Abstract
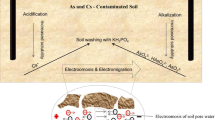
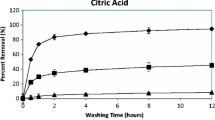
In this study, electrokinetic remediation (EKR) was carried out to remove arsenic (As) from soil washing residue. We screened various processing fluids and found that oxalic acid was most effective for As removal because it reductively dissolved Fe and As from the soil. In EKR, however, NaOH was a more effective agent for removing As, implying that the main removal mechanism of As was ion exchange between OH– and oxyanionic As. Oxalic and citric acid, both of which were efficient agents for removing As in the screening tests, did not effectively remove As by EKR, probably due to the relatively high pH and low soil-to-agent ratio. In EKR, As was mainly removed by electromigration toward the anode, even under high amounts of accumulated electro-osmotic flow. Therefore, strategies that increase electromigration have potential for enhancing As removal.








Similar content being viewed by others
References
Acar, Y. B., & Alshawabkeh, A. N. (1993). Principles of electrokinetic remediation. Environmental Science & Technology, 27, 2638–2647.
Alshawabkeh, A. N. (2009). Electrokinetic soil remediation: challenges and opportunities. Separation Science and Technology, 44, 2171–2187.
Asadi, A., Huat, B. B., & Keykhah, H. A. (2013). Theory of electroosmosis in soil. International Journal of Electrochemical Science, 8, 1016–1025.
Baek, K., Kim, D. H., Park, S. W., Ryu, B. G., Bajargal, T., & Yang, J. S. (2009). Electrolyte conditioning-enhanced electrokinetic remediation of arsenic-contaminated mine tailing. Journal of Hazardous Materials, 161, 457–462.
Chaiyaraksa, C., & Sriwiriyanuphap, N. (2004). Batch washing of cadmium from soil and sludge by a mixture of Na2S2O5 and Na2EDTA. Chemosphere, 56, 1129–1135.
Das, B. M. (2013). Advanced soil mechanics, CRC Press.
Dermont, G., Bergeron, M., Mercier, G., & Richer-Lafleche, M. (2008). Soil washing for metal removal: a review of physical/chemical technologies and field applications. Journal of Hazardous Materials, 152, 1–31.
Dixit, S., & Hering, J. G. (2003). Comparison of arsenic (V) and arsenic (III) sorption onto iron oxide minerals: implications for arsenic mobility. Environmental Science & Technology, 37, 4182–4189.
Goldberg, S., & Johnston, C. T. (2001). Mechanisms of arsenic adsorption on amorphous oxides evaluated using macroscopic measurements, vibrational spectroscopy, and surface complexation modeling. Journal of Colloid and Interface Science, 234, 204–216.
Grafe, M., Eick, M., & Grossl, P. (2001). Adsorption of arsenate (V) and arsenite (III) on goethite in the presence and absence of dissolved organic carbon. Soil Science Society of America Journal, 65, 1680–1687.
Jackson, B. P., & Miller, W. (2000). Effectiveness of phosphate and hydroxide for desorption of arsenic and selenium species from iron oxides. Soil Science Society of America Journal, 64, 1616–1622.
Jang, M., Hwang, J. S., Choi, S. I., & Park, J. K. (2005). Remediation of arsenic-contaminated soils and washing effluents. Chemosphere, 60, 344–354.
Kim, D. H., Ryu, B. G., Park, S. W., Seo, C. I., & Baek, K. (2009). Electrokinetic remediation of Zn and Ni-contaminated soil. Journal of Hazardous Materials, 165, 501–505.
Kim, D.-H., Jo, S.-U., Choi, J.-H., Yang, J.-S., & Baek, K. (2012). Hexagonal two dimensional electrokinetic systems for restoration of saline agricultural lands: a pilot study. Chemical Engineering Journal, 198, 110–121.
Kim, E. J., Lee, J. C., & Baek, K. (2015). Abiotic reductive extraction of arsenic from contaminated soils enhanced by complexation: arsenic extraction by reducing agents and combination of reducing and chelating agents. Journal of Hazardous Materials, 283, 454–461.
Ko, I., Chang, Y. Y., Lee, C. H., & Kim, K. W. (2005). Assessment of pilot-scale acid washing of soil contaminated with As, Zn and Ni using the BCR three-step sequential extraction. Journal of Hazardous Materials, 127, 1–13.
Lu, P., & Zhu, C. (2011). Arsenic Eh–pH diagrams at 25 C and 1 bar. Environmental Earth Sciences, 62, 1673–1683.
Min, Z., Bohan, L., Ming, L., ZHANG, Y., Qingru, Z., & OUYANG, B. (2008). Arsenic removal from contaminated soil using phosphoric acid and phosphate. Journal of Environmental Sciences, 20, 75–79.
Mohapatra, D., Singh, P., Zhang, W., & Pullammanappallil, P. (2005). The effect of citrate, oxalate, acetate, silicate and phosphate on stability of synthetic arsenic-loaded ferrihydrite and Al-ferrihydrite. Journal of Hazardous Materials, 124, 95–100.
Reddy, K. R., Saichek, R. E., Maturi, K., & Ala, P. (2002). Effects of soil moisture and heavy metal concentrations on electrokinetic remediation. Indian Geotechnical Journal, 32, 258–288.
Reddy, K. R., Darko-Kagya, K., & Al-Hamdan, A. Z. (2011). Electrokinetic remediation of pentachlorophenol contaminated clay soil. Water, Air, & Soil Pollution, 221, 35–44.
Ryu, B.-G., Yang, J.-S., Kim, D.-H., & Baek, K. (2009). Pulsed electrokinetic removal of Cd and Zn from fine-grained soil. Journal of Applied Electrochemistry, 40, 1039–1047.
Ryu, B.-G., Park, G.-Y., Yang, J.-W., & Baek, K. (2011). Electrolyte conditioning for electrokinetic remediation of As, Cu, and Pb-contaminated soil. Separation and Purification Technology, 79, 170–176.
Wenzel, W. W., Kirchbaumer, N., Prohaska, T., Stingeder, G., Lombi, E., & Adriano, D. C. (2001). Arsenic fractionation in soils using an improved sequential extraction procedure. Analytica Chimica Acta, 436, 309–323.
Whitacre, D. M. & Gunther, F. A. (2008). Reviews of environmental contamination and toxicology, Springer.
Yang, J. S., Lee, J. Y., Baek, K., Kwon, T. S., & Choi, J. (2009). Extraction behavior of As, Pb, and Zn from mine tailings with acid and base solutions. Journal of Hazardous Materials, 171, 443–451.
Zhang, H., & Selim, H. (2005). Kinetics of arsenate adsorption-desorption in soils. Environmental Science & Technology, 39, 6101–6108.
Acknowledgments
This work was supported by KEITI and NRF.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Shin, SY., Park, SM. & Baek, K. Electrokinetic Removal of As from Soil Washing Residue. Water Air Soil Pollut 227, 223 (2016). https://doi.org/10.1007/s11270-016-2918-8
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11270-016-2918-8