Abstract
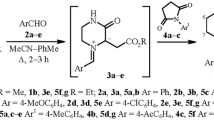
A reaction of aryl ketenes with 1-arylmethylidenepyrazolidin-1-azomethine imines, generated by the diaziridine ring opening in 6-aryl-1,5-diazabicyclo[3.1.0]hexanes catalyzed with Et2O·BF3, leads to 1,2-bis(phenylacetyl)pyrazolidine, 2-arylacetyl-1-arylidenepyrazolidin-1-ium chlorides, or a representative of 1,5-diazabicyclo[3.3.0]octan-2-ones, viz., 4-(4-eth-oxyphenyl)-3,3-diphenyl-1,5-diazabicyclo[3.3.0]octan-2-one, depending on the reaction conditions and the structure of the starting compounds. A mechanism suggested earlier for the transformation of 1,5-diazabicyclo[3.1.0]hexanes in the reaction with ketenes was confirmed.
Similar content being viewed by others
References
R. Grashey, Azomethine imines, in 1,3-Dipolar cycloaddition chemistry, Ed. A. Padwa, Wiley, New York, V. 1, Chapter 7, 1984, P. 733–817.
L. L. Rodina, A. B. Khalikova, O. A. Verzhba, I. K. Korobit-sina, in Sovremennye problemy organicheskoi khimii [Modern Problems of Organic Chemistry], Ed. K. A. Ogloblin, Leningrad Univ. Publ., 1986, Issue. 8, p. 135–171 (in Russian).
R. Huisgen, R. Grashey, J. Sauer, in The Chemistry of Alkenes, Ed. S. Patai, Interscience, London, 1964, 779.
G. Bianchi, De Micheli, R. Candolfi, in The Chemistry of Double-bonded Functional Groups, Ed. S. Patai, Wiley, New York, Part 1, Supplement A, 1977, 369.
L.N. Jungheim, S.K. Sigmund, J. Org. Chem., 1987, 52, 4007.
J. Svete, Arkivoc, 2006 (vii), 35–56.
R. C. F. Jones, S. J. Hollis, J. N. Iley, Arkivoc, 2007 (v), 152–166.
H. Dorn, A. Otto, Chem. Ber., 1968, 101, 3287.
H. Dorn, A. Otto, Angew. Chem., Int. Ed. Engl., 1968, 7, 214.
Yu. B. Koptelov, M. Kh. Kim, A. P. Molchanov, R. R. Kostikov, Zh. Org. Khim., 1999, 35, 116 [Russ. J. Org. Chem. (Engl. Transl.), 1999, 35, 110].
A. P. Molchanov, D. I. Sipkin, Yu. V. Koptelov, R. R. Kostikov, SYNLETT, 2000, 1779.
A. P. Molchanov, D. I. Sipkin, Yu. B. Koptelov, R. R. Kostikov, Zh. Org. Khim., 2001, 37, 888 [Russ. J. Org. Chem. (Engl. Transl.), 2001, 37, 841].
A. P. Molchanov, D. I. Sipkin, Yu. V. Koptelov, R. R. Kostikov, Eur. J. Org. Chem., 2002, 453.
Yu. B. Koptelov, S. P. Saik, Zh. Org. Khim., 2006, 42, 1515 [Russ. J. Org. Chem. (Engl. Transl.), 2006, 42, 1501].
Yu. B. Koptelov, Zh. Org. Khim., 2006, 42, 1524 [Russ. J. Org. Chem. (Engl. Transl.), 2006, 42, 1510].
Yu. S. Syroeshkina, V. V. Kuznetsov, K. A. Lyssenko, N. N. Makhova, Mendeleev Commun., 2008, 18, 42.
Yu. S. Syroeshkina, V. V. Kuznetsov, M. I. Struchkova, M. A. Epishina, N. N. Makhova, Mendeleev Commun., 2008, 18, 207.
Yu. S. Syroeshkina, V. V. Kuznetsov, K. A. Lyssenko, N. N. Makhova, Izv. Akad. Nauk, Ser. Khim., 2009, 362 [Russ. Chem. Bull., Int. Ed., 2009, 58, 366].
Yu. S. Syroeshkina, V. V. Kachala, I. V. Ovchinnikov, V. V. Kuznetsov, Yu. V. Nelyubina, K. A. Lyssenko, N. N. Makhova, Mendeleev Commun., 2009, 19, 276.
A. V. Shevtsov, V. V. Kuznetsov, A. A. Kislukhin, V. Yu. Petukhova, Yu. A. Strelenko, N. N. Makhova, J. Heterocyclic Chem., 2006, 43, 881.
S. Knapp, B. H. Toby, M. Sebastian, K. Krogh-Jespersen, J. A. Potenza, J. Org. Chem., 1981, 46, 2490.
C. Didierjean, A. Aubry, M. Zouikri, G. Boussard, M. Marraud, Acta Crystallogr. C, 1995, 51, 688.
A. Lecoq, G. Boussard, M. Marraud, A. Aubry, Biopolymers, 1993, 33, 1051.
A. Luttringhaus, J. Jander, R. Schneider, Chem. Ber., 1959, 92, 1756.
S. D′Andrea, Z. B. Zheng, K. DenBleyker, J. C. Fung-Tomc, H. Yang, J. Clark, D. Taylor, J. Bronson, Bioorg. Med. Chem. Lett., 2005, 15, 2834.
S. V. Usachev, G. A. Nikiforov, K. A. Lyssenko, Yu. V. Nelubina, P. A. Levkin, R. G. Kostyanovsky, Tettrahedron Asym., 2007, 18, 1540.
G. A. Olah, A. E. Pavlath, Ju. A. Olah, F. Herr, J. Org. Chem., 1957, 22, 879.
L. C. Raiford, J. N. Wickert, J. Am. Chem. Soc., 1931, 53, 3143.
V. V. Kuznetsov, S. A. Kutepov, N. N. Makhova, K. A. Lysenko, D. E. Dmitriev, Izv. Akad. Nauk, Ser. Khim., 2003, 638 [Russ. Chem. Bull., Int. Ed., 2003, 52, 665].
Author information
Authors and Affiliations
Corresponding author
Additional information
Published in Russian in Izyestiya Akademii Nauk. Seriya Khimicheskaya, No. 7, pp. 1401–1408, July, 2010.
Rights and permissions
About this article
Cite this article
Syroeshkina, Y.S., Petukhova, V.Y., Kachala, V.V. et al. Reaction of 1-arylmethylidenepyrazolidin-1-azomethine imines with aryl ketenes. Russ Chem Bull 59, 1433–1441 (2010). https://doi.org/10.1007/s11172-010-0259-0
Received:
Revised:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11172-010-0259-0
- Key words
- 6-aryl-1,5-diazabicyclo[3.1.0]hexanes
- 1-arylmethylidenepyrazolidin-1-azomethine imines
- ring expansion
- ionic liquids
- BF3·Et2O
- aryl ketenes
- 1,2-bis(phenyl-acetyl)pyrazolidine
- 2-arylacetyl-1-arylidenepyrazolidin-1-ium chlorides
- 1-(phenylacetyl)-pyrazolidine
- 1-(phenylacetyl)-4,5-dihydro-1H-pyrazole, 2,2-diphenyl-3-(4-ethoxyphenyl)-tetrahydro-1H,5H-pyrazolo[1,2-a]pyrazol-1-one