Abstract
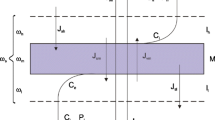
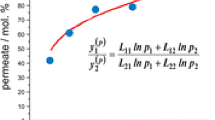
The Kedem-Katchalsky equations, modified by means of symmetric transformations of Peusner thermodynamic networks, were applied to interpret the membrane transport in concentration polarization conditions. The results from the study demonstrate that the resistance coefficients counted for membrane transport of aqueous solutions of glucose through Nephrophan membrane in horizontal plane are nonlinearly dependent on mean concentration of glucose in the membrane \({(\bar{C})}\) . It was also shown that the threshold value of concentration \({(\bar{C}_{cr})}\) existed, and for \({\bar{C} > \bar{C}_{cr}}\) , the resistance coefficients depend, while for \({\bar{C} < \bar{C}_{cr}}\) , they do not depend on the membrane system configuration. Increase of mean glucose concentration in the membrane (in the range \({\bar{C} > \bar{C}_{cr})}\) causes decrease of difference between resistance coefficients of the membrane system in homogeneous conditions (solutions mechanically stirred) and in conditions with hydrodynamic instabilities (configuration B). Besides increase of mean glucose concentration in the membrane (in the range \({\bar{C} > \bar{C}_{cr})}\) causes increase of the difference between resistance coefficients for membrane system with concentration polarization without hydrodynamic instabilities (configuration A) and membrane system in homogeneous conditions.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Abbreviations
- R ij :
-
Generalized resistance coefficients
- X i :
-
Thermodynamic forces in homogeneous conditions
- J i :
-
Thermodynamic fluxes in homogeneous conditions
- \({X_{i}^*}\) :
-
Thermodynamic forces in non-homogeneous conditions
- \({J_{i}^*}\) :
-
Thermodynamic fluxes in non-homogeneous conditions
- J v :
-
Volume flux in homogeneous conditions
- J vs :
-
Volume flux in non-homogeneous conditions
- J s :
-
Solute flux in homogeneous conditions
- J ss :
-
Solute flux in non-homogeneous conditions
- J sa :
-
Solute advective flux
- L p :
-
Hydraulic permeability coefficient
- σ :
-
Reflection coefficient
- ω :
-
Solute permeability coefficient
- ν :
-
Kinematic viscosity
- δ k :
-
Thickness of concentration boundary layers
- P h , P l :
-
Hydrostatic pressure (h higher and l lower value)
- π :
-
Osmotic pressure
- C h ,C l :
-
Solute concentrations in chambers of the membrane system
- \({\bar{C}}\) :
-
Mean solute concentration in the membrane
- R :
-
Gas constant
- R C :
-
Concentration Rayleigh number
- T :
-
Thermodynamic temperature
- D k :
-
Diffusion coefficient
- ζ p :
-
Hydraulic concentration polarization coefficient
- ζ v :
-
Osmotic concentration polarization coefficient
- ζ s :
-
Diffusive concentration polarization coefficient
- ζ a :
-
Advective concentration polarization coefficient
References
Barry P.H., Diamond J.M.: Effects of unstirred layers on membrane phenomena. Physiol. Rev. 64, 763–872 (1984)
Demirel Y.: Nonequilibrium Thermodynamics, Transport and Rate Processes in Physical and Biological Systems. Elsevier, Amsterdam (2002)
Dworecki K.: Interferometric investigations of near-membrane diffusion layers. J. Biol. Phys. 21, 37–49 (1995)
Dworecki K., Ślęzak A., Ornal-Wåsik B., Wåsik S.: Effect of hydrodynamic instabilities on solute transport in a membrane system. J. Membr. Sci. 265, 94–100 (2005)
Ewing G.W.: Instrumental methods of chemical analysis. McGraw-Hill, New York (1985)
Ginzburg B. Z., Katchalsky A.: The frictional coefficients of the flows of non- electrolytes through artificial membranes. J. Gen. Physiol. 47, 403–418 (1963)
Grzegorczyn S., Jasik-Ślęzak J., Michalska-Małecka K., A.: Transport of non-electrolyte solutions through membrane with concentration polarization. Gen. Physiol. Biophys. 27, 315–321 (2008)
Imai Y.: Network thermodynamics: analysis and synthesis of membrane transport system. Japan. J. Physiol. 46, 187–199 (1996)
Imai Y.: Graphic modeling of epithelial transport system: causality of dissipation. BioSystems 70, 9–19 (2003)
Jasik-Ślęzak J., A.: Thermodynamical description of the concentration polarization in a membrane transport of nonelectrolyte solution. Polym. Med. 40, 49–55 (2010)
Jasik-Ślęzak J., Olszówka K.M., A.: Estimation of thickness of concentration boundary layers by osmotic volume flux determination. Gen. Physiol. Biophys. 30, 186–195 (2011)
Kargol A.: Effect of boundary layers on reverse osmosis through a horizontal membrane. J. Membr. Sci. 159, 177–184 (1999)
Kargol M., Kargol A.: Membrane transport generated by the osmotic and hydrostatic pressure. Correlation relation for parameters l p , σ and ω. J. Biol. Phys. 26, 307–320 (2000)
Kargol A.: Modified Kedem-Katchalsky equations and their application. J. Membr. Sci. 174, 43–53 (2000)
Katchalsky A., Curran P. F.: Nonequilibrium Thermodynamics in Biophysics. Harvard, Cambridge (1965)
Klinkman H., Holtz M., Willgerodt W., Wilke G., Schoenfelder D.: Nephrophan—eine neue dialysemembranes. Z. Urol. 4, 285–292 (1969)
Larchet C., Nouri S., Auclair B., Dammak L., Nikonenko V.: Application of chronopotentiometry to determine the thickness of diffusion layer adjacent to an ion-exchange membrane under natural convection. Adv. Colloid Interface Sci. 139, 45–61 (2008)
Meixner J.: Network theory in its realation to thermodynamics. In: Fox, J. (ed.) Proceedings of the Symposium on Generalized Networks, pp. 13–25. Wiley, New York (1966)
Mikulecky D.: The circle that never ends: can complexity be made simple? In: Bonvchev, D.D., Rouvaray, D. (eds.) Complexity in Chemistry, Biology and Ecology, pp. 97–153. Springer, Berlin (2005)
Mishchuk N.A.: Concentration polarization of interface and non-linear electrokinetic phenomena. Adv. Colloid Interface Sci. 160, 16–39 (2010)
Newman S.A., Forgacs G.: Complexity and self-organization in biological development and evolution. In: Bonvchev, D.D., Rouvaray, D. (eds.) Complexity in Chemistry, Biology and Ecology, pp. 49–96. Springer, Berlin (2005)
Nicolis G., Prigogine I.: Self-Organization in Nonequilibrium Systems. From Dissipative Structures to Order Through Fluctuactions. Wiley, New York (1977)
Oster G.F., Perelson A., Katchalsky A.: Network thermodynamics. Nature 234, 393–399 (1971)
Perelson A.S.: Network thermodynamics. Biophys. J. 15, 667–685 (1975)
Peusner, L.: The principles of network thermodynamics and biophysical applications. Ph D Thesis, Harvard, Cambridge (1970)
Peusner L.: Hierarchies of irreversible energy conversion systems: a network thermodynamics approach. I. Linear steady state without storage. J. Theoret. Biol. 102, 7–39 (1983)
Peusner L.: Hierarchies of irreversible energy conversion systems. II. Network derivation of linear transport equations. J. Theoret. Biol. 115, 319–335 (1985a)
Peusner L.: Network representation yelding the evolution of Brownian motion with multiple particle interaction. Phys. Rev. 32, 1237–1238 (1985b)
Peusner L., Mikulecky D.C., Bunow B., Caplan S.R.: A network thermodynamic approach to hill and King-Altman reaction-diffusion kinetics. J. Chem. Phys. 83, 5559–5566 (1985)
Peusner L.: Hierarchies of irreversible energy conversion processes. III. Why are Onsager equations reciprocal? The Euclidean geometry of fluctuaction-dissipation space. J. Theoret. Biol. 122, 125–155 (1986a)
Peusner L.: Studies in Network Thermodynamics. Elsevier, Amsterdam (1986b)
Playtner H.: Analysis and Design of Engineering Systems. MIT, Cambridge (1961)
Puthenveettil B.A., Arakeri J.H.: Convection due to an unstable density difference across a permeable membrane. J. Fluid. Mech. 609, 139–170 (2008)
Puthenveettil B.A., Gunasegarane G.S., Agrawal Y.K., Arakeri J.H.: Length of near-wall plumes in turbulent convection. J. Fluid Mech. 685, 335–364 (2011)
Ramareddy G.V., Puthenveettil B.A.: The Pe ∼ 1 regime of convection across a horizontal permeable membrane. J. Fluid. Mech. 679, 476–504 (2011)
Rubinstein I., Zaltzman B.: Electro-osmotically induced convection at a permselective membrane. Phys. Rev. E 62, 2238–2251 (2000)
Soh K.C., Hatzimanikatis V.: Network thermodynamics in the post-genomic era. Curr.Opin. Microbiol. 13, 350–357 (2010)
Dworecki A., Dworecki K., Anderson J.E.: Gravitational effects on transmembrane flux: the Rayleigh-Taylor convective instability. J. Membr. Sci. 23, 71–81 (1985)
Turczyński A., Turczyński B.: Modiffication of the Kedem-Katchalsky equations. Biophys. Chem. 24, 173–178 (1986)
A.: Irreversible thermodynamic model equations of the transport across a horizontally mounted membrane. Biophys. Chem. 34, 91–102 (1989)
Ślęzak A., Jasik-Ślęzak J., Wåsik J., Sierot A., Pilis W.: Volume osmotic flows of non-electrolyte solutions through horizontally mounted membrane. Gen. Physiol. Biophys 21, 115–146 (2002)
Dworecki A., Dworecki K., Wåsik I.H., Wåsik S.: Permeability coefficient model equations of the complex: membrane-concentration boundary layers for ternary nonelectrolyte solutions. J. Membr. Sci. 267, 50–57 (2005)
Grzegorczyn A., Grzegorczyn S., Jasik-Ślęzak J., Michalska-Małecka K.: Natural convection as an asymmetrical factor of the transport through porous membrane. Transp. Porous Media 84, 685–698 (2010)
A.: Application of the network thermodynamics to interpretation of membrane transport: evaluation of the resistance coefficients of the polymeric membrane in polarization concentration conditions. Polim. Med. 41, 43–51 (2011)
Acknowledgments
This study was supported by the Ministry of Science and Higher Education under Grant No. BS/PB -622/3010/2011/P.
Open Access
This article is distributed under the terms of the Creative Commons Attribution License which permits any use, distribution, and reproduction in any medium, provided the original author(s) and the source are credited.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 2.0 International License (https://creativecommons.org/licenses/by/2.0), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
About this article
Cite this article
Ślęzak, A., Grzegorczyn, S. & Batko, K.M. Resistance Coefficients of Polymer Membrane with Concentration Polarization. Transp Porous Med 95, 151–170 (2012). https://doi.org/10.1007/s11242-012-0038-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11242-012-0038-5