Abstract
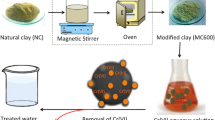
Organoclays possess unique adsorption behaviour towards hydrophobic organic contaminants. They can also remediate ionic contaminants such as heavy metals and metalloids. The objective of the present study was to prepare organoclay and organoclay mixtures efficient to adsorb both cationic and anionic contaminants. The adsorbents were characterised by X-ray diffraction and infrared spectroscopy. Trivalent (Cr3+) and hexavalent (Cr2O7 2−) chromium were selected as the model contaminants representing cationic and anionic properties. Bentonite modified with cationic surfactant hexadecyl trimethylammonium bromide at double the cation exchange capacity of the clay remarkably improved Cr2O7 2− adsorption capacity (as high as 0.49 mmol g−1). Similarly, its modification with anionic surfactant sodium dodecyl sulphate at the same dosage improved Cr3+ adsorption (as high as 0.36 mmol g−1). When these two organoclays were physically mixed in equal proportions (1:1), the resultant organoclay mixture efficiently adsorbed both Cr3+ (as high as 0.21 mmol g−1) and Cr2O7 2− (as high as 0.32 mmol g−1) implying that the mixture could remediate both anionic and cationic contaminants simultaneously. The adsorption of Cr3+ by the organoclay and organoclay mixture fitted well to the Langmuir isothermal model whereas the adsorption of Cr2O7 2− fitted well to the Freundlich model.



Similar content being viewed by others
References
Akar, S. T., Yetimoglu, Y., & Gedikbey, T. (2009). Removal of chromium (VI) ions from aqueous solutions by using Turkish montmorillonite clay: Effect of activation and modification. Desalination, 244, 97–108.
Andini, S., Cioffi, R., Montagnaro, F., Pisciotta, F., & Santor, L. (2006). Simultaneous adsorption of chlorophenol and heavy metal ions on organophilic bentonite. Applied Clay Science, 31, 126–133.
Atia, A. A. (2008). Adsorption of chromate and molybdate by cetylpyridinium bentonite. Applied Clay Science, 41, 73–84.
Bate, B., & Burns, S. E. (2010). Effect of total organic carbon content and structure on the electrokinetic behavior of organoclay suspensions. Journal of Colloid and Interface Science, 343, 58–64.
Bedelean, H., Maicaneanu, A., Burca, S., & Stanca, M. (2010). Removal of heavy metal ions from wastewaters using natural clays. Clay Minerals, 44, 487–495.
Bergaya, F., & Lagaly, G. (2006). Chapter 1 general introduction: Clays, clay minerals, and clay science. In: F. Bergaya, B. K. G. Theng, & G. Lagaly (eds.), Developments in clay science, Elsevier, pp. 1–18.
Bhattacharyya, K. G., & Gupta, S. S. (2008). Adsorption of a few heavy metals on natural and modified kaolinite and montmorillonite: A review. Advances in Colloid and Interface Science, 140, 114–131.
Borden, D., & Giese, R. F. (2001). Baseline studies of the clay minerals society source clays: Cation exchange capacity measurements by the ammonia-electrode method. Clays and Clay Minerals, 49, 444–445.
Churchman, G. J., Gates, W. P., Theng, B. K. G., & Yuan, G. (2006). Chapter 11.1 clays and clay minerals for pollution control. In: F. Bergaya, B. K. G. Theng, & G. Lagaly (Eds.), Developments in clay science, Elsevier, pp. 625–675.
de Paiva, L. B., Morales, A. R., & Valenzuela Díaz, F. R. (2008). Organoclays: Properties, preparation and applications. Applied Clay Science, 42, 8–24.
He, H., Guo, J., Xie, X., & Peng, J. (2000). Experimental study of the selective adsorption of heavy metals onto clay minerals. Chinese Journal of Geochemistry, 19, 105–109.
He, H., Frost, R. L., Bostrom, T., Yuan, P., Duong, L., Yang, D., Xi, Y., & Kloprogge, J. T. (2006a). Changes in the morphology of organoclays with HDTMA+ surfactant loading. Applied Clay Science, 31, 262–271.
He, H., Zhou, Q., Martens, W. N., Kloprogge, T. J., Yuan, P., Xi, Y., Zhu, J., & Frost, R. L. (2006b). Microstructure of HDTMA+-modified montmorillonite and its influence on sorption characteristics. Clays and Clay Minerals, 54, 689–696.
Keith, L., & Telliard, W. (1979). ES&T special report: Priority pollutants: I—A perspective view. Environmental Science and Technology, 13, 416–423.
Khenifi, A., Bouberka, Z., Sekrane, F., Kameche, M., & Derriche, Z. (2007). Adsorption study of an industrial dye by an organic clay. Adsorption, 13, 149–158.
Krishna, B. S., Murty, D. S. R., & Jai Prakash, B. S. (2000). Thermodynamics of chromium(VI) anionic species sorption onto surfactant-modified montmorillonite clay. Journal of Colloid and Interface Science, 229, 230–236.
Krishna, B. S., Murty, D. S. R., & Jai Prakash, B. S. (2001). Surfactant-modified clay as adsorbent for chromate. Applied Clay Science, 20, 65–71.
Li, Z., Willms, C. A., & Kniola, K. (2003). Removal of anionic contaminants using surfactant-modified palygorskite and sepiolite. Clays and Clay Minerals, 51, 445–451.
Li, Z., Beachner, R., McManama, Z., & Hanlie, H. (2007). Sorption of arsenic by surfactant-modified zeolite and kaolinite. Microporous and Mesoporous Materials, 105, 291–297.
Limousin, G., Gaudet, J. P., Charlet, L., Szenknect, S., Barthès, V., & Krimissa, M. (2007). Sorption isotherms: A review on physical bases, modeling and measurement. Applied Geochemistry, 22, 249–275.
Lin, S. H., & Juang, R. S. (2002). Heavy metal removal from water by sorption using surfactant-modified montmorillonite. Journal of Hazardous Materials, 92, 315–326.
Megharaj, M., Naidu, R., Xi, Y., & Sarkar, B. (2010). Modified clay sorbents. CRC CARE Pty Ltd., International Patent. World Intellectual Property Organisation International Bureau, International Publication Number: WO 2010/065996 A1.
Naidu, R., Sumner, M. E., & Harter, R. D. (1998). Sorption of heavy metals in strongly weathered soils: An overview. Environmental Geochemistry and Health, 20, 5–9.
Oyanedel-Craver, V. A., & Smith, J. A. (2006). Effect of quaternary ammonium cation loading and pH on heavy metal sorption to Ca bentonite and two organobentonites. Journal of Hazardous Materials, 137, 1102–1114.
Oyanedel-Craver, V. A., Fuller, M., & Smith, J. A. (2007). Simultaneous sorption of benzene and heavy metals onto two organoclays. Journal of Colloid and Interface Science, 309, 485–492.
Sarkar, B., Xi, Y., Megharaj, M., Krishnamurti, G. S. R., & Naidu, R. (2010a). Synthesis and characterisation of novel organopalygorskites for removal of p-nitrophenol from aqueous solution: Isothermal studies. Journal of Colloid and Interface Science, 350, 295–304.
Sarkar, B., Xi, Y., Megharaj, M., Krishnamurti, G. S. R., Rajarathnam, D., & Naidu, R. (2010b). Remediation of hexavalent chromium through adsorption by bentonite based Arquad® 2HT-75 organoclays. Journal of Hazardous Materials, 183, 87–97.
Sarkar, B., Megharaj, M., Xi, Y., & Naidu, R. (2011a). Structural characterisation of Arquad® 2HT-75 organobentonites: Surface charge characteristics and environmental application. Journal of Hazardous Materials, 195, 155–161.
Sarkar, B., Xi, Y., Megharaj, M., & Naidu, R. (2011b). Orange II adsorption on palygorskites modified with alkyl trimethylammonium and dialkyl dimethylammonium bromide—An isothermal and kinetic study. Applied Clay Science, 51, 370–374.
Sarkar, B., Megharaj, M., Xi, Y., & Naidu, R. (2012a). Surface charge characteristics of organo-palygorskites and adsorption of p-nitrophenol in flow-through reactor system. Chemical Engineering Journal, 185–186, 35–43.
Sarkar, B., Naidu, R., Rahman, M., Megharaj, M., & Xi, Y. (2012b). Organoclays reduce arsenic bioavailability and bioaccessibility in contaminated soils. Journal of Soils and Sediments, 12, 704–712.
Sarkar, B., Xi, Y., Megharaj, M., Krishnamurti, G. S. R., Bowman, M., Rose, H., & Naidu, R. (2012c). Bio-reactive organoclay: A new technology for environmental remediation. Critical Reviews in Environmental Science and Technology, 42, 435–488.
Sethunathan, N., Megharaj, M., Smith, L., Kamaludeen, S. P. B., Avudainayagam, S., & Naidu, R. (2005). Microbial role in the failure of natural attenuation of chromium(VI) in long-term tannery waste contaminated soil. Agriculture, Ecosystem and the Environment, 105, 657–661.
Stathi, P., Litina, K., Gournis, D., Giannopoulos, T. S., & Deligiannakis, Y. (2007). Physicochemical study of novel organoclays as heavy metal ion adsorbents for environmental remediation. Journal of Colloid and Interface Science, 316, 298–309.
Sullivan, E. J., Bowman, R. S., & Legiec, I. A. (2003). Sorption of arsenic from soil-washing leachate by surfactant-modified zeolite. Journal of Environmental Quality, 32, 2387–2391.
Tillman, F. D., Bartelt-Hunt, S. L., Smith, J. A., & Alther, G. R. (2004). Evaluation of an organoclay, an organoclay-anthracite blend, clinoptilolite, and hydroxy-apatite as sorbents for heavy metal removal from water. Bulletin of Environmental Contamination and Toxicology, 72, 1134–1141.
USDHHS (1987). Notice of the first priority list of hazardous substances that will be the subject of toxicological profiles. In: USDHHS-USEPA (Ed.) Federal Register, 52, pp. 12866–12874.
Vaia, R. A., Teukolsky, R. K., & Giannelis, E. P. (1994). Interlayer structure and molecular environment of alkylammonium layered silicates. Chemistry of Materials, 6, 1017–1022.
Xi, Y., Ding, Z., He, H., & Frost, R. L. (2004). Structure of organoclays—An X-ray diffraction and thermogravimetric analysis study. Journal of Colloid and Interface Science, 277, 116–120.
Xi, Y., Ding, Z., He, H., & Frost, R. L. (2005). Infrared spectroscopy of organoclays synthesized with the surfactant octadecyltrimethylammonium bromide. Spectrochimica Acta A, 61, 515–525.
Xi, Y., Frost, R. L., & He, H. (2007). Modification of the surfaces of Wyoming montmorillonite by the cationic surfactants alkyl trimethyl, dialkyl dimethyl, and trialkyl methyl ammonium bromides. Journal of Colloid and Interface Science, 305, 150–158.
Xu, L., & Zhu, L. (2009). Structures of OTMA- and DODMA-bentonite and their sorption characteristics towards organic compounds. Journal of Colloid and Interface Science, 331, 8–14.
Zadaka, D., Radian, A., & Mishael, Y. G. (2010). Applying zeta potential measurements to characterize the adsorption on montmorillonite of organic cations as monomers, micelles, or polymers. Journal of Colloid and Interface Science, 352, 171–177.
Zhitkovich, A. (2011). Chromium in drinking water: Sources, metabolism, and cancer risks. Chemical Research in Toxicology, 24, 1617–1629.
Zhu, J., He, H., Guo, J., Yang, D., & Xie, X. (2003). Arrangement models of alkylammonium cations in the interlayer of HDTMA+ pillared montmorillonites. Chinese Science Bulletin, 48, 368–372.
Zhu, J., He, H., Zhu, L., Wen, X., & Deng, F. (2005). Characterization of organic phases in the interlayer of montmorillonite using FTIR and 13C NMR. Journal of Colloid and Interface Science, 286, 239–244.
Acknowledgments
One of the authors (B. S.) is thankful to the University of South Australia for the award of the University President Scholarship (UPS) and to the Cooperative Research Centre for Contamination Assessment and Remediation of the Environment (CRC CARE) for the award of the PhD Fellowship. Help with ICP-OES analysis by Dr. Mohammad Mahmudur Rahman is also gratefully acknowledged.
Author information
Authors and Affiliations
Corresponding author
Additional information
Guest Editors: R Naidu, Euan Smith, MH Wong, Megharaj Mallavarapu, Nanthi Bolan, Albert Juhasz, and Enzo Lombi
This article is part of the Topical Collection on Remediation of Site Contamination
Rights and permissions
About this article
Cite this article
Sarkar, B., Naidu, R. & Megharaj, M. Simultaneous Adsorption of Tri- and Hexavalent Chromium by Organoclay Mixtures. Water Air Soil Pollut 224, 1704 (2013). https://doi.org/10.1007/s11270-013-1704-0
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11270-013-1704-0