Abstract
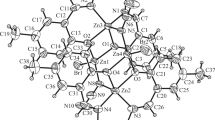
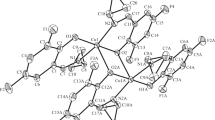
The copper aminotropones Cu[ON(R′)C7H4R-4]2 [R = H, R′ = Me (13), Et (14), n-Pr (15), n-Bu (16), Bz (17), MenOCH2CH2 (20); R = i-Pr, R′ = Me (18), n-Pr (19), MenOCH2CH2 (21)] have been prepared from the corresponding aminotropones HN(R′)OC7H4R-4 (1–7) by reacting with copper(II) acetate in aqueous ethanol. 20, 21 contain the flavourant, menthol, as part of the ligand. The structures of 5 (R = H, R′ = Bz), a hydrogen-bonded dimer, 14 and 20, both incorporating square-planar, four-coordinate copper centres, have been determined by X-ray crystallography. The antibacterial activities of complexes 13, 17, 20 and 21 have been assayed against Staphylococcus waneri, an in vitro model of plaque inhibition effects, and found to be more active than a commercial toothpaste formulation, but less active than the O,O-chelated copper(II) complex of ethylmaltol.






Similar content being viewed by others
References
Arima Y, Nakai Y, Hayakawa R, Nishino T (2003) J Antimicrob Chemother 51:113
Morita Y, Matsumura E, Okabe T, Shibata M, Sugiura M, Ohe T, Tsujibo H, Ishida N, Inamori Y (2003) Biol Pharm Bull 26:1487
Inamori Y, Sakagami Y, Morita Y, Shibata M, Sugiura M, Kumeda Y, Okabe T, Tsujibo H, Ishida N (2000) Biol Pharm Bull 23:995
Inamori Y, Tsujibo H, Ohishi H, Ishii F, Mizugaki M, Aso H, Ishida N (1993) Biol Pharm Bull 16:521
Nomiya K, Onodera K, Tsukagoshi K, Shimada K, Yoshizawa A, Tada-aki I, Sugie A, Tsuruta S, Sato R, Kasuga C (2009) Inorg Chim Acta 362:43
Irving RJ, Post ML, Povey DC (1973) J Chem Soc, Dalton Trans 2:697
Berg J-E, Pilotti A-M, Söderholm A-C, Karlsson B (1978) Acta Crystallog B34:3071
Griffith WP, Pumphrey CA, Skapski AC (1987) Polyhedron 6:891
Avdeef A, Costamanga JA, Fackler JPJ (1974) Inorg Chem 13:1854
Davis AR, Einstein FWB (1974) Inorg Chem 13:1880
Lockhart TP, Davidson F (1987) Organometallics 6:2471
Mori A, Yamamoto S-I, Takemoto M, Ujiie S (2002) Chem Lett 2:186
Mori A, Mori R, Takemoto M, Yamamoto S-I, Kuribayashi D, Uno K, Kubo K, Ujiie S (2005) J Mater Chem 15:3005
Creeth J, Molloy KC, Wright P (2000) Oral care compositions. Patent WO 00/16736
Barret MC, Mahon MF, Molloy KC, Wright P (2000) Main Group Met Chem 23:663
Barret MC, Mahon MF, Molloy KC, Steed JW, Wright P (2001) Inorg Chem 40:4384
Barret MC, Mahon MF, Molloy KC, Wright P, Creeth JE (2002) Polyhedron 21:1761
Steyl G, Muller TJ, Roodt A (2010) Acta Crystallog Sect E Struct Rep Online 66:m1508
Miyake Y, Watanabe S, Aono S, Nishinaga T, Miyazaki A, Enok T, Miyasaka H, Otani H, Iyoda M (2008) Chem Commun 44:6167
Nishinaga T, Aono T, Iomura E, Watanabe S, Miyake Y, Miyazaki A, Enoki T, Miyasaka H, Otani H, Iyoda M (2010) Dalton Trans 39:2293
Meyer N, Lohnwitz K, Zulys A, Roesky PW, Dochnahl M, Blechert S (2006) Organometallics 25:3730
Reddington MV (1998) J Chem Soc Perkin Trans II 143
Rasika-Dias HV, Jin W (1996) Inorg Chem 35:6546
Biggi G, Del Cima F, Pietra F (1973) J Am Chem Soc 95:7101
Cochran TG, Huitric AC (1971) J Org Chem 36:3046
Minardi G, Schenone P (1968) Farmaco Ed Sci 23:1181
Farrugia LJ (1999) J Appl Crystallogr 32:837
Sheldrick G (2008) Acta Cryst A64:112
Yanagisawa T, Kosakai K, Yasunami M, Takase K (1990) Chem Pharm Bull 38:3355
Hicks FA, Jenkins JC, Brookhart M (2003) Organometallics 22:3533
Steyl G (2007) Acta Crystallogr Sect E: Struct Rep Online 63:o4353
Siwatch RK, Kundu S, Kumar D, Nagendran S (2011) Organometallics 30:1998
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Barret, M.C., Bhatia, P.H., Kociok-Köhn, G. et al. New copper(II) 2-(alkylamino)troponates. Transition Met Chem 39, 543–551 (2014). https://doi.org/10.1007/s11243-014-9830-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11243-014-9830-0