Abstract
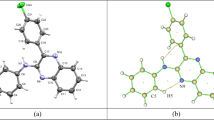
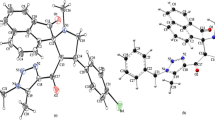
Hydrogen bonds and structure of drug Xymedone in different phase and aggregate states were studied using IR and Raman spectroscopy, IR microscopy, quantum chemical calculations (B3LYP/6-31G*), X-ray powder diffractometry, and DSC and TGA methods. It was found that the synthesized specimen of Xymedone powder is a mixture of two gauche-conformers. First one is bound by intermolecular hydrogen bonds and second one is stabilized by an intramolecular hydrogen bond. Upon the dissolution of Xymedone the conformational equilibrium shifted in favor of conformers of the first or second type depending on the solvent nature.
Similar content being viewed by others
References
S. G. Izmailov, G. A. Izmailov, M. Yu. Aver’yanov. V. S. Reznik, Ksimedon v klinicheskoi praktike [Xymedone in Clinical Practice], Izd-vo NGMA, Nizhnii Novgorod, 2001, 188 pp. (in Russian).
I. A. Litvinov, V. E. Kataev, A. T. H. Lenstra, H. J. Geise, Acta Crystallogr., 1992, C48, 1286.
V. S. Reznik, N. G. Pashkurov, Izv. Akad. Nauk SSSR, Ser. Khim., 1966, 1613 [Bull. Acad. Sci. USSR, Div. Chem. Sci. (Engl. Transl.), 1966, 15].
M. J. Frisch, G. W. Trucks, H. B. Schlegel, G. E. Scuseria, M. A. Robb, J. R. Cheeseman, J. A. Montgomery, T. Vreven, Jr., K. N. Kudin, J. C. Burant, J. M. Millam, S. S. Iyengar, J. Tomasi, V. Barone, B. Mennucci, M. Cossi, G. Scalmani, N. Rega, G. A. Petersson, H. Nakatsuji, M. Hada, M. Ehara, K. Toyota, R. Fukuda, J. Hasegawa, M. Ishida, T. Nakajima, Y. Honda, O. Kitao, H. Nakai, M. Klene, X. Li, J. E. Knox, H. P. Hratchian, J. B. Cross, C. Adamo, J. Jaramillo, R. Gomperts, R. E. Stratmann, O. Yazyev, A. J. Austin, R. Cammi, C. Pomelli, J. W. Ochterski, P. Y. Ayala, K. Morokuma, G. A. Voth, P. Salvador, J. J. Dannenberg, V. G. Zakrzewski, S. Dapprich, A. D. Daniels, M. C. Strain, O. Farkas, D. K. Malick, A. D. Rabuck, K. Raghavachari, J. B. Foresman, J. V. Ortiz, Q. Cui, A. G. Baboul, S. Clifford, J. Cioslowski, B. B. Stefanov, G. Liu, A. Liashenko, P. Piskorz, I. Komaromi, R. L. Martin, D. J. Fox, T. Keith, M. A. Al-Laham, C. Y. Peng, A. Nanayakkara, M. Challacombe, P. M. W. Gill, B. Johnson, W. Chen, M. W. Wong, C. Gonzalez, J. A. Pople, GAUSSIAN 03, Revision B.05, Gaussian, Inc., Wallingford (CT), 2004.
A. D. Becke, Phys. Rev., A, 1988, 38, 3098.
C. Lee, W. Yang, R. G. Parr, Phys. Rev., B, 1988, 37, 785.
M. V. Vol’kenshtein, Stroenie i fizicheskie svoistva molekul [Structures and Physical Properties of Molecules], Izd-vo Akad. Nauk SSSR, Moscow-Leningrad, 1955, 638 pp. (in Russian).
J. Baker, A. Jarzecki, P. Pulay, J. Phys. Chem. A, 1998, 102, 1412.
S. A. Katsyuba, E. E. Vandyukova, Chem. Phys. Lett., 2003, 377, 658.
V. A. Sipachev, J. Mol. Struct., 2001, 67, 567.
P. Popelier, A. T. H. Lenstra, C. Van Alsenoy, H. J. Geise, Struct. Chem., 1991, 2, 3.
N. B. Colthup, L. H. Daly, S. E. Wiberley, Introduction to Infrared and Raman Spectroscopy, Academic Press, New York-London, 1964, 512 pp.
M. D. Joesten, L. J. Schaad, Hydrogen Bonding, Marcel Dekker, Inc., New York, 1974, 622 pp.
Author information
Authors and Affiliations
Corresponding author
Additional information
Published in Russian in Izvestiya Akademii Nauk. Seriya Khimicheskaya, No. 6, pp. 1187–1193, June, 2012.
Rights and permissions
About this article
Cite this article
Zvereva, E.E., Vandyukova, I.I., Vandyukov, A.E. et al. IR and Raman spectra, hydrogen bonds, and conformations of N-(2-hydroxyethyl)-4,6-dimethyl-2-oxo-1,2-dihydropyrimidine (drug Xymedone). Russ Chem Bull 61, 1199–1206 (2012). https://doi.org/10.1007/s11172-012-0163-x
Received:
Revised:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11172-012-0163-x