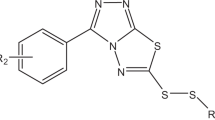
Abstract
We synthesized a number of derivatives of 1,2,3-benzotriazole, 1,2,4-triazole, and oxazolidine-2-one derivatives, among which compounds suppressing the growth of the tumor cells HeLa and H1299 were revealed. The studies of the effect of these compounds on the cell cycle and activity of caspase dependent degradation of poly(ADP-ribose) polymerase showed that these compounds at the IC50 doses exhibit cytostatic effect on tumor cells. In the MCF7 cells, these compounds induce an increase in expression of the tumor suppressor p53.
Similar content being viewed by others
References
E. Margoliash, A. Novogrodsky, Biochem. J., 1958, 68, 468.
J. V. Anjos, R. A. Neves Filho, S. C. Nascimento, R. M. Srivastava, S. J. de Melo, D. Sinou, Eur. J. Med. Chem., 2009, 44, 3571.
S. J. Yan, Y. J. Liu, Y. L. Chen, L. Liu, J. Lin, Bioorg. Med. Chem. Lett., 2010, 20, 5225.
P. C. Chen, V. Patil, W. Guerrant, P. Green, A. K. Oyeler, Bioorg. Med. Chem., 2008, 16, 4839.
L. B. Peterson, B. S. Blagg, Bioorg. Med. Chem. Lett., 2010, 20, 3957.
Y. Xia, Y. Liu, J. Wan, M. Wang, P. Rocchi, F. Qu, J. L. Iovanna, L. Peng, J. Med. Chem., 2009, 52, 6083.
K. S. Bhat, B. Poojary, D. J. Prasad, P. Naik, B. S. Holla, Eur. J. Med. Chem., 2009, 44, 5066.
M. Loiarro, S. Campo, B. Arseni, S. Rossi, V. D’Alessio, R. De Santis, C. Sette, V. Ruggiero, Prostate, 2011, 71, 32.
B. Yang, Q. J. He, D. Y. Zhu, Y. J. Lou, R. Y. Fang, Cancer Chemother. Pharmacol., 2006, 57, 268.
N. A. Al-Masoudi, B. H. Abdullah, A. H. Essa, R. Loddo, P. LaColla, Arch. Pharm., 2010, 343, 222.
J. P. Malerich, J. S. Lama, B. Hart, R. M. Fine, B. Klebansky, M. J. Tanga, A. D’Andrea, Bioor. Med. Chem. Lett., 2010, 20, 7454.
A. Kumar, I. Ahmad, B. S. Chhikara, R. Tiwari, D. Mandal, K. Parang, Bioorg. Med. Chem. Lett., 2011, 21, 1342.
S. K. De, J. L. Stebbins, L. H. Chen, M. Riel-Mehan, T. Machleidt, R. Dahl, H. Yuan, A. Emdadi, E. Barile, V. Chen, R. Murphy, M. Pellecchia, J. Med. Chem., 2009, 52, 1943.
Q. Zhang, Y. Peng, X. I. Wang, S. M. Keenan, S. Arora, W. J. Welsh, J. Med. Chem., 2007, 50, 749.
S. Arora, X. I. Wang, S. M. Keenan, C. Andaya, Q. Zhang, Y. Peng, W. I. Welsh, Cancer Res., 2009, 69, 1910.
B. Das, A. V. Rajarao, S. Rudra, A. Yadav, A. Ray, M. Pandya, A. Rattan, A. Mehta, Bioorg. Med. Chem. Lett., 2009, 19, 6424.
G. Zappia, P. Menendez, G. D. Monache, D. Misiti, L. Nevola, B. Botta, Mini Rev. Med. Chem., 2007, 7, 389.
L. C. Dias, P. R. Meira, J. Org. Chem., 2005, 70, 4762.
A. Patenaude, R. G. Deschesnes, J. L. C. Rousseau, E. Petitclerc, J. Lacroix, M.-F. Côté, R. C. Gaudreault, Cancer Res., 2007, 67, 2304.
T. Shibue, T. Hirai, I. Okamoto, N. Morita, H. Masu, I. Azumaya, O. Tamura, Chem. Eur. J., 2010, 16, 11678.
K. K. Chan, P. S. Hong, K. Tutsch, D. L. Trump, Cancer Res., 1994, 54, 6421.
J. H. Burkhalter, V. C. Stephens, L. A. R. Hall, J. Am. Chem. Soc., 1952, 74, 3868.
US Pat. 3.113.932; Chem. Abstr., 1964, 61, 7020.
L. I. Bagal, M. S. Pevzner, A. N. Frolov, N. I. Sheludyakova, Khim. Getetrotsikl. Soedin., 1970, 259 [Chem. Heterocycl. Compd. (Engl. Transl.), 1970, 6, 240].
O. H. Lowry, N. J. Rosenbrough, A. L. Farr, R. J. Randall, J. Biol. Chem., 1951, 193, 265.
S. H. Kaufmann, S. Desnoyers, Y. Ottaviano, N. E. Davidson, G. G. Poirier, Cancer Res., 1993, 53, 3976.
P. M. Chumakov, Usp. Biolog. Khim., 2007, 47, 3 [Biochemistry (Moscow) (Engl. Transl.), 2007, 72, No. 13, 1399].
A. O. Zheltukhin, P. M. Chumakov, Usp. Biolog. Khim., 2010, 50, 447 [Biochemistry (Moscow) (Engl. Transl.), 2010, 75, No. 13, 1692].
J. Wesierska-Gadek, D. Schloffer, V. Kotala, M. Horky, Int. J. Cancer, 2002, 101, 128.
T. Mitsudomi, S. M. Steinberg, M. M. Nau, D. Carbone, D. D’Amico, S. Bodner, H. K. Oie, R. L. Linnoila, J. L. Mulshine, J. D. Minna, A. F. Gazdar, Oncogene, 1992, 7, 171.
Author information
Authors and Affiliations
Corresponding author
Additional information
Published in Russian in Izvestiya Akademii Nauk. Seriya Khimicheskaya, No. 6, pp. 1141–1145, June, 2011.
Rights and permissions
About this article
Cite this article
Derevkova, V.A., Balalaeva, I.V., Papina, R.I. et al. Cytostatic action of triazole and oxazolidinone derivatives. Russ Chem Bull 60, 1166–1171 (2011). https://doi.org/10.1007/s11172-011-0183-y
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11172-011-0183-y