Abstract
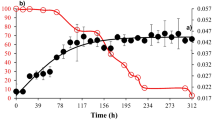
Pyrene, a four-ring polycyclic aromatic hydrocarbon that is highly resistant to degradation, persists in the environment and exerts its harmful effects toward humans, flora, and fauna when accumulated to a certain level. The ineffectiveness of conventional physical–chemical treatment methods has urged the emergence of biological treatments to degrade pyrene that persists in the environment. In this study, Pleurotus eryngii F032 was originally isolated from our laboratory due to its ability to degrade pyrene. Optimum conditions for pyrene degradation were determined using five different parameters, including pyrene concentration, incubation temperature, pH, agitation, and rhamnolipid concentration. The culture was incubated for 7, 15, 23, and 30 days, respectively, followed by pyrene extraction for degradation analysis. Results show that lower pyrene concentration requires less time for degradation by P. eryngi F032. Moreover, more time is needed for degradation when higher concentration is used, resulting in slower degradation. Optimum pyrene degradation conditions by P. eryngii F032 have been recorded at 40 °C incubation temperature, pH 3, and 2.5 % of rhamnolipid concentration with an agitation speed of 120 rpm. The capability of P. eryngii F032 to utilize pyrene as carbon and energy source depends on the presence of ligninolytic enzymes. The formation of protocatechuic acid resulting from pyrene degradation was detected via GC-MS analysis, which was further confirmed through spectrophotometric analysis.






Similar content being viewed by others
References
Bezalel, L., Hadar, Y., & Cerniglia, C. E. (1997). Enzymatic mechanisms involved in phenanthrene degradation by white-rot fungus Pleurotus ostreatus. Applied and Environmental Microbiology, 63, 2495–2501.
Bishnoi, K., Kumar, R., & Bishnoi, N. R. (2008). Biodegradation of polycyclic aromatic hydrocarbons by white rot fungus. Journal of Science Index Resource, 67, 538–542.
Eriksson, M., Dalhammar, G., & Borg-Karlson, A. K. (1999). Aerobic degradation of a hydrocarbon mixture in a natural uncontaminated potting soil by indigenous microorganisms at 20 °C and 6 °C. Applied Microbiology and Biotechnology, 51, 532–535.
Fukushima, Y., & Kirk, T. K. (1995). Laccase component of the Ceriporiospsis subvermispoa lignin-degrading enzyme. Applied and Environmental Microbiology, 61, 872–876.
Gao, L., & Liu, X. (2010). Effects of carbon concentrations and carbon to nitrogen ratios on sporulation of two biological control fungi as determined by different culture methods. Mycopathologia, 169, 475–481.
Hadibarata, T., Yusoff, A. R. M., Aris, A., & Kristanti, R. A. (2012a). Identification of naphthalene metabolism by white-rot fungus Armillaria sp. F022. Journal of Environment Science, 24, 728–732.
Hadibarata, T., Yusoff, A. R. M., Aris, A., Hidayat, T., Salmiati, & Kristanti, R. A. (2012b). Decoloration of azo, triphenylmethane and anthraquinone dyes by laccase of a newly isolated Armillaria sp. F022. Water Air Soil Pollution, 223, 1045–1054.
Hadibarata, T., Teh, Z. C., Rubiyatno, Zubir, M. M. F. A., Khudhair, A. B., Yusoff, A. R. M., et al. (2013). Identification of naphthalene metabolism by white rot fungus Pleurotus eryngii. Bioprocess and Biosystem Engineering, 36, 1455–1461.
Han, M. J., Choi, H. T., & Song, H. G. (2004). Degradation of phenanthrene by Trametes versicolor and its laccase. Journal of Microbiology, 42, 94–98.
Jadhav, U. U., Dawker, V. V., Kagalkar, A. N., & Govindwar, S. P. (2011). Effect of metals on decolorization of reactive blue HERD by Comamonas sp. UVS. Water Air Soil Pollution, 216, 621–631.
Jager, A., Croan, S., & Kirk, T. (1985). Production of ligninases and degradation of lignin in agitated submerged cultures of Phanerochaete chrysosporium. Applied Microbiology and Biotechnology, 50, 1274–1278.
Kariminiaae-Hamedaani, H. R., Sakurai, A., & Sakakibara, M. (2007). Decolorization of synthetic dyes by a new manganese peroxidase-producing white rot fungus. Dyes and Pigments, 72, 157–162.
Lange, B., Kremer, S., Anke, H., & Sterner, O. (1996). Metabolism of pyrene by basidiomycetous fungi of the genera Crinipellis, Marasmius, and Maramiellus. Canadian Journal of Microbiology, 42, 1179–1183.
Lee, C. L., Hung, H. K., Wang, J. J., & Pan, T. M. (2007). Improving the ratio of monacolin K to citrinin production of Manascus purpureus NTU 568 under dioscorea medium through the mediation of pH value and ethanol addition. Journal of Agriculture Food Chemistry, 55, 6493–6502.
Li, X. F., Cullen, W. R., Reimer, K. J., & Le, X. C. (1996). Microbial degradation of pyrene and characterization of a metabolite. Science Total Environment, 177, 17–29.
Novotny, C., Erbanova, P., Cajthaml, T., Rothschild, N., Dosoretz, C., & Sasek, V. (2000). Irpex lacteus, a white rot fungus applicable to water and soil bioremediation. Applied Microbiology and Biotechnology, 54, 850–853.
Obayori, O. S., Ilori, M. O., Adebusoye, S. A., Oyetibo, G. O., & Amund, O. O. (2008). Pyrene-degradation potentials of Pseudomonas species isolated from polluted tropical soils. World Journal of Microbiology and Biotechnology, 24, 2639–2646.
Pointing, S. B. (2001). Feasibility of bioremediation by white-rot fungi. Applied Microbiology and Biotechnology, 57, 20–33.
Pozdnyakova, N. N., Nikiforova, S. V., Makarov, O. E., Chernyshova, M. P., Pankin, K. E., & Turkovskaya, O. V. (2010). Influence of cultivation conditions on pyrene degradation by the fungus Pleurotus ostreatus D1. World Journal of Microbiology and Biotechnology, 26, 205–211.
Rajderkar, N. (1966). Certain chemical requirements for growth and sporulation of Alternaria species. Mycopathologia, 30, 172–176.
Ravelet, C., Krivobok, S., Sage, L., & Steiman, R. (2000). Biodegradation of pyrene by sediment fungi. Chemosphere, 40, 557–563.
Ryu, W. R., Shim, S. H., Jang, M. Y., Jeon, Y. J., Oh, K. K., & Cho, M. H. (2000). Biodegradation of pentachlorophenol by white rot fungi under ligninolytic and nonligninolytic conditions. Biotechnology Bioprocess Engineering, 5, 211–214.
Saraswathy, A., & Hallberg, R. (2002). Degradation of pyrene by indigenous fungi from a former gasworks site. Microbiology Letters, 210, 227–232.
Sarma, P. M., Duraja, P., Deshpande, S., & Lal, B. (2010). Degradation of pyrene by an enteric bacterium, Leclercia adecarboxyata PS4040. Biodegradation, 21, 59–69.
Sartoros, C., Yerushalmi, L., Beron, P., & Guiot, S. R. (2005). Effects of surfactant and temperature on biotransformation kinetics of anthracene and pyrene. Chemosphere, 61, 1042–1050.
Sheng, X., Chen, X., & He, L. (2008). Characteristics of an endophytic pyrene-degrading bacterium of Enterobacter sp. 12J1 from Allium macrostemon Bunge. International Biodetermination and Biodegradation, 62, 88–95.
Sho, M., Hamel, C., & Greer, C. W. (2004). Two distinct gene clusters encode pyrene degradation in Mycobacterium sp. strain S65. FEMS Microbiology Ecology, 48, 209–220.
Vila, J., Lopez, Z., Sabate, J., Minguillon, C., Solanas, A. M., & Grifoll, M. (2001). Identification of a novel metabolite in the degradation of pyrene by Mycobacterium sp. strain AP1: actions of the isolate on two and three ring PAH. Applied Microbiology and Biotechnology, 67, 5497–5505.
Wiesche, C. I. D., Martens, R., & Zadrazil, R. (1996). Two-step degradation of pyrene by white-rot fungi and soil microorganisms. Applied Microbiology and Biotechnology, 46, 653–659.
Xu, S. Y., Chen, Y. X., Lin, K. F., Chen, X. C., Lin, Q., Li, F., et al. (2009). Removal of pyrene from contaminated soils by white clover. Pedosphere, 19, 265–272.
Yan, J., Wang, L., Fu, P. P., & Yu, H. (2004). Photomutagenicity of 16 polycyclic aromatic hydrocarbons from the US EPA priority pollutant list. Mutation Research, 557, 99–108.
Zhang, D., & Zhu, L. Z. (2012). Effects of tween 80 on the removal, sorption and biodegradation of pyrene by Klebsiella oxytoca PYR-a. Environmental Pollution, 164, 169–174.
Acknowledgments
This project was partly financially supported by the Universiti Teknologi Malaysia (02H65) and the Ministry of High Education, Malaysia (4L053).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Teh, Z.C., Hadibarata, T. Enhanced Degradation of Pyrene and Metabolite Identification by Pleurotus eryngii F032. Water Air Soil Pollut 225, 1909 (2014). https://doi.org/10.1007/s11270-014-1909-x
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11270-014-1909-x