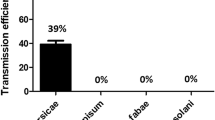
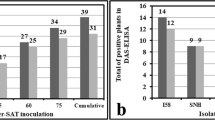
Abstract
The molecular variability of Plum pox virus (PPV) populations was compared in transgenic European plums (Prunus domestica L.) carrying the coat protein (CP) gene of PPV and non-transgenic plums in an experimental orchard in Valencia, Spain. A major objective of this study was to detect recombination between PPV CP transgene transcripts and infecting PPV RNA. Additionally, we assessed the number and species of PPV aphid vectors that visited transgenic and non-transgenic plum trees. Test trees consisted of five different P. domestica transgenic lines, i.e. the PPV-resistant C5 ‘HoneySweet’ line and the PPV-susceptible C4, C6, PT6 and PT23 lines, and non-transgenic P. domestica and P. salicina Lind trees. No significant difference in the genetic diversity of PPV populations infecting transgenic and conventional plums was detected, in particular no recombinant between transgene transcripts and incoming viral RNA was found at detectable levels. Also, no significant difference was detected in aphid populations, including viruliferous individuals, that visited transgenic and conventional plums. Our data indicate that PPV-CP transgenic European plums exposed to natural PPV infection over an 8 year period caused limited, if any, risk beyond the cultivation of conventional plums under Mediterranean conditions in terms of the emergence of recombinant PPV and diversity of PPV and aphid populations.



Similar content being viewed by others
References
Avinent L, Hermoso de Mendoza A, Llácer G (1989) Especies dominantes y curvas de vuelo de pulgones (Homoptera, Aphidinea) en campos de frutales de hueso españoles. Invest Agraria Prod Prot Veget 4:283-298
Avinent L, Hermoso de Mendoza A, Llácer G (1991) Comparison of traps for captures of alate aphids (Homoptera, Aphidinea). Agronomie 11:613–618
Avinent L, Hermoso de Mendoza A, Llácer G (1993) Comparison of sampling methods to evaluate aphid populations (Homoptera, Aphidinea) alighting on apricot trees. Agronomie 13:609–613
Badenes ML, Moustafa TA, Martínez-Calvo J, Llácer G (2006) Resistance to sharka trait in a family from selfpollination of ‘Lito’ apricot cultivar. Acta Hortic 701:381–384
Boscia D, Zeramdini H, Cambra M, Potere O, Gorris MT, Myrta A (1997) Production and characterization of a monoclonal antibody specific to the M serotype of Plum pox potyvirus. Eur J Plant Pathol 103:447–480
Cambra M, Asensio M, Gorris MT, Pérez E, Camarasa E, García JA, López-Moya JJ, López-Abella D, Vela C, Sanz A (1994) Detection of Plum pox potyvirus using monoclonal antibodies to structural and non structural proteins. Bull OEPP/EPPO Bull 24:569–577
Cambra M, Gorris MT, Marroquín C, Román MP, Olmos A, Martínez MC, Hermoso de Mendoza A, López A, Navarro L (2000) Incidence and epidemiology of Citrus tristeza virus in the Valencia Community of Spain. Virus Res 71:85–95
Cambra M, Gorris MT, Capote N, Asensio M, Martínez MC, Bertolini E, Collado C, Hermoso de Mendoza A, Matáix E, López A (2004) Epidemiology of Plum pox virus in Japanese plums in Spain. Acta Hortic 657:195–200
Cambra M, Capote N, Cambra MA, Llácer G, Botella P, López-Quílez A (2006) Epidemiology of sharka disease in Spain. Bull OEPP/EPPO Bull 36:271–275
Cambra M, Capote N, Myrta A, Llácer G (2006) Plum pox virus and the estimated costs associated with sharka disease. Bull OEPP/EPPO Bull 36:202–204
Candresse T, Cambra M (2006) Causal agent of sharka disease: historical perspective and current status of Plum pox virus strains. Bull OEPP/EPPO Bull 36:239–246
Candresse T, Macquaire G, Lanneau M, Bousalem M, Wetzel T, Quiot-Dounie L, Quiot JB, Dunez J (1994) Detection of Plum pox potyvirus and analysis of its molecular variability using immunocapture-PCR. Bull OEPP/EPPO Bull 24:585–594
Capote N, Cambra M (2005) Variability of Plum pox virus populations in PPV-resistant transgenic and non-transgenic plums. Phytopathol Pol 36:107–113
Capote N, Gorris MT, Martínez MC, Asensio M, Olmos A, Cambra M (2006) Interference between D and M types of Plum pox virus in Japanese plums assessed by specific monoclonal antibodies and quantitative real-time RT-PCR. Phytopathology 96:320–325
Cervera MT, Riechmann JL, Martin MT, García JA (1993) 3’-terminal sequence of the Plum pox virus PS and o6 isolates: evidence for RNA recombination within the Potyvirus group. J Gen Virol 74:329–334
Excoffier L, Smouse P, Quattro J (1992) Analysis of molecular variance inferred from metric distances among DNA haplotypes: Application to human DNA restriction data. Genetics 131:479–491
Fuchs M, Klas FE, McFerson JR, Gonsalves D (1998) Transgenic melon and squash expressing coat protein genes of Aphid-borne viruses do not assist the spread of an Aphid non-transmissible strain of Cucumber mosaic virus in the field. Transgenic Res 7:449–462
Glasa M, Kúdela O, Marie-Jeanne V, Quiot JB (2001) Evidence of naturally occurring recombinant Plum pox virus isolate from Slovakia. Plant Dis 85:920
Glasa M, Marie-Jeanne V, Labonne G, Šubr Z, Kúdela O, Quiot JB (2002) Natural population of recombinant Plum pox virus is stable and competitive under field conditions. Eur J Plant Pathol 108:843–853
Glasa M, Palkovics L, Komínek P, Labonne G, Pittnerová S, Kúdela O, Candresse T, Šubr Z (2004) Geographically and temporally distant natural recombinant isolates of Plum pox virus (PPV) are genetically very similar and form a unique PPV subgroup. J Gen Virol 85:2671–2681
Glasa M, Paunovic S, Jevremovic D, Myrta A, Pittnerová S, Candresse T (2005) Analysis of recombinant Plum pox virus (PPV) isolates from Serbia confirms genetic homogeneity and supports a regional origin for the PPV-Rec subgroup. Arch Virol 150:2051–2060
Greene AE, Allison FR (1994) Recombination between viral RNA and transgenic plant transcripts. Science 263:1423–1425
Greene AE, Allison FR (1996) Deletions in the 3’ untranslated region of cowpea chlorotic mottle virus transgene reduce recovery of recombinant viruses in transgenic plants. Virology 225:231–234
Hily JM, Scorza R, Malinowski T, Zawadzka B, Ravelonandro M (2004) Stability of gene silencing-based resistance to Plum pox virus in transgenic plum (Prunus domestica L.) under field conditions. Transgenic Res 13:427–436
Hily JM, Scorza R, Webb K, Ravelonandro M (2005) Accumulation of the long class of siRNA is associated with resistance to Plum pox virus in a transgenic woody perennial plum tree. Mol Plant Microbe Interact 18:794–799
James D, Glasa M (2006) Causal agent of sharka disease: new and emerging events associated with Plum pox virus characterization. Bull OEPP/EPPO Bull 36:247–250
James D, Varga A (2005) Nucleotide sequence analysis of Plum pox virus isolate W3174: Evidence of a new strain. Virus Res 110:143–150
Kegler H, Fuchs E, Gruntzig M, Schwarz S (1998) Some results of 50 years of research on the resistance to Plum pox virus. Acta Virol 42:200–215
Kunze L, Krczal H (1971) Transmisssion of Sharka virus by aphids. Ann Phytopathol HS:255–260
Labonne G, Dallot S (2006) Epidemiology of sharka disease in France. Bull OEPP/EPPO Bull 36:267–270
López-Moya JJ, Fernández-Fernández MR, Cambra M, García JA (2000) Biotechnological aspects of Plum pox virus. J Biotechnol 76:121–136
Lole KS, Bollinger RC, Paranjape RS, Gadkari D, Kulkarni SS, Novak NG, Ingersoll R, Sheppard HW, Ray SC (1999) Full-length human immunodeficiency virus type 1 genomes from subtype C-infected seroconverters in India, with evidence of intersubtype recombination. J Virol 73:152–60
Malinowski T, Zawadzka B, Ravelonandro M, Scorza R (1998) Preliminary report on the apparent breaking of resistance of a transgenic plum by chip bud inoculation of Plum pox virus PPV-S. Acta Virol 42:241–243
Malinowski T, Cambra M, Capote N, Zawadzka B, Gorris MT, Scorza R, Ravelonandro M (2006) Field trials of plum clones transformed with the Plum pox virus coat protein (PPV-CP) gene. Plant Dis 90:1012–1018
McCullagh P, Nelder JA (1989) Generalized Linear Models, 2nd edn. Cahpman & Hall, London
Myrta A, Potere O, Boscia D, Candresse T, Cambra M, Savino V (1998) Production of a monoclonal antibody specific to the El Amar strain of Plum pox virus. Acta Virol 42:248–250
Myrta A, Potere O, Crescenzi A, Nuzzaci M, Boscia D (2000) Production of two monoclonal antibodies specific to cherry strain of Plum pox virus (PPV-C). J Plant Pathol 82:95–103
Neumüller M, Hartmann W, Stösser R (2005) The hypersensitivity of European plum against PPV as a promising mechanism of resistance. Phytopathol Pol 36:77–83
Ng JCK, Perry KL (2004) Transmission of plant viruses by aphid vectors. Mol Plant Pathol 5:505–511
Olmos A, Dasí MA, Candresse T, Cambra M (1996) Print-capture PCR: A simple and highly sensitive method for the detection of Plum pox virus (PPV) in plant tissues. Nucleic Acids Res 24:2192–2193
Olmos A, Bertolini E, Gil M, Cambra M (2005) Real-time assay for quantitative detection of non-persistently transmitted Plum pox virus RNA targets in single aphids. J Virol Methods 128:151–155
Ravelonandro M, Scorza R, Bachelier JC, Labonne G, Levy L, Damsteegt V, Callahan AM, Dunez J (1997) Resistance of transgenic Prunus domestica to Plum pox virus infection. Plant Dis 81:1231–1235
Ravelonandro M, Varveri C, Delbos R, Dunez J (1998) Nucleotide sequence of the capsid protein gene of Plum pox potyvirus. J Gen Virol 69:1509–1516
Ravelonandro M, Scorza R, Minoiu N, Zagrai I, Platon I (2002) Field tests of transgenic plums in Romania. Sănătatea Plant 6:16–17
Rubio T, Borja M, Scholthof HB, Jackson AO (1999) Recombination with host transgenes and effects on virus evolution: An overview and opinion. Mol Plant Microbe Interact 12:87–92
Scorza R, Ravelonandro M, Callahan AM, Cordts JM, Fuchs M, Dunez J, Gonsalves D (1994) Transgenic plums (Prunus domestica L.) express the Plum pox virus coat protein gene. Plant Cell Rep 14:18–22
Scorza R, Callahan A, Levy L, Damsteegt V, Webb K, Ravelonandro M (2001) Post-transcriptional gene silencing in Plum pox virus resistant transgenic European plum containing the Plum pox potyvirus coat protein gene. Transgenic Res 10:201–209
Scorza R, Hily JM, Callahan A, Malinowski T, Cambra M, Capote N, Zagrai I, Damsteeg V, Briard P and Ravelonandro M (2007) Deregulation of plum pox resistant transgenic plum “HoneySweet”. Acta Hortic (in press)
Szemes M, Kalman M, Myrta A, Boscia D, Nemeth M, Kölber M, Dorgai L (2001) Integrated RT-PCR/nested PCR diagnosis for differentiating between subgroups of Plum pox virus. J Virol Methods 92:165–175
Thompson DA, Gibson TJ, Plewniak F, Jeanmougin F, Higgins DG (1997) The Clustal-X windows interface: flexible strategies for multiple sequence alignment aided by quality analysis tools. Nucleic Acids Res 24:4876–4882
Thomas PE, Hassan S, Kaniewski WK, Lawson EC, Zalewski JC (1998) A search for evidence of virus/transgene interactions in potatoes transformed with the Potato leafroll virus replicase and coat protein genes. Mol Breeding 4:407–417
Varrelman M, Palkovics L, Maiss E (2000) Transgenic or plant expression vector-mediated recombination of Plum pox virus. J Virol 74:7462–7469
Varveri C, Zintzaras E, Dimou D, Papapanagiotou A, Di Terlizzi B (2004) Monitoring and spatio-temporal analysis of PPV-M spread in two apricot orchards in Southern Greece. Ann Benaki Phytopathol Inst 20:1–9
Vigne E, Komar V, Fuchs M (2004) Field safety assessment of recombination in transgenic grapevines expressing the coat protein gene of Grapevine fanleaf virus. Transgenic Res 13:165–179
Wallis CM, Fleischer SJ, Luster D, Gildow FE (2005) Aphid (Hemiptera: Aphididae) species composition and potential aphid vectors of Plum pox virus in Pennsylvania peach orchards. J Econ Entomol 98:1441–1450
Wetzel T, Candresse T, Ravelonandro M, Dunez J (1991) A polymerase chain reaction assay adapted to Plum pox potyvirus detection. J Virol Methods 33:355–365
Wintermantel WM, Schoelz JE (1996) Isolation of recombinant viruses between cauliflower mosaic virus and a viral gene in transgenic plants under conditions of moderate selection pressure. Virology 223:156–164
Acknowledgements
This work was supported by the European project TRANSVIR (QLK3-CT-2002-02140), INIA (SC98-060 and RTA03-099) and Ministerio de Educación y Ciencia (RTA05-00190) projects. Authors thank to C. Collado (IVIA) for determination of aphid species, to B. Tamargo and J. Micó (Cooperativa Vinícola de Liria) for technical assistance in the experimental plot, to Dr. S. Elena for assistance in recombination analyses and to Dr. M. Fuchs for critical reading of the manuscript. Permissions for field release of GMO Nos. B/ES/96/16 and B/ES/05/14 were given by the Spanish Ministerio de Medio Ambiente.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Capote, N., Pérez-Panadés, J., Monzó, C. et al. Assessment of the diversity and dynamics of Plum pox virus and aphid populations in transgenic European plums under Mediterranean conditions. Transgenic Res 17, 367–377 (2008). https://doi.org/10.1007/s11248-007-9112-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11248-007-9112-0