Abstract
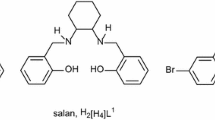
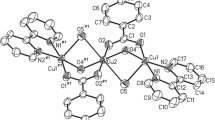
A mononuclear complex [CuL] (1), a binuclear complex [Cu2LCl2(H2O)] (2), a trinuclear complex [Cu3L2](ClO4)2 (3) involving o-phenylenediamine and salicylaldehyde and another binuclear complex of a tridentate ligand (H2L1) [Cu2L 12 ](CH3COO)2 (4) involving o-phenylenediamine and diacetylmonoxime have been synthesized, where H2L = N,N′-o-phenylenebis(salicylideneimine) and H2L1 = 3-(2-aminophenylimino)butan-2-one oxime. All the complexes have been characterized by elemental analyses, spectral and magnetic studies. The binuclear complex (2) was characterized structurally where the two Cu(II) centers are connected via an oxygen-bridged arrangement.






Similar content being viewed by others
References
Sigel H (1980) Inorg Chem 19:1411
Elder RC, Blubaugh EA Jr, Heinmann WR, Bruke PJ, McMillian RD (1983) Inorg Chem 22:2771
Gaykema WPJ, Volveda A, Hol WJH (1985) J Mol Biol 187:2255
Gaykema WPJ, Hol WGH (1989) J Mol Biol 209:249
Bu XR, Jackson CR, Derveer DV, You XZ, Meng QJ, Wang RX (1997) Polyhedron 17:2991–3001
Ray MS, Mukhopadhyay G, Bhattacharya R, Chaudhuri S, Righi L, Bocelli G, Ghosh A (2003) Polyhedron 22:617
Chattopadhyay S, Ray MS, Chaudhuri S, Mukhopadhyay G, Bocelli G, Cantoni A, Ghosh A (2006) Inorg Chim Acta 359:1367
Ray MS, Ghosh A, Bhattacharya R, Mukhopadhyay G, Drew MGB, Ribas J (2004) Dalton Trans 252
Ray MS, Mukhopadhyay G, Drew MGB, Lu T-H, Chaudhuri S, Ghosh A (2003) Inorg Chem Commun 6:961
Ray MS, Ghosh A, Chaudhuri S, Drew MGB, Ribas J (2004) Eur J Inorg Chem 15:3110
Maity D, Mukherjee P, Ghosh A, Drew MGB, Mukhopadhyay G (2008) Inorg Chim Acta 361:1515
Maity D, Chattopadhyay S, Ghosh A, Drew MGB, Mukhopadhyay G (2009) Polyhedron 28:812–818
Kanderal OM, Kozlowski H, Dobosz A, Kozlowska JS, Meyer F, Fritsky OI (2005) J Chem Soc Dalton Trans 1428
Costes JP, Dahan F, Dupuis A, Laurent JP (1998) J Chem Soc Dalton Trans 1307
Kukushkin VY, Tudela D, Pombeiro AJL (1996) Coord Chem Rev and references cited therein. 156: 333–362
Chaudhuri P (2003) Coord Chem Rev and references cited therein. 243: 143–190
Tan S-F, Ang K-P (1988) Transition Met Chem 13:64
Gruber SJ, Harris CM, Sinn E (1968) J Inorg Nucl Chem 30:1805
Lever ABP (1984) Inorganic electronic spectroscopy, 2nd edn. Elsevier Science Publishers, Amsterdam
Addison AW, Rao TN, Reedijk J, van Rijn J, Verschoor GC (1984) J Chem Soc Dalton Trans 1349
Thompson LK, Mandal SK, Tandon SS, Bridson JN, Park MK (1996) Inorg Chem 35:3117
Ruiz E, Alemany P, Alvarez S, Cano J (1997) Inorg Chem 36:3683
CrysAlis (2008) Oxford Diffraction Ltd., Abingdon, UK
Abspack Program (2007) Oxford Diffraction Ltd., Abingdon, UK
Sheldrick GM, Shelxs97 and Shelxl97, Programs for Crystallographic solution and refinement Acta Crystallogr A64:112 (2008)
Acknowledgments
We thank EPSRC and the University of Reading for funds for the X-Calibur system. Thanks are also due to Prof. Ashutosh Ghosh, Department of Chemistry, University College of Science, University of Calcutta, 92, A.P.C. Road, Kolkata-700 009, India, for his valuable suggestions during preparation of the manuscript and Sri Sanjib Giri of Indian Association for the Cultivation of Science, Jadavpur, Kolkata-700 032, India, for the magnetic measurements.
Author information
Authors and Affiliations
Corresponding author
Appendix A. Supplementary data
Appendix A. Supplementary data
Crystallographic data for the analysis have been deposited with the Cambridge Crystallographic data Centre, CCDC No 721696 (complex 2). Copies of this information may be obtained free of charge from CCDC, 12 Union Road, Cambridge, CB21EZ, UK (fax:+441223336033; email:deposit@ccdc.cam.ac.uk or www: http://www.ccdc.cam.ac.uk).
Rights and permissions
About this article
Cite this article
Maity, D., Drew, M.G.B., Godsell, J.F. et al. Synthesis and characterization of Cu(II) complexes of tetradentate and tridentate symmetrical Schiff base ligands involving o-phenelenediamine, salicylaldehyde and diacetylmonoxime. Transition Met Chem 35, 197–204 (2010). https://doi.org/10.1007/s11243-009-9314-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11243-009-9314-9