Abstract
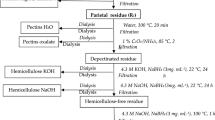
The polysaccharidic moieties of three biosorbents (Douglas fir and argan tree barks and argan endocarp) were selectively oxidized, and the subsequent modified materials were tested for their ability to bind Pb(II) or Cd(II) from aqueous solutions. Chemical modifications consisted in two selective oxidations, alone or in combination, of the following groups: primary alcohols with NaOBr catalyzed by (2,2,6,6-tetramethylpiperidin-1-yl)oxidanyl, and vicinal diols with periodate/chlorite. The sodium chlorite oxidation step induced biosorbent degradation that led to a significant decrease of mass yield. Modified materials, characterized by FT-IR spectroscopy and measurement of surface acidity, were investigated for their adsorption capabilities of Cd(II) and Pb(II). Results were compared to the capabilities of crude materials using the Langmuir adsorption model in terms of affinity (b) and maximum binding capacity (q max). Ion exchange properties were found better for lead than for cadmium before and after chemical modifications. Compared to crude barks, the best results were obtained for Douglas fir barks whose oxidation resulted in significant enhancements of q max up to × 10 in the case of lead.




Similar content being viewed by others
References
Al-Asheh, S., & Duvnjak, Z. (1997). Sorption of cadmium and other heavy metals by pine bark. Journal of Hazardous Materials, 56, 35–51.
Alloway, B. J. (1995). Heavy metals in soils. London: Blackie Academic & Professional.
Argun, M. E., Dursun, S., & Karatas, M. (2009). Removal of Cd(II), Pb(II), Cu(II) and Ni(II) from water using modified pine bark. Desalination, 249, 519–527.
Astier, C., Chaleix, V., Faugeron, C., Ropartz, D., Gloaguen, V., & Krausz, P. (2010). Grafting of aminated oligogalacturonans onto Douglas fir barks. A new route for the enhancement of their lead (II) binding capacities. Journal of Hazardous Materials, 182, 279–285.
Astier, C., Chaleix, V., Faugeron, C., Ropartz, D., Krausz, P., Gloaguen, V. (2012). Biosorption of lead (II) on modified barks explained by the hard and soft acids and bases (HSAB) theory. BioResources, 7(1), 1100–1110.
Bailey, S. E., Olin, T. J., Bricka, R. M., & Adrian, D. D. (1999). A review of potentially low-cost sorbents for heavy metals. Water Research, 33, 2469–2479.
Boehm, H. P. (1966). Chemical identification of surface groups. Advances in Catalysis and Related Subjects, 16, 179–274.
Harman, G., Patrick, R., & Spittler, T. (2007). Removal of heavy metals from polluted waters using lignocellulosic agricultural waste products. Industrial Biotechnology, 3, 366–374.
Kartel, M. T., Kupchik, L. A., & Veisov, B. K. (1999). Evaluation of pectin binding of heavy metal ions in aqueous solutions. Chemosphere, 38, 2591–2596.
Khotimchenko, M., Kovalev, V., & Khotimchenko, Y. (2007). Equilibrium studies of sorption of lead(II) ions by different pectin compounds. Journal of Hazardous Materials, 149, 693–699.
Klein-Koerkamp, C., Granet, R., Zerrouki, R., Villandier, N., Jérôme, F., Barrault, J., & Krausz, P. (2009). Efficient oxidative modification of polysaccharides in water using H2O2 activated by iron sulfophthalocyanine. Carbohydrate Polymers, 78, 938–944.
Lantenois, S., Prelot, B., Douillard, J. M., Szczodrowski, K., & Charbonnel, M. C. (2007). Flow microcalorimetry: experimental development and application to adsorption of heavy metal cations on silica. Applied Surface Science, 253, 5807–5813.
Marchetti, V., Clément, A., Gérardin, P., & Loubinoux, B. (2000). Synthesis and use of esterified sawdusts bearing carboxyl group for removal of cadmium(II) from water. Wood Science and Technology, 34, 167–173.
Martin-Dupont, F., Gloaguen, V., Granet, R., Guilloton, M., Morvan, H., & Krausz, P. (2002). Heavy metal adsorption by crude coniferous barks: a modeling study. Journal of Environmental Science and Health, Part A, 37, 1063–1073.
Mulgund, M. G., Kininge, P. T., Pillai, M. M., & Sanandam, M. R. (2011). Biosorptive removal of heavy (Cd+2, Pb+2 and Cu+2) from aqueous solutions by Cassia angustifolia bark. International Journal of Engineering, Science and Technology, 3, 1642–1647.
Pommerening, K., Rein, H., Bertram, D., & Muller, R. (1992). Estimation of dialdehyde groups in 2,3-dialdehyde bead-cellulose. Carbohydrate Research, 233, 219–223.
Volesky, B., & Holan, Z. R. (1995). Biosorption of heavy metals. Biotechnology Progress, 11, 235–250.
Wang, J., & Chen, C. (2009). Biosorbents for heavy metals removal and their future. Biotechnology Advances, 27, 195–226.
Yu, Z., Jameel, H., Chang, H. M., & Park, S. (2011). The effect of delignification of forest biomass on enzymatic hydrolysis. Bioresource Technology, 102, 9083–9089.
Acknowledgments
The authors gratefully acknowledge Dr. M. Guilloton for his help in the manuscript editing.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Hachem, K., Astier, C., Chaleix, V. et al. Optimization of Lead and Cadmium Binding by Oxidation of Biosorbent Polysaccharidic Moieties. Water Air Soil Pollut 223, 3877–3885 (2012). https://doi.org/10.1007/s11270-012-1156-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11270-012-1156-y