Abstract
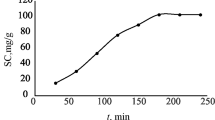
Regularities of gold sorption by the impregnated type sorbents based on a 1 M solution of benzo-15-crown-5 in nitrobenzene and benzo-15-crown-5 (30%, without diluent) were studied. The sorption isotherms were constructed, and the sorption and desorption kinetics and sorption dynamics were studied. The time of reaching sorption equilibrium, maximum capacity of the sorbents, and adsorption constants were determined. The data obtained on the time dependence of the sorption parameters are described by the pseudo-second-order kinetic curve. The conditions of gold desorption were found. Gold is quantitatively desorbed from the sorbents by a 2 M solution of ammonium thiocyanate. The sorption and desorption of gold under the dynamic conditions were studied. The dynamic and total exchange capacities of the sorbents were determined. The sorbents studied efficiently sorb gold from hydrochloric acid solutions.
Similar content being viewed by others
References
L. S. Atabekyan, V. G. Avakyan, A. K. Chibisov, S. P. Gromov, S. Z. Vatsadze, V. N. Nuriev, A. V. Medved’ko, Russ. Chem. Bull., 2019, 68, 2053; DOI: https://doi.org/10.1007/s11172-019-2666-1.
N. F. Goldshleger, M. A. Lapshina, V. E. Baulin, A. A. Shiryaev, Y. G. Gorbunova, A. Y. Tsivadze, Russ. Chem. Bull., 2020, 69, 1223; DOI: https://doi.org/10.1007/s11172-020-2893-5.
I. I. Dovhyi, D. A. Kremenchutskii, O. N. Kozlovskaia, N. A. Bezhin, V. V. Milyutin, E. A. Kozlitin, Phys. Oceanol., 2020, 36, 387.
Ž. Grahek, S. Dulanská, G. Karanović, I. Coha, I. Tucaković, M. Nodilo, E. Mátel, J. Environ. Radioact., 2018, 181, 18.
A. Zhang, C. Xiao, Y. Liu, Q. Hu, C. Chen, E. Kuraoka, J. Porous Mater., 2010, 17, 153.
V. V. Shapovalov, V. V. Kanygin, A. G. Naryshkin, N. A. Nerozin, E. Ya. Smetanin, Radiochem., 2006, 48, 53.
S. R. Izatt, J. S. McKenzie, N. E. Izatt, R. L. Bruening, K. E. Krakowiak, R. M. Izatt, Molecular Recognition Technology: a Green Chemistry Process for Separation of Individual Rare Earth Metals, White Paper on Separation of Rare Earth Elements, American Fork, 2016, 12 pp.; https://www.semanticscholar.org/paper/MOLECULAR-RECOGNITION-TECHNOLOGY-%3A-A-GREEN-PROCESS-Izatt-McKenzie/477e71c5b2b5f8cc66d396235a810d996494be4d.
S. R. Izatt, J. S. McKenzie, R. L. Bruening, R. M. Izatt, N. E. Izatt, K. E. Krakowiak, in Metal Sustainability, Ed. R. M. Izatt, John Wiley & Sons, Ltd., Chichester, 2016, pp. 317–332.
H. Poorbaygi, A. Roozbahani, K. Moradi, A. A. Habibpanah, B. Pooremad, J. Radioanal. Nucl. Chem., 2021, 327, 985.
D. Choudhury, S. Lahiri, T. N. Nag, S. Sodaye, A. Bombard, J. Radioanal. Nucl. Chem., 2021, 327, 1299.
J. Lehto, X. Hou, Chemistry and Analysis of Radionuclides: Laboratory Techniques and Methodology, Wiley-VCH Verlag GmbH & Co. KGaA, Weinheim, 2011.
R. M. Pallares, R. J. Abergel, Nano Res., 2020, 13, 2887.
Y. Zhao, B. Pang, H. Luehmann, L. Detering, X. Yang, D. Sultan, S. Harpstrite, V. Sharma, C. S. Cutler, Y. Xia, Y. Liu, Adv. Healthc. Mater., 2016, 5, 928.
M. F. Attallah, M. A. Gizawy, H. A. Shamsel-Din, N. M. A. Mohamed, A. M. I. Ali, J. Radioanal. Nucl. Chem., 2020, 325, 447.
M. Akhter, A. Mushtaq, H. M. A. Karim, M. A. Khan, Radiochim. Acta, 1994, 64, 137.
M. Sadeghi, M. R. A. Rovais, N. Zandi, M. Moradi, K. Yousefi, Appl. Radiat. Isot., 2019, 154, 108877.
S. R. Izatt, R. L. Bruening, R. M. Izatt, J. K. Kujanpää, International Precious Metals Institute 41st Annual Conference (Orlando, June 10–13, 2017), International Precious Metals Institute, Orlando, 2017, pp. 280–294.
R. M. Izatt, S. R. Izatt, N. E. Izatt, K. E. Krakowiak, R. L. Bruening, L. Navarro, Green Chem., 2015, 17, 2236.
V. V. Yakshin, O. M. Vilkova, N. A. Tsarenko, B. N. Laskorin, Dokl. Akad. Nauk, 1992, 323, 334 [Dokl. Chem. (Engl. Transl.), 1992, 323] (in Russian).
V. V. Yakshin, O. M. Vilkova, N. A. Tsarenko, B. N. Laskorin, Dokl. Akad. Nauk, 1992, 324, 144 [Dokl. Chem. (Engl. Transl.), 1992, 324] (in Russian).
V. V. Yakshin, Sb. tez. dokl. uchastnikov II Mezhdunar. foruma po nanotekhnologiyam “Rusnanotech 09” [Abstracts of Participants of II International Forum on Nanotechnologies “Rusnanotech 09”] (Moscow, October 06–08, 2009), State Corporation “Russian Corporation of Nanotechnologies,” Moscow, 2009, pp. 533–534 (in Russian).
V. V. Yakshin, O. M. Vilkova, N. A. Tsarenko, A. Yu. Tsivadze, Dokl. Chem., 2010, 430, 32.
V. V. Yakshin, O. M. Vilkova, S. M. Pluzhnik-Gladyr’, S. A. Kotlyar, Makrogeterotsikly [Macroheterocycles], 2010, 3, 114 (in Russia).
V. V. Yakshin, O. M. Vilkova, S. A. Kotlyar, A. Yu. Tsivadze, Russ. J. Coord. Chem., 2012, 38, 295.
V. V. Yakshin, O. M. Vilkova, N. A. Tsarenko, A. Yu. Tsivadze, Russ. Chem. Bull., 2012, 61, 656; DOI: https://doi.org/10.1007/s11172-012-0095-5.
A. A. Vydysh, I. I. Dovhyi, A. Yu. Lyapunov, Uch. zap. Krymskogo federal’nogo universiteta im. V. I. Vernadskogo. Ser. “Biologiya, khimiya” [Scientific Writings of V. I. Vernadsky Crimean Federal University. Ser. Biol. Chem.], 2015, 1, 197 (in Russian).
A. A. Vydysh, I. I. Dovhyi, N. A. Bezhin, Russ. Chem. Bull., 2018, 67, 2275; DOI: https://doi.org/10.1007/s11172-018-2370-6.
Pat. RF 2699631; Byul. Izobret. [Invention Bulletin], 2019, 25, 2 pp. (in Russian).
V. S. Yankovskaya, I. I. Dovhyi, V. V. Milyutin, N. A. Nekrasova, N. A. Bezhin, A. Yu. Lyapunov, J. Radioanal. Nucl. Chem., 2017, 314, 119.
V. S. Yankovskaya, I. I. Dovhyi, N. A. Bezhin, V. V. Milyutin, N. A. Nekrasova, S. V. Kapranov, V. F. Shulgin, J. Radioanal. Nucl. Chem., 2018, 318, 1085.
M. F. Attallah, E. H. Borai, K. F. Allan, Radiochem., 2009, 51, 622.
K. Periasamy, C. Namasivayam, Ind. Eng. Chem. Res., 1994, 33, 317.
Y. S. Ho, G. McKay, Process Biochem., 1999, 34, 451.
V. C. Taty-Costades, H. Fauduest, C. Porte, A. Delacroix, J. Hazard. Mater., 2003, 105, 121.
Funding
This work was carried out in terms of state assignment of the Ministry of Science and Higher Education of the Russian Federation (theme “Oceanologic Processes” No. 0555-2021-0004).
Author information
Authors and Affiliations
Corresponding author
Additional information
Published in Russian in Izvestiya Akademii Nauk. Seriya Khimicheskaya, No. 2, pp. 254–259, February, 2022.
No human or animal subjects were used in this research.
The authors declare no competing interests.
Rights and permissions
About this article
Cite this article
Dovhyi, I.I., Bezhin, N.A. Sorption of gold(iii) from hydrochloric acid solutions by the sorbents based on benzo-15-crown-5. Russ Chem Bull 71, 254–259 (2022). https://doi.org/10.1007/s11172-022-3405-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11172-022-3405-6