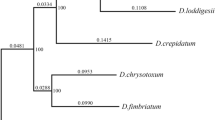
Abstract
A favorable leaf water balance can be achieved through coordinated variation in multiple leaf traits. However, the role of evolutionary history in producing this trait coordination is not well understood, especially in epiphytic species. To address these limitations, we measured 11 leaf traits for 19 Dendrobium species grown in the same environment, and used phylogenetically independent contrasts to test how the co-evolution of these traits has contributed to leaf water balance in epiphytes. Our results show that more closely correlated species had similar habitats, geographical distribution, and some leaf traits (leaf density, upper cuticle thickness, and stomatal index), whereas distantly related species did not exhibit such similarities. Species originating from forests below the altitude of 1,500 m exhibited smaller leaf area, stomatal area, and stomatal index, but thicker leaves than those from forests above the altitude of 1,500 m. Stomatal density was evolutionarily correlated with vein density, upper and lower epidermal thicknesses, and leaf density. Leaf thickness was significantly correlated with stomatal density and vein density, while leaf area was significantly correlated with epidermal cell thicknesses, stomatal density, and stomatal area. These results found that both the environment and evolutionary history significantly affected leaf functional traits and ecological characteristics in Dendrobium, and supported the hypothesis that co-evolution in leaf traits is an important contributor to leaf water balance in Dendrobium. This study provides new insights into the evolution of ecological strategies in epiphytic orchids.




Similar content being viewed by others
References
Ackerly DD (1999) Comparative plant ecology and the role of phylogenetic information. In: Press M, Scholes JD, Barker MG (eds) Physiological plant ecology. Blackwell Science, Oxford, pp 391–414
Ackerly DD, Reich PB (1999) Convergence and correlations among leaf size and function in seed plants: a comparative test using independent contrasts. Am J Bot 86:1272–1281
Akaike H (1974) A new look at the statistical model identification. IEEE Trans Autom Control 19:716–723
Baraloto C, Paine CET, Patiño S, Bonal D, Hérault B, Chave J (2010) Functional trait variation and sampling strategies in species-rich plant communities. Funct Ecol 24:208–216
Beerling DJ, Kelly CK (1996) Evolutionary comparative analyses of the relationship between leaf structure and function. New Phytol 134:35–51
Blomberg SP, Garland JR, Ives AR (2003) Testing for phylogenetic signal in comparative data: behavioral traits are more labile. Evolution 57:717–745
Blonder B, Buzzard V, Simova I et al (2012) The leaf-area shrinkage effect can bias paleoclimate and ecology research. Am J Bot 99:1756–1763
Brodribb TJ, Jordan GJ (2011) Water supply and demand remain balanced during leaf acclimation of Nothofagus cunninghamii trees. New Phytol 192:437–448
Brodribb TJ, Feild TS, Jordan GJ (2007) Leaf maximum photosynthetic rate and venation are linked by hydraulics. Plant Physiol 144:1890–1898
Conran JG, Bannister JM, Lee D (2009) Earliest orchid macrofossils: early Miocene Dendrobium and Earina (Orchidaeae: Epidendroideae) from New Zealan. Am J Bot 96:466–474
Cunningham SA, Summerhayes B, Westoby M (1999) Evolutionary divergences in leaf structure and chemistry, comparing rainfall and soil nutrient gradients. Ecol Monogr 69:569–588
Dunbar-Co S, Sporck MJ, Sack L (2009) Leaf trait diversification and design in seven rare taxa of the Hawaiian Plantago radiation. Int J Plant Sci 170:61–75
Edwards EJ (2006) Correlated evolution of stem and leaf hydraulic traits in Pereskia (Cactaceae). New Phytol 172:479–489
Felsenstein J (1985) Phylogenies and the comparative method. Am Nat 125:1–15
Franks PJ, Beerling DJ (2009) CO2-forced evolution of plant gas exchange capacity and water-use efficiency over the Phanerozoic. Geobiology 7:227–236
Freudenstein JV, Rasmussen FN (1999) What does morphology tell us about orchid relationships? A cladistic analysis. Am J Bot 86:225–248
Givnish TJ, Montgomery RA, Goldstein G (2004) Adaptive radiation of photosynthetic physiology in the Hawaiian lobeliads: light regimes, static light responses, and whole-plant compensation points. Am J Bot 91:228–246
Hetherington AM, Woodward FI (2003) The role of stomata in sensing and driving environmental change. Nature 424:901–908
Higgins D, Thompson J, Gibson T, Thompson JD, Higgins DG, Gibson TJ (1994) CLUSTALW: improving the sensitivity of progressive multiple sequence alignment through sequence weighting, position-specific gap penalties and weight matrix choice. Nucleic Acids Res 22:4673–4680
Hodges SA, Derieg NJ (2009) Adaptive radiation: from field to genomic studies. PNAS 106:9947–9954
Hsu C, Horng F, Kuo C (2002) Epiphyte biomass and nutrient capital of a moist subtropical forest in north-eastern Taiwan. J Trop Ecol 18:659–670
Ji ZH (1999) Flora of China. In: Ji ZH, Chen XQ, Lang KY, Zhu GH (eds). Subtrib. Dendrobiinae Lindl., Dendrobium Sw. Science Press of China, Beijing, pp 67–146
Kelly CK, Beerling DJ (1995) Plante life form, stomatal density and taxonomic relatedness: a reanalysis of Salisbury (1927). Funct Ecol 9:422–431
Kembel SW, Cahill JF Jr (2011) Independent evolution of leaf and root traits within and among temperate grassland plant communities. PLoS ONE 6:e19992
Kembel SW, Cowan PD, Helmus MR, Cornwell WK, Morlon H, Ackerly DD, Blomberg SP, Webb CO (2010) Picante: R tools for integrating phylogenies and ecology. Bioinformatics 26:1463–1464
Kerstiens G (1996) Cuticular water permeability and its physiological significance. J Exp Bot 47:1813–1832
Laube S, Zotz G (2003) Which abiotic factors limit vegetative growth in a vascular epiphyte? Funct Ecol 17:598–604
Nicotra AB, Cosgrove MJ, Cowling A, Schlichting CD, Jones CS (2008) Leaf shape linked to photosynthetic rates and temperature optima in South African Pelargonium species. Oecologia 154:625–635
Niinemets Ü, Sack L (2006) Structural determinants of leaf light-harvesting capacity and photosynthetic potentials. Progress in botany. Springer, Berlin, pp 385–419
Poorter H, Niinemets Ü, Poorter L, Wright IJ, Villar R (2009) Causes and consequences of variation in leaf mass per area (LMA): a meta-analysis. New Phytol 182:565–588
Posada D, Crandall KA (1998) Modeltest: testing the model of DNA substitution. Bioinformatics 14:817–818
Price T (1997) Correlated evolution and independent contrasts. Philos Trans R Soc Lond B Biol Sci 352:519–529
Riederer M, Schreiber L (2001) Protecting against water loss: analysis of the barrier properties of plant cuticles. J Exp Bot 52:2023–2032
Ronquist F, Huelsenbeck JP (2003) MrBayes 3: Bayesian phylogenetic inference under mixed models. Bioinformatics 19:1572–1574
Royer DL (2001) Stomatal density and stomatal index as indicators of paleoatmospheric CO2 concentration. Rev Palaeobot Palynol 114:1–28
Sack L, Frole K (2006) Leaf structural diversity is related to hydraulic capacity in tropical rain forest trees. Ecology 87:483–491
Sack L, Holbrook NM (2006) Leaf hydraulics. Annu Rev Plant Biol 57:361–381
Sack L, Cowan PD, Jaikumar N, Holbrook NM (2003) The ‘hydrology’ of leaves: co-ordination of structure and function in temperate woody species. Plant Cell Environ 26:1343–1356
Sack L, Scoffoni C, McKown AD, Frole K, Rawls M, Havran JC, Tran H, Tran T (2012) Developmentally based scaling of leaf venation architecture explains global ecological patterns. Nat Commun 3:837
Santiago LS, Kim SC (2009) Correlated evolution of leaf shape and physiology in the woody Sonchus alliance (Asteraceae: Sonchinae) in Macronesia. Int J Plant Sci 170:83–92
Scoffoni C, Rawls M, McKown A, Cochard M, Sack L (2011) Decline of leaf hydraulic conductance with dehydration: relationship to leaf size and venation architecture. Plant Physiol 156:832–843
Stern WL, Morris MW, Judd WS (1994) Anatomy of the thick leaves in Dendrobium section Rhizobium (Orchidaceae). Int J Plant Sci 155:716–729
Stuntz S, Simon U, Zotz G (2002) Rainforest air-conditioning: the moderating influence of epiphytes on the microclimate in tropical tree crowns. Int J Biometeorol 46:53–59
Uhl D, Mosbrugger V (1999) Leaf venation density as a climate and environmental proxy: a critical review and new data. Palaeogeogr Palaeoecol 149:15–26
Watkins JE Jr, Cardelús CL (2012) Ferns in an angiosperm world: cretaceous radiation into the epiphytic niche and diversification on the forest floor. Int J Plant Sci 173:695–710
Webb CO, Ackerly DD, Kembel SW (2008) Phylocom: software for the analysis of phylogenetic community structure and trait evolution. Bioinformatics 24:2098–2100
Witkowski ETF, Lamont BB (1991) Leaf specific mass confounds leaf density and thickness. Oecologia 88:486–493
Wright IJ, Cannon K (2001) Relationships between leaf lifespan and structural defences in a low-nutrient, sclerophyll flora. Funct Ecol 15:351–359
Wright IJ, Westoby M (2002) Leaves at low versus high rainfall: coordination of structure, lifespan and physiology. New Phytol 155:403–416
Xu ZZ, Zhou GS (2008) Responses of leaf stomatal density to water status and its relationship with photosynthesis in a grass. J Exp Bot 59:3317–3325
Zhang SB, Slik JW, Zhang JL, Cao KF (2011) Spatial patterns of wood traits in China are controlled by phylogeny and the environment. Global Ecol Biogeogr 20:241–250
Zhang JL, Poorter L, Cao KF (2012a) Productive leaf functional traits of Chinese savanna species. Plant Ecol 213:1449–1460
Zhang SB, Guan ZJ, Sun M, Zhang JJ, Cao KF, Hu H (2012b) Evolutionary association of stomatal traits with leaf vein density in Paphiopedilum, Orchidaceae. PLoS One 7:e40080
Zhang SB, Sun M, Cao KF, Hu H, Zhang JL (2014) Leaf photosynthetic rate of tropical ferns is evolutionarily linked to water transport capacity. PLoS ONE 9:e84682
Zotz G (2013) The systematic distribution of vascular epiphytes-a critical update. Bot J Linn Soc 171:453–481
Zotz G, Bader MY (2009) Epiphytic plants in a changing world-global: change effects on vascular and non-vascular epiphytes. Progress in botany. Springer, Berlin, pp 147–170
Zotz G, Hietz P (2001) The physiological ecology of vascular epiphytes: current knowledge, open questions. J Exp Bot 52:2067–2078
Acknowledgments
We would like to thank Nissa Kreidler and Julie Anberree Lebreton for helping to troubleshoot complications with translation. We would also like to thank Xiao-Jing Wang, Yan Dai, Hong Ma, and Hui Jiang for helping to collect materials and measure leaf traits. This study was supported by the National Science Foundation of China (No. 31170315 and No. 31370362) and the Natural Science Foundation of Yunnan Province (No. 2013FA044).
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by K.-F. Cao.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Sun, M., Yang, SJ., Zhang, JL. et al. Correlated evolution in traits influencing leaf water balance in Dendrobium (Orchidaceae). Plant Ecol 215, 1255–1267 (2014). https://doi.org/10.1007/s11258-014-0383-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11258-014-0383-2