Abstract
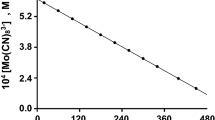
The product of the reaction of valeraldehyde with chlorine dioxide was determined, and the solvent effect on the reaction kinetics was studied. The major oxidation product is valeric acid. The reaction rate is described by the second-order equation w = k[RCHO]·[ClO2]. The rate constants were measured in the 297–328 K interval, and the activation parameters of the reaction were determined.
Similar content being viewed by others
References
J. Otto, K. Paluch, Roczniki Chem., 1965, 39, 1711.
R. A. Somsen, Tappi, 1960, 43, 154.
Y. R. Kastner, K. S. Das, C. Hu, R. J. McClendon, J. Air and Waste management association, 2003, 53, 1218.
T. A. Tumanova, Fiziko-khimicheskie osnovy otbelki tsellyulozy, Lesnaya Prom-st’ [Physicochemical Foundations of Cellulose Bleaching], Moscow, 1984, 213 pp. (in Russian).
A. J. Gordon, R. A. Ford, A Handbook of Practical Data, Techniquies and References, Wiley, New York, 1972.
C. Reichardt, Solvents and solvents effects in organic chemistry, Weinheim, VCH Verlagsgesellschaft mbH, 1988, 763 pp.
Handbook of Solvents, Ed. G. Wypych, ChemTec Publishing, Toronto, 2001, 1675 pp.
R. Kopitzky, H. Grothe, H. Willner, Chem. Eur. J., 2002, 8, 5601.
Author information
Authors and Affiliations
Corresponding author
Additional information
Published in Russian in Izvestiya Akademii Nauk. Seriya Khimicheskaya, No. 11, pp. 2287–2289, November, 2008.
Rights and permissions
About this article
Cite this article
Ganieva, E.S., Ganiev, I.M., Grabovskiy, S.A. et al. Oxidation of valeraldehyde by chlorine dioxide. Russ Chem Bull 57, 2332–2334 (2008). https://doi.org/10.1007/s11172-008-0331-1
Received:
Revised:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11172-008-0331-1