Abstract
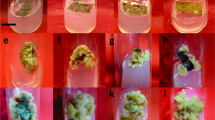
Copper (Cu) accumulation, subcellular distribution and the chemical forms of Cu, and amino acids metabolism were investigated in castor bean (Ricinus communis L.) callus via in vitro culture. The castor bean callus was obtained from the embryo cultured in Murashige and Skoog (MS) medium and then transferred to MS medium with different Cu doses (0, 20, 40, and 60 mg L−1; Cu0, Cu20, etc.) for 28-days cultivation. The stress from Cu inhibited the growth of the castor bean callus, and the Cu content in the castor bean callus increased with the increasing Cu dose, reaching a maximum value of 293.2 mg kg−1 fresh weight (FW) in the 60 mg L−1 Cu treatment. Concentrations of Cu in the cell wall, organelles, and cytoplasm increased significantly with the elevated Cu dose, with the cell wall containing 50.2 % of the total Cu in the 60 mg L−1 Cu treatment. The major Cu fractions were C (bound to the exchangeable polar compounds) (28.1 %) and E (bound to the structural polar compounds and nucleic acids) (27.5 %) in the control treatment, and the main fraction was C (51.2 %) in the treatment with 60 mg L−1 Cu. The concentration of free amino acids in the cytoplasm was closely related to the Cu content in the castor bean callus. In addition, most of the Cu in the cell wall bound with functional groups of the cell chemical components, hydroxyl (–OH), acylamino (–CONH2), and carboxylate ion (–COO−). The castor bean exhibited a strong tolerance to Cu, which accumulated mainly in the cell wall.






Similar content being viewed by others
Abbreviations
- 6-BA:
-
6-Benzylaminopurine
- NAA:
-
1-Naphthaleneacetic acid
- MS medium:
-
Murashige and Skoog medium
- FTIR:
-
Fourier transform infrared spectroscopy
- AAS:
-
Atomic absorption spectrometry
- MTs:
-
Metallothioneins
- Tris:
-
Tris (hydroxymethyl) aminomethane
References
Abreu CA, Coscione AR, Pires AM, Paz-Ferreiro J (2012) Phytoremediation of a soil contaminated by heavy metals and boron using castor bean oil plants and organic matter amendments. J Geochem Explor 123:3–7
Carra A, Sajeva M, Abbate L, Siragusa M, Sottile F, Do FC (2012) In vitro plant regeneration of caper (Capparis spinosa L.) from floral explants and genetic stability of regenerants. Plant Cell Tissue Organ Cult 109(2):373–381
Cobbett C, Goldsbrough P (2002) Phytochelatins and metallothioneins: roles in heavy metal detoxification and homeostasis. Annu Rev Plant Biol 53(1):159–182
Cohu CM, Pilon M (2007) Regulation of superoxide dismutase expression by copper availability. Physiol Plant 129:747–755
Fatima N, Ahmad N, Anis M (2011) Enhanced in vitro regeneration and change in photosynthetic pigments, biomass and proline content in Withania somnifera L. (Dunal) induced by copper and zinc ions. Plant Physiol Bioch 49:1465–1471
Fernández R, Fernández-Fuego D, Bertrand A, González A (2014) Strategies for Cd accumulation in Dittrichia viscosa (L.) Greuter: Role of the cell wall, non-protein thiols and organic acids. Plant Physiol Bioch 78:63–70
Gori P, Schiff S, Santandrea G, Bennici A (1998) Response of in vitro cultures of Nicotiana tabacum L. to copper stress and selection of plants from Cu-tolerant callus. Plant Cell Tissue Organ Cult 53:161–169
Grill E, Winnacker EL, Zenk MH (1985) Phytochelatins: the principal heavy-metal complexing peptides of higher plants. Science 230(4726):674–676
Haydon MJ, Cobbett CS (2007) Transporters of ligands for essential metal ions in plants. New Phytol 174(3):499–506
Huang GY, Guo GG, Yao SY, Zhang N, Hu HQ (2016) Organic acids, amino acids compositions in the root exudates and Cu-accumulation in castor bean (Ricinus communis L.) under Cu stress. Int J Phytoremediat 18(1):33–40
Iqbal M, Saeed A, Zafar SI (2009) FTIR spectrophotometry, kinetics and adsorption isotherms modeling, ion exchange, and EDX analysis for understanding the mechanism of Cd2+ and Pb2+ removal by mango peel waste. J Hazard Mater 164(1):161–171
Jeong AS, Damodaran PN, Kwang SR (2014) Influence of exogenous application of glutathione on rubisco and rubisco activase in heavy metal-stressed tobacco plant grown in vitro. Saudi J Biol Sci 21(1):89–97
Jiang L, Shi W, Yang X, Fu C, Chen W (2002) Cu hyperaccumulators in mining area. Chin J Appl Ecol 13(7):906–908
Johnson A, Gunawardana B, Singhal N (2009) Amendments for enhancing copper uptake by Brassica juncea and Lolium perenne from solution. Int J Phytoremediat 11(3):215–234
Kang W, Zheng J (2011) Ricinus communis, a new copper hyperaccumulator. J Anhui Agr Sci 39(3):1449–1451, 1466
Kang W, Bao JG, Zheng J, Hu HQ, Du J (2015) Distribution and chemical forms of copper in the root cells of castor bean seedlings and their tolerance to copper phytotoxicity in hydroponic culture. Environ Sci Pollut Res 22(10):7726–7734
Kartel MT, Kupchik LA, Veisov BK (1999) Evaluation of pectin binding of heavy metal ions in aqueous solutions. Chemosphere 38:2591–2596
Konno H, Nakashima S, Katoh K (2010) Metal-tolerant moss Scopelophila cataractae accumulates copper in the cell wall pectin of the protonema. J Plant Physiol 167(5):358–364
Kumari KG, Ganesan M, Jayabalan N (2008) Somatic organogenesis and plant regeneration in Ricinus communis. Biol Plantarum 52(1):17–25
Kuroda K, Shibasaki S, Ueda M, Tanaka A (2001) Cell surface-engineered yeast displaying a histidine oligopeptide (hexa-His) has enhanced adsorption of and tolerance to heavy metal ions. Appl Microbiol Biot 57(5):697–701
Lasat MM (2002) Phytoextraction of toxic metals: a review of biological mechanisms. J Environ Qual 31(1):109–120
Liu J, Duan CQ, Zhang XH, Zhu YN, Hu C (2009) Subcellular distribution of chromium in accumulating plant Leersia hexandra Swartz. Plant Soil 322(1):187–195
Lukatkin A, Egorova I, Michailova I, Malec P, Strzałka K (2014) Effect of copper on pro- and antioxidative reactions in radish (Raphanus sativus L.) in vitro and in vivo. J Trace Elem Med Bio 28(1):80–86
Mackie KA, Müller T, Kandeler E (2012) Remediation of copper in vineyards: a mini review”. Environ Pollut 167:16–26
Majumdar SS, Das SK, Saha T, Panda GC, Bandyopadhyoy T, Guha AK (2008) Adsorption behavior of copper ions on Mucor rouxii biomass through microscopic and FTIR analysis. Colloids Surf B Biointerfaces 63(1):138–145
Martins JPR, Martins AD, Pires MF, Junio RAB, Reis RO, Dias GDMG, Pasqual M (2016) Anatomical and physiological responses of Billbergia zebrina (Bromeliaceae) to copper excess in a controlled microenvironment. Plant Cell Tissue Organ Cult 126:43–57
Megateli S, Semsari S, Couderchet M (2009) Toxicity and removal of heavy metals (cadmium, copper, and zinc) by Lemna gibba. Ecotox. Environ Safe 72(6):1774–1780
Merdy P, Guillon E, Dumonceau J, Aplincourt M (2002) Spectroscopic study of copper (II)–wheat straw cell wall residue surface complexes. Environ Sci Technol 36(8):1728–1733
Mohan Ram HY, Satsangi A (1963) Induction of cell divisions in the mature endosperm of Ricinus communis during germination. Curr Sci 32:28–30
Ni C, Chen Y, Lin Q, Tian G (2005) Subcellular localization of copper in tolerant and non-tolerant plant. J Environ Sci China 117(3):452–456
Niu ZX, Sun LN, Sun TH, Li YS WH (2007) Evaluation of phytoextracting cadmium and lead by sunflower, ricinus, alfalfa and mustard in hydroponic culture. J. Environ Sci China 19:961–967
Olivares AR, Carrillo-González R, González-Chávez MCA, Hernández RMS (2013) Potential of castor bean (Ricinus communis L.) for phytoremediation of mine tailings and oil production. J Environ Manage 114:316–323
Onrubia M, Cusido RM, Ramirez K, Hernandez-Vazquez L, Moyano E, Bonfill M, Palazon J (2013) Bioprocessing of plant in vitro systems for the mass production of pharmaceutically important metabolites: paclitaxel and its derivatives. Curr Med Chem 20(7):880–891
Patel MK, Joshi M, Mishra A, Jha B (2015) Ectopic expression of SbNHX1 gene in transgenic castor (Ricinus communis L.) enhances salt stress by modulating physiological process. Plant Cell Tissue Organ Cult 122(2):477–490
Satsangi A, Mohan Ram HY (1965) A continuously growing tissue culture from the mature endosperm of Ricinus communis L. Phytomorph 15:26–30
Schaeffer GW, Sharpe FT (1997) Free and bound amino acids and proteins in developing grains of rice with enhanced lysine/proteins. Theor Appl Genet (Tag) 94(6):878–881
Sekomo CB, Nkurang E, Rousseau DP, Lens PN (2011) Fate of heavy metals in an urban natural wetland: the Nyabugogo Swamp (Rwanda). Water Air Soil Poll 214:321–333
Shaik S, Singh N, Nicholas A (2011) HPLC and GC analyses of in vitro-grown leaves of the cancer bush Lessertia (Sutherlandia) frutescens L. reveal higher yields of bioactive compounds. Plant Cell Tissue Organ Cult 105:431–438
Shakoor MB, Ali S, Farid M, Farooq MA, Tauqeer HM, Iftikhar U, Hannan F, Bharwana SA (2013) Heavy metal pollution, a global problem and its remediation by chemically enhanced phytoremediation: a review. J Biol Environ Sci 3(3):12–20
Sharma SS, Dietz KJ (2006) The significance of amino acids and amino acid-derived molecules in plant responses and adaptation to heavy metal stress. J Exp Bot 57:711–726
Shi J, Wu B, Yuan X, Cao Y, Chen X, Chen Y, Hu T (2008) An X-ray absorption spectroscopy investigation of speciation and biotransformation of copper in Elsholtzia splendens. Plant Soil 302(1):163–174
Sujatha M, Reddy TP, Mahasi MJ (2008) Role of biotechnological interventions in the improvement of castor (Ricinus communis L.) and Jatropha curcas L. Biotechnol Adv 26(5):424–435
Šukalović VHT, Vuletić M, Veljović-Jovanović S, Vučinića Z (2010) The effects of manganese and copper in vitro and in vivo on peroxidase catalytic cycles. J Plant Physiol 167:1550–1557
Sun C, Liu J, Wang Y, Sun L, Yu H (2013) Multivariate and geostatistical analyses of the spatial distribution and sources of heavy metals in agricultural soil in Dehui, Northeast China. Chemosphere 92:517–523
Tang SR (2000) Distribution of heavy metal in leaf extractive of Elsholtzia splendens and Commelina suffruticosa. Plant Physiol Commu 32(2):128–129
Weng SH (2010) Fourier transform infrared spectrum analysis. Chemical Industry Press, Beijing
Xiong ZT, Liu C, Geng B (2006) Phytotoxic effects of copper on nitrogen metabolism and plant growth in Brassica pekinensis Rupr. Ecotox. Environ Safe 64(3):273–280
Yang J, Chen Z, Wu S, Cui Y, Zhang L, Dong H, Yang C, Li C (2015) Overexpression of the Tamarix hispida ThMT3 gene increases copper tolerance and adventitious root induction in Salix matsudana Koidz. Plant Cell Tissue Organ Cult 121(2):469–479
Yruela I (2005) Copper in plants. Braz J Plant Physiol 17(1):145–146
Yruela I (2009) Copper in plants: acquisition, transport and interactions. Funct Plant Biol 36(5):409–430
Zenk MH (1996) Heavy metal detoxification in higher plants. Gene 179:21–30
Zlenko VA, Kotikov IV, Troshin LP (2005) Effects of IAA and BA on development of globular, heart- and torpedo-stage embryos from cell suspensions of three grape genotypes. Sci Hortic Amsterdam 104(3):237–247
Zuzarte MR, Dinis AM, Cavaleiro C, Salgueiro LR, Canhoto JM (2010) Trichomes, essential oils and in vitro propagation of Lavandula pedunculata (Lamiaceae). Ind Crop Prod 32:580–587
Acknowledgments
This project was financially supported by National Natural Science Foundation of China (41371470), National High Technology Research and Development Program (863 project) (2012AA101402) and the fundamental research funds for the central universities (2012MBDX005). The authors also want to acknowledge Professor Zdenko Rengel (The University of Western Australia) and Professor Ping Liu (Huazhong Agricultural University) for their help in paper’s languages modification.
Author contributions
Guoyong Huang and Yong Jin designed the experiment and performed all analysis, data interpretation and literature search; Guoyong Huang wrote and revised the manuscript; Wei Kang, Yonghong Liu, and Tao Zou gave some good suggestions on the experiments and participated in the part of experiments; Jin Zheng, Hongqing Hu participated in the experimental design and in the discussion of the manuscript.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Huang, G., Jin, Y., Zheng, J. et al. Accumulation and distribution of copper in castor bean (Ricinus communis L.) callus cultures: in vitro. Plant Cell Tiss Organ Cult 128, 177–186 (2017). https://doi.org/10.1007/s11240-016-1097-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11240-016-1097-z