Abstract
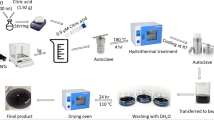
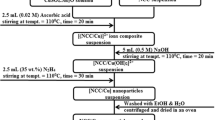
Recently, nanoparticles as adsorbents have received great attention due to their notable properties. In this regard, the acrylic acid adsorption from aqueous medium was examined by utilizing copper oxide (CuO) nanoparticles. In this research, initially, CuO nanoparticles were synthesized using a simple precipitation technique. CuO nanoparticles were characterized by Brunauer-Emmett-Teller (BET), Fourier transform infrared spectroscopy (FTIR), and scanning electron microscope (SEM) analyzes. CuO nanoparticles synthesized were in nano scale size ranged between 140 and 180 nm. FTIR analysis also confirmed the functional groups of CuO nanoparticles. Lastly, the effects of contact time (30–240 min), concentration of acrylic acid (2–10% w/w), temperature (25–55 °C), and CuO nanoparticle dosage (0.05–0.25 g) on the adsorption of acrylic acid with CuO nanoparticles were examined. The optimum adsorption conditions were obtained as the contact time of 180 min, the concentration of acrylic acid of 10% (w/w), nanoparticle dosage of 0.05 g, and temperature of 25 °C. At these conditions, the maximum adsorption capacity of CuO nanoparticles for acrylic acid was found as 202.67 mg g−1. This result confirmed that the synthesized CuO nanoparticles exhibited good adsorption performance towards to acrylic acid.












Similar content being viewed by others
References
Ahamed, M.J., Senkal, D., Shkel, A.M., (2014). Effect of annealing on mechanical quality factor of fused quartz hemispherical resonator, 2014 International Symposium on Inertial Sensors and Systems (ISISS). IEEE, 1-4.
Aroguz, A. Z., Gülen, J., & Evers, R. (2008). Adsorption of methylene blue from aqueous solution on pyrolyzed petrified sediment. Bioresource Technology, 99(6), 1503–1508.
Aşçı, Y. S., & İncİ, İ. (2010). Extraction equilibria of acrylic acid from aqueous solutions by amberlite LA-2 in various diluents. Journal of Chemical & Engineering Data, 55(7), 2385–2389.
Baylan, N., & Meriçboyu, A. E. (2016). Adsorption of lead and copper on bentonite and grapeseed activated carbon in single-and binary-ion systems. Separation Science and Technology, 51(14), 2360–2368.
Bhatia, D., Datta, D., Joshi, A., Gupta, S., & Gote, Y. (2019). Adsorption of isonicotinic acid from aqueous solution using multi-walled carbon nanotubes/Fe3O4. Journal of Molecular Liquids, 276, 163–169.
Bournel, F., Laffon, C., Parent, P., & Tourillon, G. (1996). Adsorption of acrylic acid on aluminium at 300 K: A multi-spectroscopic study. Surface Science, 352, 228–231.
Chiban, M., Soudani, A., Sinan, F., & Persin, M. (2011). Single, binary and multi-component adsorption of some anions and heavy metals on environmentally friendly Carpobrotus edulis plant. Colloids and Surfaces B: Biointerfaces, 82(2), 267–276.
Danner, H., Ürmös, M., Gartner, M., & Braun, R. (1998). Biotechnological production of acrylic acid from biomass. Applied Biochemistry and Biotechnology, 70(1), 887–894.
De, B., Wasewar, K., Dhongde, V., Madan, S., & Gomase, A. (2018). Recovery of acrylic acid using calcium peroxide nanoparticles: Synthesis, characterisation, batch study, equilibrium, and kinetics. Chemical and Biochemical Engineering Quarterly, 32(1), 29–39.
Farghali, A., Bahgat, M., Allah, A. E., & Khedr, M. (2013). Adsorption of Pb (II) ions from aqueous solutions using copper oxide nanostructures. Beni-Suef University Journal of Basic and Applied Sciences, 2(2), 61–71.
Ghaedi, M., Ghaedi, A., Hossainpour, M., Ansari, A., Habibi, M., & Asghari, A. (2014). Least square-support vector (LS-SVM) method for modeling of methylene blue dye adsorption using copper oxide loaded on activated carbon: Kinetic and isotherm study. Journal of Industrial and Engineering Chemistry, 20(4), 1641–1649.
Ghareib, M., Abdallah, W., Tahon, M., & Tallima, A. (2019). Biosynthesis of copper oxide nanoparticles using the preformed biomass of aspergillus fumigatus and their antibacterial and photocatalytic activities. Digest Journal of Nanomaterials & Biostructures (Djnb), 14(2), 291–303.
Gomes, T., Pereira, C. G., Cardoso, C., Pinheiro, J. P., Cancio, I., & Bebianno, M. J. (2012). Accumulation and toxicity of copper oxide nanoparticles in the digestive gland of Mytilus galloprovincialis. Aquatic Toxicology, 118, 72–79.
Goswami, A., Raul, P., & Purkait, M. (2012). Arsenic adsorption using copper (II) oxide nanoparticles. Chemical Engineering Research and Design, 90(9), 1387–1396.
Gülen, J., & Zorbay, F. (2017). Methylene blue adsorption on a low cost adsorbent-carbonized peanut shell. Water Environment Research, 89(9), 805–816.
Gülen, J., Aroguz, A., & Dalgın, D. (2005). Adsorption kinetics of azinphosmethyl from aqueous solution onto pyrolyzed Horseshoe sea crab shell from the Atlantic Ocean. Bioresource Technology, 96(10), 1169–1174.
Gülen, J., Altın, Z., & Özgür, M. (2013). Adsorption of amitraz on the clay. American Journal of Engineering Research, 2(6), 1–8.
Hassan, K. H., Jarullah, A. A., & Saadi, S. K. (2017). Synthesis of copper oxide nanoparticle as an adsorbent for removal of Cd (II) and Ni (II) ions from binary system. International Journal of Applied Environmental Sciences, 12(11), 1841–1861.
Hosseini, R., Sayadi, M. H., & Shekari, H. (2019). Adsorption of nickel and chromium from aqueous solutions using copper oxide nanoparticles: Adsorption isotherms, kinetic modeling, and thermodynamic studies. Avicenna Journal of Environmental Health Engineering, 6(2), 1–9.
Ijagbemi, C. O., Baek, M. H., & Kim, D. S. (2009). Montmorillonite surface properties and sorption characteristics for heavy metal removal from aqueous solutions. Journal of Hazardous Materials, 166(1), 538–546.
Keshav, A., Chand, S., & Wasewar, K. L. (2009). Reactive extraction of acrylic acid using tri-n-butyl phosphate in different diluents. Journal of Chemical & Engineering Data, 54(6), 1782–1786.
Lunelli, B.H., Duarte, E.R., De Toledo, E.V., Maciel, M.W., Maciel Filho, R., (2007). A new process for acrylic acid synthesis by fermentative process. Applied Biochemistry and Biotecnology. Springer, 487-499.
Mahmoodi, N. M., Hayati, B., Arami, M., & Lan, C. (2011). Adsorption of textile dyes on pine cone from colored wastewater: Kinetic, equilibrium and thermodynamic studies. Desalination, 268(1–3), 117–125.
Mao, Y., & Fung, B. (1997). A study of the adsorption of acrylic acid and maleic acid from aqueous solutions onto alumina. Journal of Colloid and Interface Science, 191(1), 216–221.
Mironyuk, I., Tatarchuk, T., Naushad, M., Vasylyeva, H., & Mykytyn, I. (2019). Highly efficient adsorption of strontium ions by carbonated mesoporous TiO2. Journal of Molecular Liquids, 285, 742–753.
Musante, C., & White, J. C. (2012). Toxicity of silver and copper to Cucurbita pepo: Differential effects of nano and bulk-size particles. Environmental Toxicology, 27, 510–517.
Naushad, M. (2014). Surfactant assisted nano-composite cation exchanger: Development, characterization and applications for the removal of toxic Pb2+ from aqueous medium. Chemical Engineering Journal, 235, 100–108.
Naushad, M., Alqadami, A. A., AlOthman, Z. A., Alsohaimi, I. H., Algamdi, M. S., & Aldawsari, A. M. (2019). Adsorption kinetics, isotherm and reusability studies for the removal of cationic dye from aqueous medium using arginine modified activated carbon. Journal of Molecular Liquids, 293, 111442.
Nayak, A. K., & Pal, A. (2019). Development and validation of an adsorption kinetic model at solid-liquid interface using normalized Gudermannian function. Journal of Molecular Liquids, 276, 67–77.
Nekouei, F., Nekouei, S., Tyagi, I., & Gupta, V. K. (2015). Kinetic, thermodynamic and isotherm studies for acid blue 129 removal from liquids using copper oxide nanoparticle-modified activated carbon as a novel adsorbent. Journal of Molecular Liquids, 201, 124–133.
Ojedokun, A. T., & Bello, O. S. (2017). Kinetic modeling of liquid-phase adsorption of Congo red dye using guava leaf-based activated carbon. Applied Water Science, 7(4), 1965–1977.
Rajput, V., Minkina, T., Fedorenko, A., Sushkova, S., Mandzhieva, S., Lysenko, V., Duplii, N., Fedorenko, G., Dvadnenko, K., & Ghazaryan, K. (2018a). Toxicity of copper oxide nanoparticles on spring barley (Hordeum sativum distichum). Science of the Total Environment, 645, 1103–1113.
Rajput, V.D., Minkina, T., Fedorenko, A., Tsitsuashvili, V., Mandzhieva, S., Sushkova, S., Azarov, A., (2018b). Metal oxide nanoparticles: Applications and effects on soil ecosystems. Soil contamination: Sources, assessment and remediation. Nova Science Publishers, Hauppauge, 81–106.
Rashad, M., & Al-Aoh, H. A. (2019). Promising adsorption studies of bromophenol blue using copper oxide nanoparticles. Desalination and Water Treatment, 139, 360–368.
Sebeia, N., Jabli, M., Ghith, A., & Saleh, T. A. (2020). Eco-friendly synthesis of Cynomorium coccineum extract for controlled production of copper nanoparticles for sorption of methylene blue dye. Arabian Journal of Chemistry, 13(2), 4263–4274.
Singh, S., Kumar, N., Kumar, M., Agarwal, A., & Mizaikoff, B. (2017). Electrochemical sensing and remediation of 4-nitrophenol using bio-synthesized copper oxide nanoparticles. Chemical Engineering Journal, 313, 283–292.
Sivaraj, R., Rahman, P. K., Rajiv, P., Salam, H. A., & Venckatesh, R. (2014). Biogenic copper oxide nanoparticles synthesis using Tabernaemontana divaricate leaf extract and its antibacterial activity against urinary tract pathogen. Spectrochimica Acta Part A: Molecular and Biomolecular Spectroscopy, 133, 178–181.
Straathof, A. J., Sie, S., Franco, T. T., & Van der Wielen, L. A. (2005). Feasibility of acrylic acid production by fermentation. Applied Microbiology and Biotechnology, 67(6), 727–734.
Suárez-Cerda, J., Espinoza-Gómez, H., Alonso-Núñez, G., Rivero, I. A., Gochi-Ponce, Y., & Flores-López, L. Z. (2017). A green synthesis of copper nanoparticles using native cyclodextrins as stabilizing agents. Journal of Saudi Chemical Society, 21(3), 341–348.
Xiaobo, X., Jianping, L., & Peilin, C. (2006). Advances in the research and development of acrylic acid production from biomass. Chinese Journal of Chemical Engineering, 14(4), 419–427.
Zhang, H., Gong, Y., Li, C., Zhang, L., & Zhu, C. (2011). Liquid−liquid equilibria for the quaternary system water (1)+ acrylic acid (2)+ acetic acid (3)+ cyclohexane (4) at (293.15, 303.15, and 313.15) K. Journal of Chemical & Engineering Data, 56(5), 2332–2336.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Baylan, N., İlalan, İ. & İnci, İ. Copper Oxide Nanoparticles as a Novel Adsorbent for Separation of Acrylic Acid from Aqueous Solution: Synthesis, Characterization, and Application. Water Air Soil Pollut 231, 465 (2020). https://doi.org/10.1007/s11270-020-04832-3
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11270-020-04832-3