Abstract
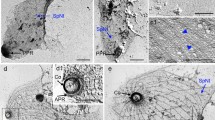
The first merogony of Eimeria bovis takes place in lymphatic endothelial cells of the ileum, resulting in the formation of macromeronts up to 250 μm. In this study, we investigated the host cell cytoskeleton (actin filaments, microtubules, spectrin, vimentin intermediate filaments) associated with parasitic development in vitro by confocal laser scanning microscopy (CLSM) using primary bovine umbilical vein endothelial cells (BUVEC) and bovine spleen lymphatic endothelial cells (BSLEC) as host cells. No prominent changes in the host cell cytoskeleton were detected 1–3 days after E. bovis sporozoite invasion. With ongoing meront maturation a significant increase in microtubules and actin filaments close to the parasitophorous vacuole (PV) was found. Mature macromeronts within the PV were completely enclosed by these cytoskeletal elements. Our findings suggest, that in order to guarantee the survival of the host cell on the enlargement of macromeronts, E. bovis needs not only to augment but also to rearrange its cytoskeletal system.


Similar content being viewed by others
Abbreviations
- BSA:
-
bovine serum albumin
- BSLEC:
-
bovine spleen lymphatic endothelial cells
- BUVEC:
-
bovine umbilical vein endothelial cells
- CLSM:
-
confocal laser scanning microscopy
- DMSO:
-
dimethylsulphoxide
- ECGM:
-
endothelial cell growth medium
- FCS:
-
foetal calf serum
- PBS:
-
phosphate-buffered saline
- p. i.:
-
post-infection
- PV:
-
parasitophorous vacuole
- RT:
-
room temperature
References
Andrade, E.F., Stumbo, A.C., Monteiro-Leal, L.H., Carvalho, L. and Barbosa, H.S., 2001. Do microtubules around the Toxoplasma gondii-containing parasitophorous vacuole in skeletal muscle cells form a barrier for the phagolysosomal fusion?, Journal of Submicroscopic Cytology and Pathology, 33(3), 337–341
Bart, B.J. and Lussky, R.C., 2005. Bart syndrome with associated anomalies, American Journal of Perinatology, 22(2), 365–369
Bennett, V., 1985. The membrane skeleton of human erythrocytes and its implication for more complex cells, Annual Review of Biochemistry, 54, 273–304
Bonnet, C., Boucher, D., Lazereg, S., Pedrotii, B., Islam, K., Denoulet, P. and Larcher, J.C., 2001. Differential binding regulation of microtubule-associated proteins MAP1A, MAP1B, and MAP2 by tubulin polyglutamylation, Journal of Biological Chemistry, 276, 12839–12848
Coppens, I., Dunn, J.D., Romano, J.D., Pypaert, M., Zhang, H., Boothroyd, J.C. and Joiner, K.A., 2006. Toxoplasma gondii sequesters lysosomes from mammalian hosts in the vacuolar space, Cell, 125, 261–274.
Daugschies, A. and Najdrowski, M., 2005. Eimeriosis in cattle: current understanding, Journal of Veterinary Medicine, 52, 417–427
Deng, M., Lancto, C.A. and Abrahamsen, M.S., 2004. Cryptosporidium parvum regulation of human epithelial cell expression, International Journal of Parasitology, 34, 73–82
Downing, K.H. and Nogales, E., 1998a. Tubulin and microtubule structure, Current Opinion in Cell Biology, 10, 16–22
Downing, K.H. and Nogales, E., 1998b. Tubulin structure: insights into microtubule properties and functions, Current Opinion in Structural Biology, 8, 785–791
Elliot, D.A. and Clark, D.P., 2000. Cryptosporidium parvum induces host cell actin accumulation at the host-parasite interface, Infection and Immunity, 68, 2315–2322
Elliot, D.A., Coleman, D.J., Lane, M.A., May, R.C., Machesky, L.M. and Clark, D.P., 2001. Cryptosporidium parvum infection requires host cell actin polymerization, Infection and Immunity, 69, 5940–5942
Faber, J.E., Kollmann, D., Heise, A., Bauer, C., Failing, K., Bürger, H.J. and Zahner, H., 2002. Eimeria infections in cows in the periparturient phase and their calves: oocyst excretion and levels of specific serum and colostrum antibodies, Veterinary Parasitology, 104(1), 1–17
Feng, H., Nie, W., Bonilla, R., Widmer, G., Sheoran, A. and Tzipori, S., 2006. Quantitative tracking of Cryptosporidium infection in cell culture, Journal of Parasitology, 92, 1350–1354
Fiege, N., Klatte, D., Kollmann, D., Zahner, H. and Bürger, H.J., 1992. Eimeria bovis in cattle: colostral transfer of antibodies and immune response to experimental infections, Parasitology Research, 78, 32–38
Forney, J.R., DeWall, D.B.,Yang, S., Speer, C.A. and Healy, M.C., 1999. A role for host phoshoinositide 3-kinase and cytoskeletal remodelling during Cryptosporidium parvum infection, Infection and Immunity, 67, 844–852
Ghazali, M., Rodier, M.H., el Moudni, B.E., Babin, P., Fernandez, B. and Jacquemin, J.L., 1995. Detection and immunolocalization of human erythrocyte spectrin immunoanalogues in Toxoplasma gondii (Protozoan, Parasite), Journal of Eukaryotic Microbiology, 42, 427–433
Halonen, S.K. and Weidner, E., 1994. Overcoating of Toxoplasma parasitophorous vacuoles with host cell vimentin type intermediate filaments, Journal of Eukaryotic Microbiology, 41, 65–71
Halonen, S.K., Weiss, L.M. and Chiu, F.C., 1998. Association of host cell intermediate filaments with Toxoplasma gondii cysts in murine astrocytes in vitro, International Journal of Parasitology, 28, 815–823
Hammond, D.M., Davis, L.R. and Bowmann, L., 1964. Experimental infections with Eimeria bovis in calves, American Journal of Veterinary Research, 5, 303–311
Hammond, D.M. and Fayer, R., 1968. Cultivation of Eimeria bovis in three established cell lines and bovine tracheal cell line culture, Journal of Parasitolology, 54, 559–568
Hermosilla, C., Barbisch, B., Heise, A., Kowalik, S. and Zahner, H., 2002. Development of Eimeria bovis in vitro: suitability of several bovine, human and porcine endothelial cell lines, bovine gastrointestinal, Madin-Darby bovine kidney (MDBK) and African green monkey kidney (VERO) cells, Parasitology Research, 88, 301–307
Hermosilla, C., Zahner, H. and Taubert, A., 2006. Eimeria bovis modulates adhesion molecule gene transcription in and PMN adhesion to infected bovine endothelial cells, International Journal of Parasitology, 36, 423–431
Janmey, P.A., 1991. Mechanical properties of cytoskeletal polymers, Current Opinion in Cell Biology, 3, 4–11
Kelly, R., 1990. Microtubules, membrane traffic and cell organization, Cell, 61, 5–7
Kreis, T.E., 1990. Role of microtubules in the organization of the Golgi apparatus, Cell Motility and the Cytoskeleton, 15, 67–70
Kreitzer, G., Liao, G. and Gundersen, G.G., 1999. Detyrosination of tubulin regulates the interaction of intermediate filaments with microtubules in vivo via a kinesin-dependent mechanism, Molecular Biology of the Cell, 10, 1105–1118
Lingelbach, K. and Joiner, K.A., 1998. The parasitophorous vacuole membrane surrounding Plasmodium and Toxoplasma: an unusual compartment in infected cells, Journal of Cell Science, 111, 1447–1475
Matthews, G. and Allen, M., 2005. Epidermolysis bullosa in cattle in the UK, Veterinary Record, 157, 456
Melo, E.J., Carvalho, T.M. and De Souza, W., 2001. Behavior of microtubules in cells infected with Toxoplasma gondii, Biocell, 25(1), 53–59
Morisaki, J.H., Heuser, J.E. and Sibley, L.D., 1995. Invasion of Toxoplasma gondii occurs by active penetration of the host cell, Journal of Cell Science, 108, 2457–2464
Morrissette, N.S., Mitra, A., Sept, D. and Sibley, L.D., 2004. Dinitroanilines bind a-tubulin to disrupt microtubules, Molecular Biology of the Cell, 15, 1960–1968
Reduker, D.W. and Speer, C.A., 1986. Antigens of in-vitro produced first generation merozoites of Eimeria bovis (Apicomplexa), Journal of Parasitology, 72, 782–785
Sibley, L.D., 2003. Toxoplasma gondii: perfecting an intracellular life style, Traffic, 4(9), 581–586
Speer, C.A., Reduker, D.W., Burgess, D.E., Whitmire, W.M. and Splitter, G.A., 1985. Lymphokine-induced inhibition of Eimeria bovis and Eimeria papillata (Apicomplexa) in cultured bovine monocytes, Infection and Immunity, 50, 566–571
Steinert, P.M. and Roop, D.R., 1988. Molecular and cellular biology of intermediate filaments, Annual Review of Biochemistry, 57, 593–625
Acknowledgements
We acknowledge the kind supply of BSLEC by K. Preissner (Justus Liebig University Giessen) and of bovine umbilical cords by H. Bollwein and A. Koch (University of Veterinary Medicine Hannover). We are indebted to B. Hofmann, C. Scheld and M. Buss for their excellent technical assistance in cell culture. Further, we are grateful to K. Failing (Justus Liebig University Giessen) for support in statistical analyses of the data.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Hermosilla, C., Schröpfer, E., Stowasser, M. et al. Cytoskeletal changes in Eimeria bovis-infected host endothelial cells during first merogony. Vet Res Commun 32, 521–531 (2008). https://doi.org/10.1007/s11259-008-9054-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11259-008-9054-x