Abstract
Objective
To evaluate the effect of Pygeum africanum on oxidative stress and functional changes of the bladder after diabetes induction.
Materials and methods
Thirty-two adult Wistar male rats were treated daily for 8 weeks and grouped as follows: Control group (n = 6), Streptozotocin-induced diabetic group (n = 10), diabetes plus P. africanum group (n = 10), and control plus P. africanum group (n = 6). After diabetes induction for 4 weeks, the diabetes plus P. africanum and control plus P. africanum groups were fed with P. africanum (100 mg/kg, orally) in peanut oil for another 4 weeks. The catalase, superoxide dismutase activity, and malondialdehyde levels were measured as a marker of lipid peroxidation. The levels of inducible nitric oxide synthase were also evaluated. Urodynamic studies were performed to evaluate the functional changes of diabetic bladders after P. africanum treatment.
Results
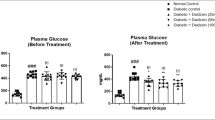
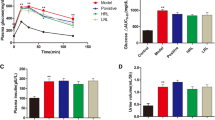
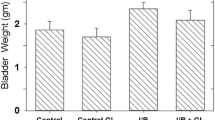
The catalase and superoxide dismutase activities significantly increased (P < 0.05) and maleic dialdehyde levels significantly decreased from diabetic plus P. africanum group compared with diabetic group (P < 0.05). Immunohistochemical studies showed a significantly decreased number of inducible nitric oxide synthase-positive cells in diabetic plus P. africanum group compared with diabetic group (P < 0.05). In diabetic plus P. africanum group, maximal bladder volume significantly decreased, while bladder pressure and maximal bladder pressure significantly increased compared with diabetic group (P < 0.05).
Conclusions
Early treatment with P. africanum could effectively suppress the oxidative stress status in diabetic bladder and may slow down the process of diabetic cystopathy.


Similar content being viewed by others
References
Brown JS, Wessells H, Chancellor MB, Howards SS, Stamm WE, Stapleton AE, Steers WD, Van Den Eeden SK, McVary KT (2005) Urologic complications of diabetes. Diabetes Care 28(1):177–185
Olapade-Olaopa EO, Morley RN, Carter CJ, Walmsley BH (1997) Diabetic cystopathy presenting as primary acute urinary retention in a previously undiagnosed young male diabetic patient. J Diabetes Complicat 11(6):350–351
Jiang YJ, Gong DX, Liu HB, Yang CM, Sun ZX, Kong CZ (2008) Ability of alpha-lipoic acid to reverse the diabetic cystopathy in a rat model. Acta Pharmacol Sin 29(6):713–719
Yoshimura N, Chancellor MB, Andersson KE, Christ GJ (2005) Recent advances in understanding the biology of diabetes-associated bladder complications and novel therapy. BJU Int 95(6):733–738
Bradley WE (1980) Diagnosis of urinary bladder dysfunction in diabetes mellitus. Ann Intern Med 92(2 Pt 2):323–326
Ueda T, Yoshimura N, Yoshida O (1997) Diabetic cystopathy: relationship to autonomic neuropathy detected by sympathetic skin response. J Urol 157(2):580–584
Yongzhi L, Benkang S, Jianping Z, Lingxia R, Wei B, Yaofeng Z, Keqin Z, Laudon V (2008) Expression of transforming growth factor beta1 gene, basic fibroblast growth factor gene and hydroxyproline in diabetes-induced bladder dysfunction in a rat model. Neurourol Urodyn 27(3):254–259
Beshay E, Carrier S (2004) Oxidative stress plays a role in diabetes-induced bladder dysfunction in a rat model. Urology 64(5):1062–1067
Slatter DA, Bolton CH, Bailey AJ (2000) The importance of lipid-derived malondialdehyde in diabetes mellitus. Diabetologia 43(5):550–557
Kristal BS, Koopmans SJ, Jackson CT, Ikeno Y, Park BJ, Yu BP (1997) Oxidant-mediated repression of mitochondrial transcription in diabetic rats. Free Radic Biol Med 22(5):813–822
Bonnefont-Rousselot D, Bastard JP, Jaudon MC, Delattre J (2000) Consequences of the diabetic status on the oxidant/antioxidant balance. Diabetes Metab 26(3):163–176
Romanenko A, Morimura K, Wanibuchi H, Salim EI, Kinoshita A, Kaneko M, Vozianov A, Fukushima S (2000) Increased oxidative stress with gene alteration in urinary bladder urothelium after the chernobyl accident. Int J Cancer 86(6):790–798
Barlet A, Albrecht J, Aubert A, Fischer M, Grof F, Grothuesmann HG, Masson JC, Mazeman E, Mermon R, Reichelt H et al (1990) Efficacy of Pygeum africanum extract in the medical therapy of urination disorders due to benign prostatic hyperplasia: evaluation of objective and subjective parameters. A placebo-controlled double-blind multicenter study. Wien Klin Wochenschr 102(22):667–673
Donkervoort T, Sterling A, van Ness J, Donker PJ (1977) A clinical and urodynamic study of tadenan in the treatment of benign prostatic hypertrophy. Eur Urol 3(4):218–225
Breza J, Dzurny O, Borowka A, Hanus T, Petrik R, Blane G, Chadha-Boreham H (1998) Efficacy and acceptability of tadenan (Pygeum africanum extract) in the treatment of benign prostatic hyperplasia (BPH): a multicentre trial in central Europe. Curr Med Res Opin 14(3):127–139
Levin RM, Das AK, Haugaard N, Novitsky Y, Horan P, Leggett RE, Riffaud JP, Longhurst PA (1997) Beneficial effects of tadenan therapy after 2 weeks of partial obstruction in the rabbit. Neurourol Urodyn 16(6):583–599
Levin RM, Das AK (2000) A scientific basis for the therapeutic effects of Pygeum africanum and Serenoa repens. Urol Res 28(3):201–209
Levin RM, Riffaud JP, Bellamy F, Rohrmann D, Habib M, Krasnopolsky L, Zhao Y, Wein AJ (1996) Protective effect of tadenan on bladder function secondary to partial outlet obstruction. J Urol 155(4):1466–1470
Mossman BT, Wilson GL, Craighead JE (1985) Chlorozocin. A diabetogenic analogue of streptozocin with dissimilar mechanisms of action on pancreatic beta cells. Diabetes 34(6):602–610
Bolaffi JL, Nowlain RE, Cruz L, Grodsky GM (1986) Progressive damage of cultured pancreatic islets after single early exposure to streptozocin. Diabetes 35(9):1027–1033
Kolb-Bachofen V, Epstein S, Kiesel U, Kolb H (1988) Low-dose streptozocin-induced diabetes in mice. Electron microscopy reveals single-cell insulitis before diabetes onset. Diabetes 37(1):21–27
Persson K, Andersson KE (1992) Nitric oxide and relaxation of pig lower urinary tract. Br J Pharmacol 106(2):416–422
Acknowledgments
This study was supported by Nature and Science Foundation of Shandong Province, China (no. Y2005C09).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Wang, D., Li, Y., Hou, G. et al. Pygeum africanum: effect on oxidative stress in early diabetes-induced bladder. Int Urol Nephrol 42, 401–408 (2010). https://doi.org/10.1007/s11255-009-9610-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11255-009-9610-5