Abstract
Purpose
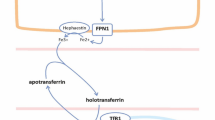
Disturbed iron homeostasis contributes to resistance to recombinant human erythropoietin (rHuEpo) in hemodialysis (HD) patients. Although increased hepcidin, which downregulates the iron exporter ferroportin, had been incriminated, such an association has not been confirmed. Albeit not universally accepted, it has been supported that in case of copper deficiency, decreased activity of multicopper oxidases induces endocytosis and degradation of ferroportin. Ferroportin in monocytes, serum copper, ceruloplasmin and markers of iron status were measured, and associations with rHuEpo resistance index (ERI) were evaluated.
Methods
After a 4-week washout period from iron treatment, 34 HD patients and 20 healthy volunteers enrolled in the study. Ferroportin was assessed by means of Western blotting, copper colorimetrically, whereas ceruloplasmin with enzyme-linked immunosorbent assay. Hemoglobin, serum iron, ferritin and transferrin saturation (TSAT) were also measured.
Results
Ferroportin in monocytes of HD patients was decreased. Serum copper, ceruloplasmin, iron and TSAT were decreased. No correlation between copper or ceruloplasmin and ferroportin was detected. ERI was negatively correlated with ferroportin and all the markers of iron adequacy, but not with copper or ceruloplasmin.
Conclusion
Although copper deficiency and decreased ferroportin are common in HD patients, copper might not play role in ferroportin level in monocytes and in iron metabolism in this population.


Similar content being viewed by others
References
Eleftheriadis T, Liakopoulos V, Antoniadi G, Kartsios C, Stefanidis I (2009) The role of hepcidin in iron homeostasis and anemia in hemodialysis patients. Semin Dial 22(1):70–77. doi:10.1111/j.1525-139X.2008.00532.x
Kanbay M, Perazella MA, Kasapoglu B, Koroglu M, Covic A (2010) Erythropoiesis stimulatory agent- resistant anemia in dialysis patients: review of causes and management. Blood Purif 29(1):1–12. doi:10.1159/000245041
Eleftheriadis T, Kartsios C, Liakopoulos V, Antoniadi G, Ditsa M, Papadopoulos C, Anifandis G, Skirta A, Markala D, Stefanidis I (2006) Does hepcidin affect erythropoiesis in hemodialysis patients? Acta Haematol 116(4):238–244. doi:10.1159/000095873
Ford BA, Eby CS, Scott MG, Coyne DW (2010) Intra-individual variability in serum hepcidin precludes its use as a marker of iron status in hemodialysis patients. Kidney Int 78(8):769–773. doi:10.1038/ki.2010.254
Tessitore N, Girelli D, Campostrini N, Bedogna V, Pietro Solero G, Castagna A, Melilli E, Mantovani W, De Matteis G, Olivieri O, Poli A, Lupo A (2010) Hepcidin is not useful as a biomarker for iron needs in haemodialysis patients on maintenance erythropoiesis-stimulating agents. Nephrol Dial Transpl 25(12):3996–4002. doi:10.1093/ndt/gfq321
van der Weerd NC, Grooteman MP, Bots ML, van den Dorpel MA, den Hoedt CH, Mazairac AH, Nube MJ, Penne EL, Gaillard CA, Wetzels JF, Wiegerinck ET, Swinkels DW, Blankestijn PJ, Ter Wee PM (2012) Hepcidin-25 in chronic hemodialysis patients is related to residual kidney function and not to treatment with erythropoiesis stimulating agents. PLoS One 7(7):e39783. doi:10.1371/journal.pone.0039783
Eleftheriadis T, Pissas G, Remoundou M, Filippidis G, Antoniadi G, Oustampasidou N, Liakopoulos V, Stefanidis I (2014) Ferroportin in monocytes of hemodialysis patients and its associations with hepcidin, inflammation, markers of iron status and resistance to erythropoietin. Int Urol Nephrol 46(1):161–167. doi:10.1007/s11255-013-0497-9
Ward DM, Kaplan J (2012) Ferroportin-mediated iron transport: expression and regulation. Biochim Biophys Acta 1823(9):1426–1433. doi:10.1016/j.bbamcr.2012.03.004
McDermott JA, Huber CT, Osaki S, Frieden E (1968) Role of iron in the oxidase activity of ceruloplasmin. Biochim Biophys Acta 151(3):541–557
Osaki S, Johnson DA, Frieden E (1971) The mobilization of iron from the perfused mammalian liver by a serum copper enzyme, ferroxidase I. J Biol Chem 246(9):3018–3023
Collins JF, Klevay LM (2011) Copper. Adv Nutr 2(6):520–522. doi:10.3945/an.111.001222
Sarkar J, Seshadri V, Tripoulas NA, Ketterer ME, Fox PL (2003) Role of ceruloplasmin in macrophage iron efflux during hypoxia. J Biol Chem 278(45):44018–44024. doi:10.1074/jbc.M304926200
De Domenico I, Ward DM, di Patti MC, Jeong SY, David S, Musci G, Kaplan J (2007) Ferroxidase activity is required for the stability of cell surface ferroportin in cells expressing GPI-ceruloplasmin. EMBO J 26(12):2823–2831. doi:10.1038/sj.emboj.7601735
Vulpe CD, Kuo YM, Murphy TL, Cowley L, Askwith C, Libina N, Gitschier J, Anderson GJ (1999) Hephaestin, a ceruloplasmin homologue implicated in intestinal iron transport, is defective in the sla mouse. Nat Genet 21(2):195–199. doi:10.1038/5979
di Patti MC, Maio N, Rizzo G, De Francesco G, Persichini T, Colasanti M, Polticelli F, Musci G (2009) Dominant mutants of ceruloplasmin impair the copper loading machinery in aceruloplasminemia. J Biol Chem 284(7):4545–4554. doi:10.1074/jbc.M805688200
Prohaska JR, Broderius M (2012) Copper deficiency has minimal impact on ferroportin expression or function. Biometals 25(4):633–642. doi:10.1007/s10534-012-9521-2
Erceg-Hurn DM, Mirosevich VM (2008) Modern robust statistical methods: an easy way to maximize the accuracy and power of your research. Am Psychol 63(7):591–601. doi:10.1037/0003-066X.63.7.591
Andrews NC (1999) Disorders of iron metabolism. N Engl J Med 341(26):1986–1995. doi:10.1056/NEJM199912233412607
Lin TH, Chen JG, Liaw JM, Juang JG (1996) Trace elements and lipid peroxidation in uremic patients on hemodialysis. Biol Trace Elem Res 51(3):277–283. doi:10.1007/BF02784082
Navarro-Alarcon M, Reyes-Perez A, Lopez-Garcia H, Palomares-Bayo M, Olalla-Herrera M, Lopez-Martinez MC (2006) Longitudinal study of serum zinc and copper levels in hemodialysis patients and their relation to biochemical markers. Biol Trace Elem Res 113(3):209–222. doi:10.1385/BTER:113:3:209
Hsieh YY, Shen WS, Lee LY, Wu TL, Ning HC, Sun CF (2006) Long-term changes in trace elements in patients undergoing chronic hemodialysis. Biol Trace Elem Res 109(2):115–121. doi:10.1385/BTER:109:2:115
Ongajooth L, Ongajyooth S, Likidlilid A, Chantachum Y, Shayakul C, Nilwarangkur S (1996) Role of lipid peroxidation, trace elements and anti-oxidant enzymes in chronic renal disease patients. J Med Assoc Thai 79(12):791–800
Emenaker NJ, DiSilvestro RA, Nahman NS Jr, Percival S (1996) Copper-related blood indexes in kidney dialysis patients. Am J Clin Nutr 64(5):757–760
Weissgarten J, Berman S, Bilchinsky R, Modai D, Averbukh Z (2001) Total cell-associated Zn2+ and Cu2+ and proliferative responsiveness of peripheral blood mononuclear cells from patients on chronic hemodialysis. Metabolism 50(3):270–276
Kalantar-Zadeh K, Streja E, Miller JE, Nissenson AR (2009) Intravenous iron versus erythropoiesis-stimulating agents: friends or foes in treating chronic kidney disease anemia? Adv Chronic Kidney Dis 16(2):143–151. doi:10.1053/j.ackd.2008.12.008
Eleftheriadis T, Antoniadi G, Liakopoulos V, Kartsios C, Stefanidis I (2007) Disturbances of acquired immunity in hemodialysis patients. Semin Dial 20(5):440–451. doi:10.1111/j.1525-139X.2007.00283.x
Torti FM, Torti SV (2002) Regulation of ferritin genes and protein. Blood 99(10):3505–3516
Collins JF, Prohaska JR, Knutson MD (2010) Metabolic crossroads of iron and copper. Nutr Rev 68(3):133–147. doi:10.1111/j.1753-4887.2010.00271.x
Higuchi T, Matsukawa Y, Okada K, Oikawa O, Yamazaki T, Ohnishi Y, Fujita T, Fukuda N, Soma M, Matsumoto K (2006) Correction of copper deficiency improves erythropoietin unresponsiveness in hemodialysis patients with anemia. Intern Med 45(5):271–273
Conflict of interest
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Eleftheriadis, T., Pissas, G., Antoniadi, G. et al. Serum copper and ferroportin in monocytes of hemodialysis patients are both decreased but unassociated. Int Urol Nephrol 46, 1825–1831 (2014). https://doi.org/10.1007/s11255-014-0725-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11255-014-0725-y