Abstract
Objective
To investigate factors influencing the volume response of everolimus and sirolimus in tuberous sclerosis complex (TSC) associated-angiomyolipomas (AML).
Methods
A retrospective analysis of 30 cases of TSC-AML treated by mTOR inhibitors (everolimus 18 cases, and sirolimus 12 cases) between April 2014 and November 2017 at our center was carried out. Epidemiological data, therapeutic response and influence factors were reviewed and analyzed. Age, sex, associated with SEGA and/or LAM or not, plasma rapamycin concentration, AML volume at baseline, and mean CT value of AML in the maximum cross-section at baseline were analyzed as potential influencing factors.
Results
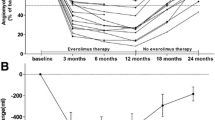
Eighteen patients with 32 lesions in everolimus group and 12 patients with 15 lesions in sirolimus group were included. There was no statistically significant difference of baseline characteristics except for involved side (P = 0.008) between two groups. The mean volume of AML was 1000 ± 1276 cm3 at baseline and 633 ± 1121 cm3 at 6 months after treatment (P < 0.001) in everolimus group, and 1984 ± 2861 cm3 at baseline and 1733 ± 2533 cm3 at 6 months after treatment (P = 0.001) in sirolimus group, respectively. The mean volume reduction of the AML in everolimus and sirolimus groups were 55.56% ± 23.79% and 30.5% ± 22.8% (P = 0.001). Stepwise multiple linear regression analysis revealed that factors influencing the short-term volume response of everolimus and sirolimus for TSC-associated AML were AML volume at baseline (P < 0.001 and 0.038, respectively) and mean CT value at baseline (P < 0.001 and 0.020, respectively). The rates of ≥ 50% volume reduction in high CT value group was much higher than that in low CT value group (90.5% vs. 18.2%, P < 0.001).
Conclusions
Everolimus at 10 mg daily might be more effective than sirolimus at 2 mg daily in treatment of patients with TSC-AML. AML volume and mean CT value at baseline were factors influencing the short-term volume response of everolimus or sirolimus for TSC-AML.

Similar content being viewed by others
Data availability
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Abbreviations
- AML:
-
Angiomyolipomas
- LAM:
-
Lymphangioleiomyomatosis
- SEGA:
-
Subependymal giant cell astrocytoma
- TSC:
-
Tuberous sclerosis complex
References
Sasongko TH, Ismail NF, Zabidi-Hussin Z (2016) Rapamycin and rapalogs for tuberous sclerosis complex. Cochrane Database Syst Rev 7:CD011272
Osborne JP, Fryer A, Webb D (1991) Epidemiology of tuberous sclerosis. Ann NY Acad Sci 615:125–127
Northrup H, Krueger DA, International Tuberous Sclerosis Complex Consensus Group (2013) Tuberous sclerosis complex diagnostic criteria update: recommendations of the 2012 international tuberous sclerosis complex consensus conference. Pediatr Neurol 49(4):243–254
Budde K, Gaedeke J (2012) Tuberous sclerosis complex-associated angiomyolipomas: focus on mTOR inhibition. Am J Kidney Dis 59(2):276–283
Bissler JJ, Kingswood JC, Radzikowska E et al (2013) Everolimus for angiomyolipoma associated with tuberous sclerosis complex or sporadic lymphangioleiomyomatosis (EXIST-2): a multicentre, randomised, double-blind, placebo-controlled trial. Lancet 381(9869):817–824
Bissler JJ, McCormack FX, Young LR et al (2008) Sirolimus for angiomyolipoma in tuberous sclerosis complex or lymphangioleiomyomatosis. N Engl J Med 358(2):140–151
McCormack FX, Inoue Y, Moss J et al (2011) Efficacy and safety of sirolimus in lymphangioleiomyomatosis. N Engl J Med 364(17):1595–1606
Dabora SL, Franz DN, Ashwal S et al. (2011) Multicenter phase 2 trial of sirolimus for tuberous sclerosis: kidney angiomyolipomas and other tumors regress and VEGF-D levels decrease. PLoS ONE 6(9):e23379
Davies DM, de Vries PJ, Johnson SR et al (2011) Sirolimus therapy for angiomyolipoma in tuberous sclerosis and sporadic lymphangioleiomyomatosis: a phase 2 trial. Clin Cancer Res 17(12):4071–4081
Koenig MK, Hebert AA, Roberson J et al (2012) Topical rapamycin therapy to alleviate the cutaneous manifestations of tuberous sclerosis complex: a double-blind, randomized, controlled trial to evaluate the safety and efficacy of topically applied rapamycin. Drugs R&D 12(3):121–126
Cabrera-López C, Martí T (2012) Assessing the effectiveness of rapamycin on angiomyolipoma in tuberous sclerosis: a two years trial. Orphanet J Rare Dis 7:87
van Slegtenhorst M, de Hoogt R, Hermans C et al (1997) Identification of the tuberous sclerosis gene TSC1 on chromosome 9q34. Science 277(5327):805–808
European Chromosome 16 Tuberous Sclerosis Consortium (1993) Identification and characterization of the tuberous sclerosis gene on chromosome 16. Cell 75(7):1305–1315
Dabora SL, Jozwiak S, Franz DN et al (2001) Mutational analysis in a cohort of 224 tuberous sclerosis patients indicates increased severity of TSC2, compared with TSC1, disease in multiple organs. Am J Hum Genet 68(1):64–80
Cai Y, Li H, Zhang Y (2017) Assessment if tuberous sclerosis complex associated with renal lesions by targeted next-generation sequencing in mainland China. Urology 101:170.e1–170.e7
Gao X, Pan D (2001) TSC1 and TSC2 tumor suppressors antagonize insulin signaling in cell growth. Genes Dev 15(11):1383–1392
Kozłowska J, Okoń K (2008) Renal tumors in postmortem material. Pol J Pathol 59(1):21–25
Dixon BP, Hulbert JC, Bissler JJ (2011) Tuberous sclerosis complex renal disease. Nephron Exp Nephrol 118(1):e15–e20
Aydin H, Magi-Galluzzi C, Lane BR et al (2009) Renal angiomyolipoma: clinicopathologic study of 194 cases with emphasis on the epithelioid histology and tuberous sclerosis association. Am J Surg Pathol 33(2):289–297
Curatolo P, Bjørnvold M, Dill PE et al (2016) The role of mTOR inhibitors in the treatment of patients with tuberous sclerosis complex: evidence-based and expert opinions. Drugs 76(5):551–565
Krueger DA, Northrup H, International Tuberous Sclerosis Complex Consensus Group (2013) Tuberous sclerosis complex surveillance and management: recommendations of the 2012 international tuberous sclerosis complex consensus conference. Pediatr Neurol 49(4):255–265
MacKeigan JP, Krueger DA (2015) Differentiating the mTOR inhibitors everolimus and sirolimus in the treatment of tuberous sclerosis complex. Neuro Oncol 17(12):1550–1559
Budde K, Zonnenberg BA, Frost M et al (2016) Pharmacokinetics and pharmacodynamics of everolimus in patients with renal angiomyolipoma and tuberous sclerosis complex or lymphangioleiomyomatosis. Br J Clin Pharmacol 81(5):958–970
Malinowska IA, Lee N, Kumar V et al (2013) Similar trends in serum VEGF-D levels and kidney angiomyolipoma responses with longer duration sirolimus treatment in adults with tuberous sclerosis. PLoS ONE 8(2):e56199
Brugarolas JB, Vazquez F, Reddy A et al (2003) TSC2 regulates VEGF through mTOR-dependent and -independent pathways. Cancer Cell 4(2):147–158
Acknowledgements
We thanked Dr. Xu Wang in Department of Urology, Peking Union Medical College Hospital for participating in manuscript revision work.
Funding
The present study was supported by National Natural Science Foundation of China (No. 81670611).
Author information
Authors and Affiliations
Contributions
YZ and YC conceived and designed the study. HL helped to evaluate the feasibility of the study, and guided the implementation. WW and HG performed the data collection. WW conducted data analysis and produced the manuscript. BS and HS helped to provide the image data.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no competing interests.
Ethics approval
The study was approved by the Ethics Committee of Peking Union Medical College Hospital (Beijing, China).
Informed consent
Consent for publication was obtained from all the authors.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Wang, W., Guo, H., Shi, B. et al. CT characteristics predict the response to everolimus or sirolimus of renal angiomyolipomas in patients with tuberous sclerosis complex. Int Urol Nephrol 51, 671–676 (2019). https://doi.org/10.1007/s11255-019-02093-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11255-019-02093-6