Abstract
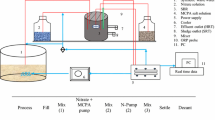
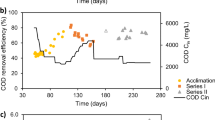
Biodegradation kinetics of 3-chlorophenol (3-CP) were studied in two identical lab scale sequencing batch reactors (SBR) fed with the compound as the sole energy and carbon source and operated at different filling time (1 h for SBR1 and 2 h for SBR2). High removal efficiency was always obtained in both SBRs in the range of feed concentration of 300–960 mg L−1. Increased feed load to 1,200 mg L−1 3-CP could also be removed in SBR1 despite the presence of inhibition, whereas determined failure of SBR2. Long filling time and high biomass concentration were shown to have beneficial effect on process kinetics since they allowed to avoid substrate concentration peaks at the end of the fill phase. However, longer filling time (in the present case higher than 1 h) did not allow to select and enrich robust microbial population. The Haldane equation well fitted the kinetic test data measured in the presence of inhibition, i.e., at 960 and 1,200 mg L−1 3-CP in SBR1.






Similar content being viewed by others
Abbreviations
- SBR:
-
Sequencing batch reactor
- CFSTR:
-
Continuous flow stirred tank reactor
- DO:
-
Dissolved oxygen concentration (mg O2 L−1)
- OUR:
-
Oxygen uptake rate (mg O2/L−1 h−1)
- V TOT :
-
Working volume (L)
- t FILL :
-
Filling time (h)
- V IN :
-
Influent volume (L)
- 3-CP:
-
3-chlorophenol
- C IN :
-
3-CP influent concentration (mg L−1)
- COD:
-
Chemical oxygen demand (mg L−1)
- MLSS:
-
Mixed liquor suspended solids (mg L−1)
- MLVSS:
-
Mixed liquor volatile suspended solids (mg L−1)
- TSS:
-
Total suspended solids (mg L−1)
- VER:
-
Volumetric exchange rate (adim.)
- VSS:
-
Volatile suspended solids (mg L−1)
- Y :
-
Growth yield coefficient (mg VSS mg COD−1)
- b :
-
Endogenous respiration rate (day−1)
- SVI:
-
Sludge volume index (mL g−1)
- F co :
-
Influent organic loading factor (mg COD mg VSS−1 day−1)
- F cv :
-
Volumetric organic loading factor (g COD L−1 day−1)
- k max :
-
Maximum specific removal rate (mg COD mg VSS−1 day−1)
- k*:
-
Real maximum specific removal rate (mg COD mg VSS−1 day−1)
- U(S):
-
Substrate specific utilization rate (mg COD mg VSS−1 h−1)
- K s :
-
Saturation constant (mg L−1)
- K I :
-
Inhibition constant (mg L−1)
- μ :
-
Maximum specific growth rate (day−1)
- μ*:
-
Real maximum specific growth rate (day−1)
- C*:
-
Critical substrate concentration (mg L−1)
- \( C_{{3} - {\rm{CP,}}i}^{{ \exp }} \) :
-
Experimental 3-CP concentration (mg L−1)
- \( C_{{3} - {\rm{CP,}}i}^{\rm{cal}} \) :
-
Modeled 3-CP concentration (mg L−1)
References
Andrews, J. F. (1968). A mathematical model for the continuous culture of microorganisms utilizing inhibitory substrates. Biotechnology Bioengineering, 10(6), 707–723.
APHA/AWWA. (1998). Standard methods for the examination of water and wastewater (20th ed.). Washington: American Public health Association.
Carucci, A., Chiavola, A., Majone, M., & Rolle, E. (1999). Treatment of tannery wastewater in a sequencing batch reactor. Water Science and Technology, 40(1), 253–259.
Chiavola, A., McSwain, B. S., Irvine, R. L., Boni, M. R., & Baciocchi, R. (2003). Biodegradation of 3-chlorophenol in a Sequencing Batch Reactor. Journal of Environmental Science & Health, Part A, A38(10), 2113–2123.
Chiavola, A., Baciocchi, R., Irvine, R. L., Gavasci, R., & Sirini, P. (2004). Aerobic biodegradation of 3-chlorophenol in a Sequencing Batch Reactor. Effect of cometabolism. Water Science and Technology, 50(10), 235–242.
Chiesa, S. C., Irvine, R. L., & Manning, J. F. (1984). Feast/famine growth environments and activated sludge population selection. Biotechnology and Bioengineering, XXVII, 562–569.
Eweis, J. B., Ergas, S. J., Chang, D. P. Y., & Schroeder, E. D. (1998). Bioremediation principles. New York: McGraw-Hill.
Haggblom, M. M., & Valo, R. J. (1995). Microbial transformation and degradation of organic chemicals. In L. Y. Young & C. E. Cerniglia (Eds.), Bioremediation of chlorophenol wastes (pp. 389–434). New York: Wiley-Liss, Inc.
Lee, C. M., Lu, C. J., & Chuang, M. S. (1994). Effects of immobilized cells on the biodegradation of chlorinated phenols. Water Science and Technology, 30(9), 87–90.
Lu, C. J., & Tsai, Y. H. (1993). The effects of a secondary carbon source on the biodegradation of recalcitrant compounds. Water Science and Technology, 28(7), 97–101.
Lu, C. J., Lee, C. M., & Huang, C. Z. (1996). Biodegradation of chlorophenols by immobilized pure-culture microorganisms. Water Science and Technology, 34(10), 67–72.
Katayama-Hirayama, K., Tobita, S., & Hirayama, K. (1994). Biodegradation of phenol and monochlorophenols by yeast Rhodotorula Glutinis. Water Science and Technology, 30(9), 59–66.
Macé, S., & Mata-Alvarez, J. (2002). Utilization of SBR technology for wastewater treatment: An overview. Industrial & Engineering Chemistry Research, 41(23), 5539–5553.
Metcalf and Eddy. (2003). Wastewater engineering. Treatment and Reuse (4th ed.). New York: McGraw-Hill.
Nuhoglu, A., & Beste, Y. (2005). Modelling of phenol removal in a batch reactor. Process Biochemistry, 40, 1233–1239.
Tomei, M. C., Annesini, M. C., Luberti, R., Cento, G., & Senia, A. (2003). Kinetics of 4-nitrophenol biodegradation in a sequencing batch reactor. Water Research, 37, 3803–3814.
Tomei, M. C., Annesini, M. C., & Bussoletti, S. (2004). 4-nitrophenol biodegradation in a sequencing batch reactor: kinetic study and effect of filling time. Water Research, 38, 375–384.
Wilderer, P. A., Irvine, R. L., & Goronszy, M. C. (2001). Sequencing batch reactor technology. Scientific and technical report N. 10. London: IWA.
Wei, Y., Van Houten, R. T., Borger, A. R., Eikelboom, D. H., & Fan, Y. (2003). Minimization of excess sludge production for biological wastewater treatment. Water Research, 37, 4453–4467.
Yang, X. F., Xie, M. L., & Liu, Y. (2003). Metabolic uncouplers reduce excess sludge production in an activated sludge process. Process Biochemistry, 38(9), 1373–1377.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Chiavola, A., Baciocchi, R. & Barducci, F. 3-Chlorophenol Biodegradation in a Sequencing Batch Reactor: Kinetic Study and Effect of the Filling Time. Water Air Soil Pollut 212, 219–229 (2010). https://doi.org/10.1007/s11270-010-0334-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11270-010-0334-z