosporium. Appl Environ Microbiol 49:299–304
Frank T, Etienne O (1988) Influence of veratryl alcohol and hydrogen peroxide on ligninase activity and ligninase production by Phanerochaete chrysosporium. Appl Environ Microbiol 54(2):466–472
Gill PK, Arora DS (2003) Effect of culture conditions on manganese peroxidase production and activity by some white rot fungi. J Ind Microbiol Biotechnol 30:28–33
Guha S, Jaffe PR (1998) Biodegradation kinetics of phenanthrene partitioned into the micellar phase of non-ionic surfactants. Environ Sci Technol 30:605–611. doi:10.1021/es950385z
Hamman OB, De la RT, Mart’ınez J (1999) The effect of manganese on the production of Phanerochaete flavido-alba ligninolytic peroxidases in nitrogen limited cultures. FEMS Microbiol Lett 177:137–142. doi:10.1111/j.1574-6968.1999.tb13724.x
Hodgson J, Rho D, Guiot SR, Ampleman G, Thiboutot S, Hawari J (2000) Tween 80 enhanced TNT mineralization by Phanerochaete chrysosporium. Can J Microbiol 46:110–118. doi:10.1139/cjm-46-2-110
Hofrichter M (2002) Lignin conversion by manganese peroxidase (MnP). Enzyme Microb Technol 30:454–466. doi:10.1016/S0141-0229(01)00528-2
Hu X, Zhao X, Hwang H (2006) Comparative study of immobilized Trametes versicolor laccase on nanoparticles and kaolinite. Chemosphere 66:1618–1626. doi:10.1016/j.chemosphere.2006.08.004
Jäger A, Croan C, Kirk TK (1985) Production of ligninases and degradation of lignin in agitated submerged cultures of Phanerochaete chrysosporium. Appl Environ Microbiol 50:1274–1278
Kapich AN, Prior BA, Botha A, Galkin S, Lundell T, Hatakka A (2004) Effect of lignocelluloses-containing substrates on production of ligninolytic peroxidases in submerged cultures of Phanerochaete chrysosporium ME-446. Enzyme Microb Technol 34: 187–195. doi:10.1016/j.enzmictec.2003.10.004
Kersten P, Cullen D (2006) Extracellular oxidative systems of the lignin-degrading basidiomycete Phanerochaete chrysosporium. Fungal Genet Biol 44:77–87. doi:10.1016/j.fgb.2006.07.007
Kirk TK, Croan S, Tien M, Murtagh KE Farrell RL (1986) Production of multiple ligninases by Phanerochaete chrysosporium: effect of selected growth conditions and use of a mutant strain. Enzyme Microb Technol 8:27–32. doi:10.1016/0141-0229(86)90006-2
Kuwahara M, Glenn LK, Morgan MA, Gold MH (1984) Separation and characterization of two extracellular H2O2-dependent oxidases from ligninolytic cultures of Phanerochaete chrysosporium. FEBS Lett 169:247–250. doi:10.1016/0014-5793(84)80327-0
Laemmli UK (1970) Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 227:680–685. doi:10.1038/227680a0
Lestan D, Cernilec M, Strancar A, Perdih A (1993) Influence of some surfactants and related compounds on ligninolytic activity of Phanerochaete chrysosporium. FEMS Microbiol Lett 106:17–22. doi:10.1111/j.1574-6968.1993.tb05929.x
Lowry OH, Rosenbrough NJ, Farr AL, Randall RJ (1951) Protein measurement with the Folin phenol reagent. J Biol Chem 193:265–275
Novotný Č, Svobodová K, Erbanová P, Cajthaml T, Kasinath A, Lang E, Šašek V (2004) Ligninolytic fungi in bioremediation: extracellular enzyme production and degradation rate. Soil Biol Biochem 36:1545–1551. doi:10.1016/j.soilbio.2004.07.019
Pazarlioglu NK, Urek RO, Ergun F (2005) Biodecolourization of direct blue 15 by immobilized Phanerochaete chrysosporium. Process Biochem 40:1923–1929. doi:10.1016/j.procbio.2004.07.005
Rodriguez CS, Rivela I, Munoz MR, Sanroman A (2000) Stimulation of ligninolytic enzyme production and the ability to decolourise Poly R-478 in semi-solid-state cultures of Phanerochaete chrysosporium. Bioresour Technol 74:159–164. doi:10.1016/S0960-8524(99)00156-X
Ron EZ, Rosenberg E (2002) Biosurfactants and bioremediation. Curr Opin Biotechnol 13:249–252. doi:10.1016/S0958-1669(02)00316-6
Swamy J, Ramsay JA (1999) Effects of Mn2+ and NH4+ concentrations on laccase and manganese peroxidase production and Amaranth decoloration by Trametes versicolor. Appl Microbiol Biotechnol 51:391–396. doi:10.1007/s002530051408
Ten-Have R, Hartmans S, Teunissen PJM (1998) Purification and characterization of two lignin peroxidase isozymes produced by Bjerkandera sp. FEMS Microbiol Lett 422:391–394
Tien M, Kirk TK (1988) Lignin peroxydase of Phanerochaete chrysosporium. Method 1988; 161:238–249
Xiong X, Wen X, Bai Y, Qian Y (2008) Effects of culture conditions on ligninolytic enzymes and protease production by Phanerochaete chrysosporium in air. J Environ Sci 20:94–100. doi:10.1016/S1001-0742(08)60014-5
Zheng ZM, Obbard JP (2001) Effect of nonionic surfactants on elimination of polycyclic aromatic hydrocarbons (PAHs) in soilslurry by Phanerochaete chrysosporium. J Chem Technol Biotechnol 76:423–429. doi:10.1002/jctb.396
Acknowledgments
The research is supported by the Department of Energy (DOE-MAT-SBI Program), grant number DE-FG36-05G085002 with subcontract# 07-08-001 to JSU from University of Mississippi (P.I. Dr. Clint Williford), via management of Mr. Sumesh Arora of Mississippi Technology Alliance.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Wang, P., Hu, X., Cook, S. et al. Effect of culture conditions on the production of ligninolytic enzymes by white rot fungi Phanerochaete chrysosporium (ATCC 20696) and separation of its lignin peroxidase. World J Microbiol Biotechnol 24, 2205–2212 (2008). https://doi.org/10.1007/s11274-008-9731-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11274-008-9731-5