Abstract
Purpose
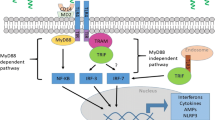
The urothelium is a frontline sensor of the lower urinary tract, sampling the bladder lumen and stimulating an immune response to infectious and noxious agents. Pattern recognition receptors (PRRs) recognize such agents and coordinate the innate response, often by forming inflammasomes that activate caspase-1 and the release of interleukin-1. We have shown the presence of one PRR (NLRP3) in the urothelia and its central role in the inflammatory response to cyclophosphamide. The purpose of this study was to (1) assess the likely range of the PPR response by assessing the repertoire present in the rat bladder and (2) determine the utility of the MYP3 rat urothelia cell line for in vitro studies by assessing its PPR repertoire and functional responsiveness.
Methods
Immunohistochemistry was performed for seven PPRs (NLRP1, NLRP3, NLRP6, NLRP7, NLRP12, NLRC4 and AIM2) on bladder sections and MYP3 cells. For functionality, MYP3 cells were challenged with the quintessential NLRP3 activator ATP and assessed for caspase-1 activation.
Results
All PPRs examined were expressed in the bladder and localized to the urothelial layer with several also in the detrusor (none in the interstitia). MYP3 cells also expressed all PRRs with a variable intracellular location. ATP-stimulated caspase-1 activity in MYP3 cells in a dose-dependent manner was reduced by knockdown of NLRP3 expression.
Conclusion
The results suggest that the bladder possesses the capacity to initiate an innate immune response to a wide array of uropathological agents and the MYP3 cells will provide an excellent investigational tool for this field.










Similar content being viewed by others
References
Birder LA (2006) Urinary bladder urothelium: molecular sensors of chemical/thermal/mechanical stimuli. Vasc Pharmacol 45:221–226. doi:10.1016/j.vph.2005.08.027
Barnett BJ, Stephens DS (1997) Urinary tract infection: an overview. Am J Med Sci 314:245–249
Hooton TM, Stamm WE (1997) Diagnosis and treatment of uncomplicated urinary tract infection. Infect Dis Clin N Am 11:551–581
Foxman B, Barlow R, D’Arcy H, Gillespie B, Sobel JD (2000) Urinary tract infection: self-reported incidence and associated costs. Ann Epidemiol 10:509–515
Foxman B, Brown P (2003) Epidemiology of urinary tract infections: transmission and risk factors, incidence, and costs. Infect Dis Clin N Am 17:227–241
Song J, Abraham SN (2008) Innate and adaptive immune responses in the urinary tract. Eur J Clin Invest 38(Suppl 2):21–28. doi:10.1111/j.1365-2362.2008.02005.x
Davis BK, Wen H, Ting JP (2011) The inflammasome NLRs in immunity, inflammation, and associated diseases. Annu Rev Immunol 29:707–735. doi:10.1146/annurev-immunol-031210-101405
Unterholzner L, Keating SE, Baran M, Horan KA, Jensen SB, Sharma S et al (2010) IFI16 is an innate immune sensor for intracellular DNA. Nat Immunol 11:997–1004. doi:10.1038/ni.1932
Song J, Abraham SN (2008) TLR-mediated immune responses in the urinary tract. Curr Opin Microbiol 11:66–73. doi:10.1016/j.mib.2007.12.001
Kummer JA, Broekhuizen R, Everett H, Agostini L, Kuijk L, Martinon F et al (2007) Inflammasome components NALP 1 and 3 show distinct but separate expression profiles in human tissues suggesting a site-specific role in the inflammatory response. J Histochem Cytochem 55:443–452. doi:10.1369/jhc.6A7101.2006
Hughes FM Jr, Vivar NP, Kennis JG, Pratt-Thomas JD, Lowe DW, Shaner BE et al (2014) Inflammasomes are important mediators of cyclophosphamide-induced bladder inflammation. Am J Physiol Renal Physiol 306:F299–F308. doi:10.1152/ajprenal.00297.2013
Bauernfeind F, Hornung V (2013) Of inflammasomes and pathogens–sensing of microbes by the inflammasome. EMBO Mol Med 5:814–826. doi:10.1002/emmm.201201771
Kawamata H, Kameyama S, Nan L, Kawai K, Oyasu R (1993) Effect of epidermal growth factor and transforming growth factor beta 1 on growth and invasive potentials of newly established rat bladder carcinoma cell lines. Int J Cancer 55:968–973
Cohen SM, Arnold LL, Uzvolgyi E, Cano M, St John M, Yamamoto S et al (2002) Possible role of dimethylarsinous acid in dimethylarsinic acid-induced urothelial toxicity and regeneration in the rat. Chem Res Toxicol 15:1150–1157
Nascimento MG, Suzuki S, Wei M, Tiwari A, Arnold LL, Lu X et al (2008) Cytotoxicity of combinations of arsenicals on rat urinary bladder urothelial cells in vitro. Toxicology 249:69–74. doi:10.1016/j.tox.2008.04.007
Hornung V, Latz E (2010) Critical functions of priming and lysosomal damage for NLRP3 activation. Eur J Immunol 40:620–623. doi:10.1002/eji.200940185
Sutterwala FS, Haasken S, Cassel SL (2014) Mechanism of NLRP3 inflammasome activation. Ann N Y Acad Sci 1319:82–95. doi:10.1111/nyas.12458
Martinon F, Burns K, Tschopp J (2002) The inflammasome: a molecular platform triggering activation of inflammatory caspases and processing of proIL-beta. Mol Cell 10:417–426
Boyden ED, Dietrich WF (2006) Nalp1b controls mouse macrophage susceptibility to anthrax lethal toxin. Nat Genet 38:240–244. doi:10.1038/ng1724
Faustin B, Lartigue L, Bruey JM, Luciano F, Sergienko E, Bailly-Maitre B et al (2007) Reconstituted NALP1 inflammasome reveals two-step mechanism of caspase-1 activation. Mol Cell 25:713–724. doi:10.1016/j.molcel.2007.01.032
Gregory SM, Davis BK, West JA, Taxman DJ, Matsuzawa S, Reed JC et al (2011) Discovery of a viral NLR homolog that inhibits the inflammasome. Science 331:330–334. doi:10.1126/science.1199478
Martinon F, Agostini L, Meylan E, Tschopp J (2004) Identification of bacterial muramyl dipeptide as activator of the NALP3/cryopyrin inflammasome. Curr Biol 14:1929–1934. doi:10.1016/j.cub.2004.10.027
Franchi L, Kanneganti TD, Dubyak GR, Nunez G (2007) Differential requirement of P2X7 receptor and intracellular K + for caspase-1 activation induced by intracellular and extracellular bacteria. J Biol Chem 282:18810–18818. doi:10.1074/jbc.M610762200
Barbe F, Douglas T, Saleh M (2014) Advances in nod-like receptors (NLR) biology. Cytokine Growth Factor Rev. doi:10.1016/j.cytogfr.2014.07.001
Chen GY (2014) Role of Nlrp6 and Nlrp12 in the maintenance of intestinal homeostasis. Eur J Immunol 44:321–327. doi:10.1002/eji.201344135
Elinav E, Strowig T, Kau AL, Henao-Mejia J, Thaiss CA, Booth CJ et al (2011) NLRP6 inflammasome regulates colonic microbial ecology and risk for colitis. Cell 145:745–757. doi:10.1016/j.cell.2011.04.022
Hilt EE, McKinley K, Pearce MM, Rosenfeld AB, Zilliox MJ, Mueller ER et al (2014) Urine is not sterile: use of enhanced urine culture techniques to detect resident bacterial flora in the adult female bladder. J Clin Microbiol 52:871–876. doi:10.1128/JCM.02876-13
Xu W, Yang L, Lee P, Huang WC, Nossa C, Ma Y et al (2014) Mini-review: perspective of the microbiome in the pathogenesis of urothelial carcinoma. Am J Clin Exp Urol 2:57–61
Wlodarska M, Thaiss CA, Nowarski R, Henao-Mejia J, Zhang JP, Brown EM et al (2014) NLRP6 inflammasome orchestrates the colonic host-microbial interface by regulating goblet cell mucus secretion. Cell 156:1045–1059. doi:10.1016/j.cell.2014.01.026
Parsons CL (2007) The role of the urinary epithelium in the pathogenesis of interstitial cystitis/prostatitis/urethritis. Urology 69:9–16. doi:10.1016/j.urology.2006.03.084
Khare S, Dorfleutner A, Bryan NB, Yun C, Radian AD, de Almeida L et al (2012) An NLRP7-containing inflammasome mediates recognition of microbial lipopeptides in human macrophages. Immunity 36:464–476. doi:10.1016/j.immuni.2012.02.001
Anderson GG, Dodson KW, Hooton TM, Hultgren SJ (2004) Intracellular bacterial communities of uropathogenic Escherichia coli in urinary tract pathogenesis. Trends Microbiol 12:424–430. doi:10.1016/j.tim.2004.07.005
Anderson GG, Martin SM, Hultgren SJ (2004) Host subversion by formation of intracellular bacterial communities in the urinary tract. Microbes Infect 6:1094–1101. doi:10.1016/j.micinf.2004.05.023
Hannan TJ, Totsika M, Mansfield KJ, Moore KH, Schembri MA, Hultgren SJ (2012) Host-pathogen checkpoints and population bottlenecks in persistent and intracellular uropathogenic Escherichia coli bladder infection. FEMS Microbiol Rev 36:616–648. doi:10.1111/j.1574-6976.2012.00339.x
Vladimer GI, Weng D, Paquette SW, Vanaja SK, Rathinam VA, Aune MH et al (2012) The NLRP12 inflammasome recognizes Yersinia pestis. Immunity 37:96–107. doi:10.1016/j.immuni.2012.07.006
Williams KL, Lich JD, Duncan JA, Reed W, Rallabhandi P, Moore C et al (2005) The CATERPILLER protein monarch-1 is an antagonist of toll-like receptor-, tumor necrosis factor alpha-, and mycobacterium tuberculosis-induced pro-inflammatory signals. J Biol Chem 280:39914–39924. doi:10.1074/jbc.M502820200
Miao EA, Mao DP, Yudkovsky N, Bonneau R, Lorang CG, Warren SE et al (2010) Innate immune detection of the type III secretion apparatus through the NLRC4 inflammasome. Proc Natl Acad Sci USA 107:3076–3080. doi:10.1073/pnas.0913087107
Buttner D (2012) Protein export according to schedule: architecture, assembly, and regulation of type III secretion systems from plant- and animal-pathogenic bacteria. Microbiol Mol Biol Rev 76:262–310. doi:10.1128/MMBR.05017-11
Kofoed EM, Vance RE (2011) Innate immune recognition of bacterial ligands by NAIPs determines inflammasome specificity. Nature 477:592–595. doi:10.1038/nature10394
Lage SL, Longo C, Branco LM, da Costa TB, Buzzo Cde L, Bortoluci KR (2014) Emerging concepts about NAIP/NLRC4 inflammasomes. Front Immunol 5:309. doi:10.3389/fimmu.2014.00309
Zhao Y, Yang J, Shi J, Gong YN, Lu Q, Xu H et al (2011) The NLRC4 inflammasome receptors for bacterial flagellin and type III secretion apparatus. Nature 477:596–600. doi:10.1038/nature10510
Tenthorey JL, Kofoed EM, Daugherty MD, Malik HS, Vance RE (2014) Molecular basis for specific recognition of bacterial ligands by NAIP/NLRC4 inflammasomes. Mol Cell 54:17–29. doi:10.1016/j.molcel.2014.02.018
Miyazaki J, Ba-Thein W, Kumao T, Akaza H, Hayashi H (2002) Identification of a type III secretion system in uropathogenic Escherichia coli. FEMS Microbiol Lett 212:221–228
Chen TC, Tsai JP, Huang HJ, Teng CC, Chien SJ, Kuo HC et al (2011) Regulation of cyclooxygenase-2 expression in human bladder epithelial cells infected with type I fimbriated uropathogenic E.coli. Cell Microbiol 13:1703–1713. doi:10.1111/j.1462-5822.2011.01650.x
McDermott C, Chess-Williams R, Mills KA, Kang SH, Farr SE, Grant GD et al (2013) Alterations in acetylcholine, PGE2 and IL6 release from urothelial cells following treatment with pyocyanin and lipopolysaccharide. Toxicol In Vitro 27:1693–1698. doi:10.1016/j.tiv.2013.04.015
Wheeler MA, Hausladen DA, Yoon JH, Weiss RM (2002) Prostaglandin E2 production and cyclooxygenase-2 induction in human urinary tract infections and bladder cancer. J Urol 168:1568–1573. doi:10.1097/01.ju.0000030583.31299.80
Fernandes-Alnemri T, Yu JW, Datta P, Wu J, Alnemri ES (2009) AIM2 activates the inflammasome and cell death in response to cytoplasmic DNA. Nature 458:509–513. doi:10.1038/nature07710
Hornung V, Ablasser A, Charrel-Dennis M, Bauernfeind F, Horvath G, Caffrey DR et al (2009) AIM2 recognizes cytosolic dsDNA and forms a caspase-1-activating inflammasome with ASC. Nature 458:514–518. doi:10.1038/nature07725
Jin T, Perry A, Jiang J, Smith P, Curry JA, Unterholzner L et al (2012) Structures of the HIN domain:DNA complexes reveal ligand binding and activation mechanisms of the AIM2 inflammasome and IFI16 receptor. Immunity 36:561–571. doi:10.1016/j.immuni.2012.02.014
Sauer JD, Witte CE, Zemansky J, Hanson B, Lauer P, Portnoy DA (2010) Listeria monocytogenes triggers AIM2-mediated pyroptosis upon infrequent bacteriolysis in the macrophage cytosol. Cell Host Microbe 7:412–419. doi:10.1016/j.chom.2010.04.004
Warren SE, Armstrong A, Hamilton MK, Mao DP, Leaf IA, Miao EA et al (2010) Cutting edge: cytosolic bacterial DNA activates the inflammasome via Aim2. J Immunol 185:818–821. doi:10.4049/jimmunol.1000724
Duewell P, Kono H, Rayner KJ, Sirois CM, Vladimer G, Bauernfeind FG et al (2010) NLRP3 inflammasomes are required for atherogenesis and activated by cholesterol crystals. Nature 464:1357–1361. doi:10.1038/nature08938
Wen C, Yang X, Yan Z, Zhao M, Yue X, Cheng X et al (2013) Nalp3 inflammasome is activated and required for vascular smooth muscle cell calcification. Int J Cardiol 168:2242–2247. doi:10.1016/j.ijcard.2013.01.211
Rawat R, Cohen TV, Ampong B, Francia D, Henriques-Pons A, Hoffman EP et al (2010) Inflammasome up-regulation and activation in dysferlin-deficient skeletal muscle. Am J Pathol 176:2891–2900. doi:10.2353/ajpath.2010.090058
Mezzaroma E, Toldo S, Farkas D, Seropian IM, Van Tassell BW, Salloum FN et al (2011) The inflammasome promotes adverse cardiac remodeling following acute myocardial infarction in the mouse. Proc Natl Acad Sci USA 108:19725–19730. doi:10.1073/pnas.1108586108
Acknowledgments
The authors would like to thank Margret Romano for help with the immunocytochemistry. This work was supported by internal funds from the Medical University of South Carolina.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
All applicable international, national, and/or institutional guidelines for the care and use of animals were followed. All procedures performed in studies involving animals were in accordance with the ethical standards of the Medical University of South Carolina. All experimental procedures were reviewed and approved by the Institutional Animal Care and Use Committee (IACUC) at the Medical University of South Carolina and were performed in accordance with the guidelines set forth in the NIH Guide for the Care and Use of Laboratory Animals, published by the Public Health Service of the USA.
Rights and permissions
About this article
Cite this article
Hughes, F.M., Turner, D.P. & Todd Purves, J. The potential repertoire of the innate immune system in the bladder: expression of pattern recognition receptors in the rat bladder and a rat urothelial cell line (MYP3 cells). Int Urol Nephrol 47, 1953–1964 (2015). https://doi.org/10.1007/s11255-015-1126-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11255-015-1126-6