Abstract
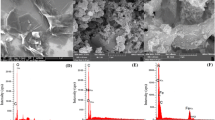
Recent studies show that carbonaceous materials have gained interest because of their superior features over the alternative adsorbents. Therefore, it is of great value to synthesize novel carbonaceous adsorbents. In the present study, graphene oxide nanopowders (GON) were synthesized through a modified Hummer’s method. The material has been characterized by X-ray diffraction (XRD), scanning electron microscopy (SEM), and Fourier transform infrared spectroscopy (FTIR) methods. Naproxen has been selected as the model pharmaceutical to investigate the adsorption performance of GON. The highest adsorption removal was found to be 65.28% under the optimum conditions (0.03 g of GON for the adsorption of 10 mg/L naproxen solution at 100 rpm mixing the speed of shaking bath). The relevant adsorption system was an exothermic, spontaneous, and chemisorption process depending on the kinetic (pseudo-first order, pseudo-second order, intraparticular, and Elovich models), equilibrium (Langmuir, Freundlich, Temkin, Dubinin-Radushkevich, Harkin-Jura, and Halsey isotherm equations), and thermodynamic parameters.







Similar content being viewed by others
References
Amin, M., Alazba, A., Shafiq, M., et al. (2015). Adsorptive removal of reactive black 5 from wastewater using bentonite clay: isotherms, kinetics and thermodynamics. Sustainability, 7, 15302–15318. https://doi.org/10.3390/su71115302.
Bele, S., Samanidou, V., & Deliyanni, E. (2016). Effect of the reduction degree of graphene oxide on the adsorption of Bisphenol A. Chemical Engineering Research and Design, 109, 573–585. https://doi.org/10.1016/j.cherd.2016.03.002.
Bian, Y., Bian, Z.-Y., Zhang, J.-X., et al. (2015). Effect of the oxygen-containing functional group of graphene oxide on the aqueous cadmium ions removal. Applied Surface Science, 329, 269–275. https://doi.org/10.1016/J.APSUSC.2014.12.090.
Chowdhury, S., Mishra, R., Saha, P., & Kushwaha, P. (2011). Adsorption thermodynamics, kinetics and isosteric heat of adsorption of malachite green onto chemically modified rice husk. Desalination, 265, 159–168. https://doi.org/10.1016/j.desal.2010.07.047.
Çolak, F., Atar, N., & Olgun, A. (2009). Biosorption of acidic dyes from aqueous solution by Paenibacillus macerans: kinetic, thermodynamic and equilibrium studies. Chemical Engineering Journal, 150, 122–130. https://doi.org/10.1016/j.cej.2008.12.010.
Domínguez-Vargas, J. R., Gonzalez, T., Palo, P., & Cuerda-Correa, E. M. (2013). Removal of carbamazepine, naproxen, and trimethoprim from water by Amberlite XAD-7: a kinetic study. Clean: Soil, Air, Water, 41, 1052–1061. https://doi.org/10.1002/clen.201200245.
Dordio, A. V., Miranda, S., Prates Ramalho, J. P., & Carvalho, A. J. P. (2017). Mechanisms of removal of three widespread pharmaceuticals by two clay materials. Journal of Hazardous Materials, 323, 575–583. https://doi.org/10.1016/J.JHAZMAT.2016.05.091.
Emik, S. (2014). Preparation and characterization of an IPN type chelating resin containing amino and carboxyl groups for removal of Cu(II) from aqueous solutions. Reactive and Functional Polymers, 75, 63–74. https://doi.org/10.1016/j.reactfunctpolym.2013.12.006.
Freundlich, H. (1907). Über die Adsorption in Lösungen. Zeitschrift für Physikalische Chemie, 57U, 385–470. https://doi.org/10.1515/zpch-1907-5723.
Gao, Y., Li, Y., Zhang, L., et al. (2012). Adsorption and removal of tetracycline antibiotics from aqueous solution by graphene oxide. Journal of Colloid and Interface Science, 368, 540–546. https://doi.org/10.1016/j.jcis.2011.11.015.
Ghadim, E. E., Manouchehri, F., Soleimani, G., et al. (2013). Adsorption properties of tetracycline onto graphene oxide: equilibrium, kinetic and thermodynamic studies. PLoS One, 8, e79254. https://doi.org/10.1371/journal.pone.0079254.
Gupta, V. K., Gupta, M., & Sharma, S. (2001). Process development for the removal of lead and chromium from aqueous solutions using red mud—an aluminium industry waste. Water Research, 35, 1125–1134. https://doi.org/10.1016/S0043-1354(00)00389-4.
Halsey, G. (1948). Physical adsorption on non-uniform surfaces. The Journal of Chemical Physics, 16, 931–937. https://doi.org/10.1063/1.1746689.
Hasan, Z., & Jhung, S. H. (2015). Removal of hazardous organics from water using metal-organic frameworks (MOFs): plausible mechanisms for selective adsorptions. Journal of Hazardous Materials, 283, 329–339. https://doi.org/10.1016/J.JHAZMAT.2014.09.046.
Hasan, Z., Jeon, J., & Jhung, S. H. (2012). Adsorptive removal of naproxen and clofibric acid from water using metal-organic frameworks. Journal of Hazardous Materials, 209–210, 151–157. https://doi.org/10.1016/J.JHAZMAT.2012.01.005.
Hasan, Z., Choi, E.-J., & Jhung, S. H. (2013). Adsorption of naproxen and clofibric acid over a metal–organic framework MIL-101 functionalized with acidic and basic groups. Chemical Engineering Journal, 219, 537–544. https://doi.org/10.1016/j.cej.2013.01.002.
Ho, Y.-S. (2006). Review of second-order models for adsorption systems. Journal of Hazardous Materials, 136, 681–689. https://doi.org/10.1016/j.jhazmat.2005.12.043.
Ho, Y., & McKay, G. (1999). Pseudo-second order model for sorption processes. Process Biochemistry, 34, 451–465. https://doi.org/10.1016/S0032-9592(98)00112-5.
İlbay, Z., Şahin, S., Kerkez, Ö., & Bayazit, Ş. S. (2015). Isolation of naproxen from wastewater using carbon-based magnetic adsorbents. International journal of Environmental Science and Technology, 12. https://doi.org/10.1007/s13762-015-0775-4.
Joguet, L., Sondi, I., & Matijević, E. (2002). Preparation of nanosized drug particles by the coating of inorganic cores: naproxen and ketoprofen on alumina. Journal of Colloid and Interface Science, 251, 284–287. https://doi.org/10.1006/JCIS.2002.8408.
Kaman, S. P. D., Tan, I. A. W., & Lim, L. L. P. (2017). Palm oil mill effluent treatment using coconut shell–based activated carbon: adsorption equilibrium and isotherm. In MATEC Web of Conferences (Vol. 87, p. 03009). EDP sciences.
Khazri, H., Ghorbel-Abid, I., Kalfat, R., & Trabelsi-Ayadi, M. (2017). Removal of ibuprofen, naproxen and carbamazepine in aqueous solution onto natural clay: equilibrium, kinetics, and thermodynamic study. Applied Water Science, 7, 3031–3040. https://doi.org/10.1007/s13201-016-0414-3.
Kurtulbaş, E., Bilgin, M., Şahin, S., & Bayazit, Ş. S. (2017). Comparison of different polymeric resins for naproxen removal from wastewater. Journal of Molecular Liquids, 241, 633–637. https://doi.org/10.1016/J.MOLLIQ.2017.06.070.
Langmuir, I. (1918). The adsorption of gases on plane surfaces of glass, mica and platinum. Journal of the American Chemical Society, 40, 1361–1403. https://doi.org/10.1021/ja02242a004.
Lin, Y., Xu, S., & Li, J. (2013). Fast and highly efficient tetracyclines removal from environmental waters by graphene oxide functionalized magnetic particles. Chemical Engineering Journal, 225, 679–685. https://doi.org/10.1016/j.cej.2013.03.104.
Lu, X., Shao, Y., Gao, N., & Ding, L. (2015). Equilibrium, thermodynamic, and kinetic studies of the adsorption of 2,4-dichlorophenoxyacetic acid from aqueous solution by MIEX resin. Journal of Chemical & Engineering Data, 60, 1259–1269. https://doi.org/10.1021/je500902p.
Mohammadi Nodeh, M. K., Radfard, M., Zardari, L. A., & Rashidi Nodeh, H. (2018). Enhanced removal of naproxen from wastewater using silica magnetic nanoparticles decorated onto graphene oxide; parametric and equilibrium study. Separation Science and Technology, 53, 2476–2485. https://doi.org/10.1080/01496395.2018.1457054.
Önal, Y., Akmil-Başar, C., & Sarıcı-Özdemir, Ç. (2007). Elucidation of the naproxen sodium adsorption onto activated carbon prepared from waste apricot: kinetic, equilibrium and thermodynamic characterization. Journal of Hazardous Materials, 148(3), 727–734. https://doi.org/10.1016/j.jhazmat.2007.03.037.
Ozdemir, O. K. (2019). A novel method to produce few layers of graphene as support materials for platinum catalyst. Chemical Papers, 73, 387–395. https://doi.org/10.1007/s11696-018-0588-2.
Phasuphan, W., Praphairaksit, N., & Imyim, A. (2019). Removal of ibuprofen, diclofenac, and naproxen from water using chitosan-modified waste tire crumb rubber. Journal of Molecular Liquids, 294, 111554.
Saadi, R., Saadi, Z., Fazaeli, R., & Fard, N. E. (2015). Monolayer and multilayer adsorption isotherm models for sorption from aqueous media. Korean Journal of Chemical Engineering, 32, 787–799. https://doi.org/10.1007/s11814-015-0053-7.
Şahin, S., & Emik, S. (2018). Fast and highly efficient removal of 2,4-D using amino-functionalized poly (glycidyl methacrylate) adsorbent: optimization, equilibrium, kinetic and thermodynamic studies. Journal of Molecular Liquids, 260, 195–202. https://doi.org/10.1016/J.MOLLIQ.2018.03.091.
Salem Attia, T. M., Hu, X. L., & Yin, D. Q. (2013). Synthesized magnetic nanoparticles coated zeolite for the adsorption of pharmaceutical compounds from aqueous solution using batch and column studies. Chemosphere, 93, 2076–2085. https://doi.org/10.1016/J.CHEMOSPHERE.2013.07.046.
Sarker, M., Song, J. Y., & Jhung, S. H. (2018). Adsorptive removal of anti-inflammatory drugs from water using graphene oxide/metal-organic framework composites. Chemical Engineering Journal, 335, 74–81. https://doi.org/10.1016/J.CEJ.2017.10.138.
Seo, P. W., Bhadra, B. N., Ahmed, I., et al. (2016). Adsorptive removal of pharmaceuticals and personal care products from water with functionalized metal-organic frameworks: remarkable adsorbents with hydrogen-bonding abilities. Scientific Reports, 6, 34462. https://doi.org/10.1038/srep34462.
Song, J. Y., & Jhung, S. H. (2017). Adsorption of pharmaceuticals and personal care products over metal-organic frameworks functionalized with hydroxyl groups: quantitative analyses of H-bonding in adsorption. Chemical Engineering Journal, 322, 366–374. https://doi.org/10.1016/J.CEJ.2017.04.036.
Soto, M. L., Moure, A., Domínguez, H., & Parajó, J. C. (2011). Recovery, concentration and purification of phenolic compounds by adsorption: a review. Journal of Food Engineering, 105, 1–27. https://doi.org/10.1016/j.jfoodeng.2011.02.010.
Subramanyam, B., & Das, A. (2009). Study of the adsorption of phenol by two soils based on kinetic and isotherm modeling analyses. Desalination, 249, 914–921. https://doi.org/10.1016/j.desal.2009.05.020.
Tang, Y., Guo, H., Xiao, L., et al. (2013). Synthesis of reduced graphene oxide/magnetite composites and investigation of their adsorption performance of fluoroquinolone antibiotics. Colloids and Surfaces A: Physicochemical and Engineering Aspects, 424, 74–80. https://doi.org/10.1016/j.colsurfa.2013.02.030.
Tanhaei, B., Ayati, A., Lahtinen, M., & Sillanpää, M. (2015). Preparation and characterization of a novel chitosan/Al2O3/magnetite nanoparticles composite adsorbent for kinetic, thermodynamic and isotherm studies of methyl orange adsorption. Chemical Engineering Journal, 259, 1–10. https://doi.org/10.1016/J.CEJ.2014.07.109.
Temkin, I. M. (1940). Kinetics of ammonia synthesis on promoted iron catalysts. Acta Physiochimica URSS, 12, 327–356.
Vadi, M. (2013). Adsorption isotherms of some non-steroidal drugs on single wall carbon nanotube. Asian Journal of Chemistry, 25(6), 3431.
Wu, S., Zhao, X., Li, Y., et al. (2013). Adsorption of ciprofloxacin onto biocomposite fibers of graphene oxide/calcium alginate. Chemical Engineering Journal, 230, 389–395. https://doi.org/10.1016/j.cej.2013.06.072.
Yu, Z., Peldszus, S., & Huck, P. M. (2008). Adsorption characteristics of selected pharmaceuticals and an endocrine disrupting compound—naproxen, carbamazepine and nonylphenol—on activated carbon. Water Research, 42, 2873–2882. https://doi.org/10.1016/J.WATRES.2008.02.020.
Yu, Z., Peldszus, S., & Huck, P. M. (2009). Adsorption of selected pharmaceuticals and an endocrine disrupting compound by granular activated carbon. 2. Model prediction. Environmental Science & Technology, 43, 1474–1479. https://doi.org/10.1021/es7032185.
Yusan, S., & Erenturk, S. (2011). Adsorption characterization of strontium on PAN/zeolite composite adsorbent Chemometric methods view project Measurment of radioactivity of soil, sediment and aqueous samples view project adsorption characterization of strontium on PAN/zeolite composite adsorbent. World Journal of Nuclear Science and Technology, 1, 6–12. https://doi.org/10.4236/wjnst.2011.11002.
Zhao, G., Wen, T., Yang, X., et al. (2012). Preconcentration of U( vi ) ions on few-layered graphene oxide nanosheets from aqueous solutions. Dalton Transactions, 41, 6182–6188. https://doi.org/10.1039/C2DT00054G.
Zolgharnein, J., Dalvand, K., Rastgordani, M., & Zolgharnein, P. (2017). Adsorptive removal of phosphate using nano cobalt hydroxide as a sorbent from aqueous solution; multivariate optimization and adsorption characterization. Journal of Alloys and Compounds, 725, 1006–1017. https://doi.org/10.1016/J.JALLCOM.2017.07.228.
Acknowledgements
We would like to thank my master student Ms. Berna Erim for preparing a schematic representation of GON synthesis.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Ciğeroğlu, Z., Özdemir, O.K., Şahin, S. et al. Naproxen Adsorption onto Graphene Oxide Nanopowders: Equilibrium, Kinetic, and Thermodynamic Studies. Water Air Soil Pollut 231, 101 (2020). https://doi.org/10.1007/s11270-020-04472-7
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11270-020-04472-7