Abstract
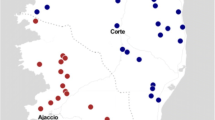
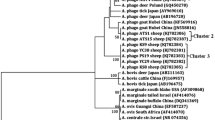
The aim of this cross-sectional study was to determine the molecular prevalence and associated risk factors in sheep populations of Iran. To this end, between March 2017 and February 2018 jugular vein blood samples were collected from 1842 apparently healthy sheep from 327 herds in nine provinces in four ecological zones of Iran. A specific nested-PCR targeting the msp4 gene of A. ovis was employed. Fourteen variables were subjected to logistic regression analyses (univariate and multivariate) to specify the potential risk factors for infection. Statistically significant variables in univariate analyses (P ≤ 0.20) were assessed by multivariable logistic regression to control the confounding factors. Anaplasma ovis DNA was detected in 51.1% of herds (167/327) and 28.3% of animals (521/1842). Among geographical zones, herd and animal prevalence was highest in the Persian-Gulf zone (P < 0.001), and among provinces, Lorestan (in west) and Khuzestan (in south-west) had the highest prevalence (P < 0.001). Analysis of factors associated with A. ovis infection revealed that distance from other farms (OR = 2.52, P < 0.001), presence of other animal species in the farm (OR = 2.03, P = 0.046), season (OR = 1.40, P = 0.005), breed (OR = 3.762, P < 0.001), and age of sheep (OR = 1.20, P = 0.049) are potential risks in Iran. The spatial scan statistic in SaTScan recognized two high risks clusters for A. ovis infection in central (Semnan province) and the Persian-Gulf (Khuzestan province) zones amongst the study areas (P < 0.001). Sequence and phylogenetic analysis of the msp4 gene confirmed the detection of A. ovis. This research is the largest study focusing on ovine anaplasmosis in Iran and shows that infected sheep are present in all geographic zones, bioclimatic areas, and provinces.



Similar content being viewed by others
Data availability
The data that support the findings of this study are however available from the corresponding author upon reasonable request. The provided GenBank accession number for our nucleotide sequence is MZ383975.
Code availability
Not applicable.
References
Aktas, M. and Özübek, S., 2018. Anaplasma ovis genetic diversity detected by major surface protein 1a and its prevalence in small ruminants, Veterinary Mcrobiology, 217, 13-17.
Alanazi, A.D., Nguyen, V.L., Alyousif, M.S., Manoj, R.R., Alouffi, A.S., Donato, R., Sazmand, A., Mendoza-Roldan, J.A., Dantas-Torres, F. and Otranto, D., 2020. Ticks and associated pathogens in camels (Camelus dromedarius) from Riyadh Province, Saudi Arabia, Parasites & Vectors, 13, 1-9.
Altay, K., Dumanli, N., Aktas, M. and Ozubek, S., 2014. Survey of Anaplasma infections in small ruminants from East part of Turkey, Kafkas Universitesi Veteriner Fakultesi Dergisi, 20, 1-4.
Belkahia, H., Ben Said, M., El Hamdi, S., Yahiaoui, M., Gharbi, M., Daaloul-Jedidi, M., Mhadhbi, M., Jedidi, M., Darghouth, M.A. and Klabi, I., 2014. First molecular identification and genetic characterization of Anaplasma ovis in sheep from Tunisia, Small Ruminant Research, 121, 404-410.
Belkahia, H., Ben Said, M., El Mabrouk, N., Saidani, M., Cherni, C., Ben Hassen, M., Bouattour, A. and Messadi, L., 2017. Seasonal dynamics, spatial distribution and genetic analysis of Anaplasma species infecting small ruminants from Northern Tunisia, Infection, Genetics and Evolution, 54, 66-73.
Belkahia, H., Ben Said, M., Ghribi, R., Selmi, R., Asker, A.B., Yahiaoui, M., Bousrih, M., Daaloul-Jedidi, M. and Messadi, L., 2019. Molecular detection, genotyping and phylogeny of Anaplasma spp. in Rhipicephalus ticks from Tunisia, Acta Tropica, 191, 38-49.
Ben Said, M., Belkahia, H., Alberti, A., Zobba, R., Bousrih, M., Yahiaoui, M., Daaloul-Jedidi, M., Mamlouk, A., Gharbi, M. and Messadi, L., 2015. Molecular survey of Anaplasma species in small ruminants reveals the presence of novel strains closely related to A. phagocytophilum in Tunisia, Vector-Borne and Zoonotic Diseases, 15, 580-590.
Ben Said, M., Belkahia, H. and Messadi, L., 2018. Anaplasma spp. in North Africa: a review on molecular epidemiology, associated risk factors and genetic characteristics, Ticks and Tick-borne Diseases, 9, 543-555.
Cabezas-Cruz, A., Gallois, M., Fontugne, M., Allain, E., Denoual, M., Moutailler, S., Devillers, E., Zientara, S., Memmi, M., Chauvin, A., Agoulon, A., Vayssier-Taussat, M. and Chartier, C., 2019. Epidemiology and genetic diversity of Anaplasma ovis in goats in Corsica, France, Parasites & Vectors, 12, 1-11.
Changizi, E., 2015. Prevalence, intensity and associated risk factors for ovine tick infestation in two districts of Semnan area, Iranian Journal of Veterinary Medicine, 8, 287-292.
Chochlakis, D., Ioannou, I., Tselentis, Y. and Psaroulaki, A., 2010. Human anaplasmosis and Anaplasma ovis variant, Emerging infectious diseases, 16, 1031-1032.
Ciani, E., Alloggio, I., Petazzi, F. and Pieragostini, E., 2013. Looking for prognosticators in ovine anaplasmosis: discriminant analysis of clinical and haematological parameters in lambs belonging to differently susceptible breeds experimentally infected with Anaplasma ovis, Acta Veterinaria Scandinavica, 55, 1-5.
Dantas-Torres, F. and Otranto, D., 2017. Anaplasmosis. In: C.B. Marcondes (ed), Arthropod Borne Diseases, 2017, (Springer, Cham, Switzerland), 215-222.
de la Fuente, J., Atkinson, M.W., Naranjo, V., de Mera, I.G.F., Mangold, A.J., Keating, K.A. and Kocan, K.M., 2007. Sequence analysis of the msp4 gene of Anaplasma ovis strains, Veterinary Mcrobiology, 119, 375-381.
de la Fuente, J., Estrada-Pena, A., Venzal, J.M., Kocan, K.M. and Sonenshine, D.E., 2008. Overview: ticks as vectors of pathogens that cause disease in humans and animals, Frontiers in Bioscience, 13, 6938-6946.
Delpy, L.-P., 1939. Agents pathogènes observés en Iran dans le sang des animaux domestiques, Archives of Razi Institute, 1, 72-77.
Eydivandi, S., Sahana, G., Momen, M., Moradi, M. and Schönherz, A., 2020. Genetic diversity in Iranian indigenous sheep vis-à-vis selected exogenous sheep breeds and wild mouflon, Animal Genetics, 51, 772-787.
Farahani, A., Razi-Jalali, M.H., Hamidinejat, H. and Tabandeh, M.R., 2020. Determination of fauna of hard ticks on sheep in Khuzestan province, Iran, Journal of Isfahan Medical School, 37, 1376-1380.
Friedhoff, K., 1997. Tick-borne diseases of sheep and goats caused by Babesia, Theileria or Anaplasma spp, Parassitologia, 39, 99-109.
Ghaffar, A., Ijaz, M., Ali, A., Farooqi, S.H., Rehman, A., Ali, M.M., Zafar, M.Z. and Naeem, M.A., 2020. First report on molecular characterization of anaplasmosis in small ruminants in Pakistan, Journal of Parasitology, 106, 360-368.
Gharbi, M., Omri, H., Jedidi, M., Zorii, S. and Darghouth, M., 2015. Epidemiological study of sheep anaplasmosis (Anaplasma ovis infection) in Kairouan, central Tunisia, Journal of Advances in Parasitology, 2, 30-34.
Hall, T.A., 1999. BioEdit: a user-friendly biological sequence alignment editor and analysis program for Windows 95/98/NT, Nucleic Acids Symposium Series, 41, 95-98.
Hosseini-Vasoukolaei, N., Oshaghi, M.A., Shayan, P., Vatandoost, H., Babamahmoudi, F., Yaghoobi-Ershadi, M.R., Telmadarraiy, Z. and Mohtarami, F., 2014. Anaplasma infection in ticks, livestock and human in Ghaemshahr, Mazandaran Province, Iran, Journal of Arthropod-Borne Diseases, 8, 204-211.
Jalali, S., Khaki, Z., Kazemi, B., Bandehpour, M., Rahbari, S., Razi Jalali, M. and Yasini, S., 2013. Molecular detection and identification of Anaplasma species in sheep from Ahvaz, Iran, Iranian Journal of Veterinary Research, 14, 50-56.
Jiménez, C., Benito, A., Arnal, J., Ortín, A., Gómez, M., López, A., Villanueva-Saz, S. and Lacasta, D., 2019. Anaplasma ovis in sheep: Experimental infection, vertical transmission and colostral immunity, Small Ruminant Research, 178, 7-14.
Kocan, K.M., de la Fuente, J., Blouin, E.F., Coetzee, J.F. and Ewing, S.A., 2010. The natural history of Anaplasma marginale, Veterinary Parasitology, 167, 95-107.
Kulldorff, M., 2010. SaTScan user guide for version 9.0. Available at: http://www.scribd.com/doc/88420107/Current-Version-SaTScan-v9-1-1-Released-March-92011#scribd.
Mlimbe, M., Hyera, E., Ochanga, P., Nguluma, A., Marwa, L., Rugaimukamu, A., Godfrey, J., Ngabo, M. and Shirima, E., 2021. Study on the causes and pattern of sheep mortality under farm conditions in Northern Tanzania, Livestock Research for Rural Development, 32, 2020.
Mohammadian, B., Noaman, V. and Emami, S.J., 2021. Molecular survey on prevalence and risk factors of Anaplasma spp. infection in cattle and sheep in West of Iran, Tropical Animal Health and Production, 53, 1-7.
Nabian, S., Rahbari, S., Shayan, P. and Hadadzadeh, H., 2007. Current status of tick fauna in north of Iran, Iranian Journal of Parasitology, 2, 12-17.
Niaz, S., Zia Ur Rahman, I.A., Cossío-Bayúgar, R., Amaro-Estrada, I., Alanazi, A.D., Khattak, I., Zeb, J., Nasreen, N. and Khan, A., 2021. Molecular prevalence, characterization and associated risk factors of Anaplasma spp. and Theileria spp. in small ruminants in Northern Pakistan, Parasite, 28, 1-13.
Noaman, V., 2012. Identification of hard ticks collected from sheep naturally infected with Anaplasma ovis in Isfahan province, central Iran, Comparative Clinical Pathology, 21, 367-369.
Noaman, V., 2018. Molecular detection of novel genetic variants associated to Anaplasma ovis among dromedary camels in Iran, Archives of Razi Institute, 73, 11-18.
Noaman, V., 2020. Epidemiological study on Anaplasma phagocytophilum in cattle: molecular prevalence and risk factors assessment in different ecological zones in Iran, Preventive Veterinary Medicine, 183, 105118.
Noaman, V., Abdigoudarzi, M. and Nabinejad, A., 2017. Abundance, diversity, and seasonal dynamics of hard ticks infesting cattle in Isfahan Province, Iran, Archives of Razi Institute, 72, 15-21.
Noaman, V. and Bastani, D., 2016. Molecular study on infection rates of Anaplasma ovis and Anaplasma marginale in sheep and cattle in West-Azerbaijan province, Iran, Veterinary Research Forum, 7, 163-167.
Noaman, V. and Moradi, M., 2019. Molecular epidemiology and risk factors assessment of Anaplasma spp. on dairy cattle in southwest of Iran, Acta Veterinaria Eurasia, 45, 30-36.
Noaman, V., Shayan, P. and Shahmoradi, A., 2009. Detection of Anaplasma ovis based on 16S rRNA gene by PCR-RFLP in sheep from central part of Iran, Journal of Veterinary Laboratory Research, 1, 27-37.
Rahbari, S., 1995. Studies on some ecological aspects of Tick West Azarbidjan, Iran, Journal of Applied Animal Research, 7, 189-194.
Reinbold, J.B., Coetzee, J.F., Hollis, L.C., Nickell, J.S., Riegel, C.M., Christopher, J.A. and Ganta, R.R., 2010. Comparison of iatrogenic transmission of Anaplasma marginale in Holstein steers via needle and needle-free injection techniques, American Journal of Veterinary Research, 71, 1178-1188.
Renneker, S., Abdo, J., Salih, D., Karagenç, T., Bilgiç, H., Torina, A., Oliva, A., Campos, J., Kullmann, B. and Ahmed, J., 2013. Can Anaplasma ovis in small ruminants be neglected any longer?, Transboundary and Emerging Diseases, 60, 105-112.
Sazmand, A., Harl, J., Eigner, B., Hodžić, A., Beck, R., Hekmatimoghaddam, S., Mirzaei, M., Fuehrer, H.-P. and Joachim, A., 2019. Vector-borne bacteria in blood of camels in Iran: new data and literature review, Comparative Immunology, Microbiology and Infectious Diseases, 65, 48-53.
Selmi, R., Said, M.B., Dhibi, M., Yahia, H.B., Abdelaali, H. and Messadi, L., 2020. Genetic diversity of groEL and msp4 sequences of Anaplasma ovis infecting camels from Tunisia, Parasitology International, 74, 101980.
Soosaraei, M., Haghi, M.M., Etemadifar, F., Fakhar, M., Teshnizi, S.H., Asfaram, S. and Esboei, B.R., 2020. Status of Anaplasma spp. infection in domestic ruminants from Iran: A systematic review with meta-analysis. Parasite Epidemiology and Control, 11, e00173.
Tamura, K. and Nei, M., 1993. Estimation of the number of nucleotide substitutions in the control region of mitochondrial DNA in humans and chimpanzees, Molecular Biology and Evolution, 10, 512-526.
Tamura, K., Peterson, D., Peterson, N., Stecher, G., Nei, M. and Kumar, S., 2011. MEGA5: molecular evolutionary genetics analysis using maximum likelihood, evolutionary distance, and maximum parsimony methods, Molecular Biology and Evolution, 28, 2731-2739.
Thrusfield, M. and Christley, R., 2018. Veterinary Epidemiology, (John Wiley & Sons, NJ, USA.)
Torina, A., Agnone, A., Blanda, V., Alongi, A., D’Agostino, R., Caracappa, S., Marino, A.M., Di Marco, V. and de la Fuente, J., 2012. Development and validation of two PCR tests for the detection of and differentiation between Anaplasma ovis and Anaplasma marginale, Ticks and Tick-borne Diseases, 3, 283-287.
Yasini, S., Khaki, Z., Rahbari, S., Kazemi, B., Amoli, J.S., Gharabaghi, A. and Jalali, S., 2012. Hematologic and clinical aspects of experimental ovine anaplasmosis caused by Anaplasma ovis in Iran, Iranian Journal of Parasitology, 7, 91-98.
Yousefi, A., Rahbari, S., Shayan, P., Sadeghi-dehkordi, Z. and Bahonar, A., 2017. Molecular detection of Anaplasma marginale and Anaplasma ovis in sheep and goat in west highland pasture of Iran, Asian Pacific Journal of Tropical Biomedicine, 7, 455-459.
Zhou, M., Cao, S., Sevinc, F., Sevinc, M., Ceylan, O., Ekici, S., Jirapattharasate, C., Moumouni, P.F.A., Liu, M. and Wang, G., 2017. Molecular detection and genetic characterization of Babesia, Theileria and Anaplasma amongst apparently healthy sheep and goats in the central region of Turkey, Ticks and Tick-borne Diseases, 8, 246-252.
Acknowledgements
The authors would like to thank farmers, veterinarians, and laboratory colleagues who assisted us with sampling.
Author information
Authors and Affiliations
Contributions
VN conceived the study and performed fieldwork. VN performed laboratory work. VN and AS performed and analyzed data. VN and AS wrote the first draft of the manuscript. AS reviewed the manuscript. VN and AS read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics approval
Blood samples were taken according to the methods and guidelines of animal ethics approved by the Animal Ethics Committee of AREEO. All sampling was done with the permission of the livestock owner.
Consent to participate
Before each interview, verbal informed consent was acquired from all livestock owners.
Consent for publication
All authors read and consent to the publication of the manuscript.
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Noaman, V., Sazmand, A. Anaplasma ovis infection in sheep from Iran: molecular prevalence, associated risk factors, and spatial clustering. Trop Anim Health Prod 54, 6 (2022). https://doi.org/10.1007/s11250-021-03007-4
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11250-021-03007-4