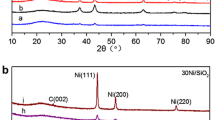
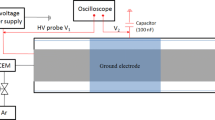
Abstract
Activity control for CO dissociation over supported Ni catalysts is very important for steam reforming, CO2 reforming, methanation, carbon formation, and other processes. In this work, a series of Ni/SiO2 catalysts were designed through thermal decomposition or dielectric barrier discharge (DBD) plasma decomposition. After a high temperature reduction, the main difference between the two catalysts was the structure of Ni nanoparticles, instead of the particles size. The plasma decomposed catalyst possesses smooth surface on Ni nanoparticles with less defect sites, leading to the activity control of CO dissociation. DBD plasma decomposed Ni/SiO2 catalysts have a low activity for CO dissociation with a low final carbon yield. The as-produced carbon materials over thermally decomposed or plasma decomposed are in the form of carbon onion particles or carbon nanotubes, separately. The growth mechanism of these two different carbon materials was also discussed.






Similar content being viewed by others
References
Liu CJ, Ye JY, Jiang JJ, Pan YX (2011) ChemCatChem 3:529–541
Kameshima S, Tamura K, Ishibashi Y, Nozaki T (2015) Catal Today 256:67–75
Nozaki T, Okazaki K (2013) Catal Today 211:29–38
Cheng DG, Zhu XL, Ben YH, He F, Cui L, Liu CJ (2006) Catal Today 115:205–210
Guo F, Xu JQ, Chu W (2015) Catal Today 256:124–129
Liu L, Wang S, Guo Y, Wang BL, Rukundo P, Wen SP, Wang ZJ (2016) Int J Hydrog Energy 41:17361–17369
Pan YX, Kuai PY, Liu Y, Ge QF, Liu CJ (2010) Energy Environ Sci 3:1322–1325
Liu DP, Quek XY, Cheo WNE, Lau R, Borgna A, Yang YH (2009) J Catal 266:380–390
Sumi H, Lee YH, Muroyama H, Matsui T, Kamijo M, Mimuro S, Yamanaka M, Nakajima Y, Eguchi K (2011) J Power Sour 196:4451–4457
Balakos MW, Chuang SSC (1993) React Kinet Catal Lett 49:7–12
Liu Q, Zhong ZY, Gu FN, Wang XY, Lu XP, Li HF, Xu GW, Su FB (2016) J Catal 337:221–232
Yan XL, Zhao BR, Liu Y, Li YN (2015) Catal Today 256:29–40
Bartholomew CH (1982) Catal Rev Sci Eng 24:67–112
McCarty JG, Wise H (1979) J Catal 57:406–416
Mirodatos C, Praliaud H, Primet M (1987) J Catal 107:275–287
Tavares MT, Alstrup I, Bernardo CA, Rostrup-Nielsen JR (1996) J Catal 158:402–410
Pan YX, Liu CJ, Shi P (2008) J Power Sour 176:46–53
Zhu XL, Huo PP, Zhang YP, Cheng DG, Liu CJ (2008) Appl Catal B 81:132–140
Pan YX, Liu CJ, Cui L (2008) Catal Lett 123:96–101
Zhu XL, Cheng DG, Kuai PY (2008) Energy Fuels 22:1480–1484
Zhao BR, Yan XL, Zhou Y, Liu CJ (2013) Ind Eng Chem Res 52:8182–8188
YanXL, Liu Y, Zhao BR, Wang Z, Wang Y, Liu CJ (2013) Int J Hydrog Energy 38:2283–2291
Yan XL, Liu Y, Zhao BR, Wang Y, Liu CJ (2013) Phys Chem Chem Phys 15:12132–12138
Zhou R, Rui N, Fan ZG, Liu CJ (2016) Int J Hydrogen Energy 41:22017–22025
Yan XL, Liu CJ (2013) Diam Relat Mater 31:50–57
Guo QT, With P, Liu Y, Gläser R, Liu CJ (2013) Catal Today 211:156–161
Liu Y, Wang Z, Liu CJ (2015) Catal Today 256:137–141
Liu CJ, Li MY, Wang JQ, Zhou XT, Guo QT, Yan JM, Li YZ (2016) Chin J Catal 37:340–348
Liu Y, Pan YX, Wang ZJ, Kuai PY, Liu CJ (2010) Catal Commun 11:551–554
Zou JJ, Liu CJ, Zhang YP (2006) Langmuir 22:2334–2339
Chen Y, Wang B, Li LJ, Yang YH, Ciuparu D, Lim SY, Haller GL, Pfefferle LD (2007) Carbon 45:2217–2228
Kitiyanan B, Alvarez WE, Harwell JH, Resasco DE (2000) Chem Phys Lett 317:497–503
Lin W, Moon KS, Zhang SJ, Ding Y, Shang JT, Chen MX, Wong CP (2010) ACS Nano 4:1716–1722
Reddy ALM, Shaijumon MM, Ramaprabhu S (2006) Nanotechnology 17:5299–5305
Cao ZS, Qiu L, Yang YZ, Chen YK, Liu XG (2015) Appl Surf Sci 353:873–881
Zhang Y, Wang W, Wang ZY, Zhou XT, Wang Z, Liu CJ (2015) Catal Today 256:130–136
Andersson MP, Abild-Pedersen F, Remediakis IN, Bligaard T, Jones G, Engbæk J, Lytken O, Horch S, Nielsen JH, Sehested J, Rostrup-Nielsen JR, Nørskov JK, Chorkendorff I (2008) J Catal 255:6–19
Wang ZX, Qiao QA, Chen SG, Zhang WX (2002) Surf Sci 517:29–42
Steinrück HP, Develyn MP, Madix RJ (1986) Surf Sci 172:561–567
Tibbetts GG (1984) J Cryst Growth 66:632–638
Jiang KL, Feng C, Liu K, Fan SS (2007) J Nanosci Nanotechnol 7:1494–1504
Nolan PE, Michael JS, Lynch DCS (1995) Carbon 33:79–85
Nasibulin AG, Moisal A, Brown DP, Kauppinen EI (2003) Carbon 41:2711–2724
Acknowledgements
We acknowledge the National Natural Science Foundation of China (Nos. 21406153, 21406177, 21576177, and 21546009), Shanxi Province Science Foundation for Youths (No. 2014021014-2), the Supported by Program for the Outstanding Innovative Teams of Higher Learning Institutions of Shanxi, Scientific Research Program Funded by Shaanxi Provincial Education Department (Program No. 14JK1749) and the support from Taiyuan University of Technology (1205-04020202).
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Yan, X., Bao, J., Zhao, B. et al. CO Dissociation on Ni/SiO2: The Formation of Different Carbon Materials. Top Catal 60, 890–897 (2017). https://doi.org/10.1007/s11244-017-0754-8
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11244-017-0754-8