Abstract
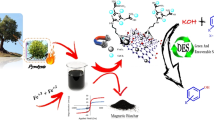
In this study, a magnetic nitrogen-doped carbon-based copper (MNC-Cu) catalyst was fabricated so that natural silk cocoons undergo thermal processes and then activate by combining with Fe3O4 MNPs as a suitable substrate for placement copper metal. The efficiency of the generated catalyst in the synthesis of 5-substituted 1H-tetrazole derivatives was evaluated by the [3 + 2] cycloaddition reaction of aromatic aldehydes, azide ions, and hydroxylamines. FT-IR, FE-SEM, EDS, TEM, XRD, TGA, and VSM techniques have been adopted to identify and validate this heterogeneous catalyst. The observation from EDS, elemental mapping, XRD, and FT-IR analysis confirms the immobilization of Cu metals on the MNC surface and uniform distribution of species and then no aggregation occurs during functionalization. VSM results show the magnetic feature of fabricated catalyst, and based on the leaching test, the amount of catalyst leaching was negligible. Moreover, this reaction is a one-pot multicomponent reaction (MCRs) or more precisely a one-pot, three-component reaction, which is one of the advantages of this work. The fabricated catalyst shows high efficiency, good stability, and considerable reactivity in synthesizing 5-substituted 1H-tetrazole derivatives. So it can be seen that the fabricated magnetic catalyst is a useful and practical catalyst for synthesizing [3 + 2] cycloaddition reactions. Reusability and recyclability for five sequential runs without notable increase and reduction in activity are other advantages. In addition, the magnetic properties of this heterogeneous catalyst led to easy and quickly recovered and striking copper leaching.
Graphical abstract














Similar content being viewed by others
Availability of data and material
My manuscript has data included as supplementary electronic material.
References
S. Gopi, K. Giribabu, M. Kathiresan, ACS Omega 3, 6251 (2018)
B. Karimi, H. Barzegar, H. Vali, Chem. Commun. 54, 7155 (2018)
B. Sakintuna, Y. Yürüm, Ind. Eng. Chem. Res. 44, 2893 (2005)
F. Bai, Y. Xia, B. Chen, H. Su, Y. Zhu, Carbon N. Y. 79, 213 (2014)
Z. Lan, Y. Fang, Y. Zhang, X. Wang, Angew. Chemie 130, 479 (2018)
J. Lee, J. Kim, T. Hyeon, Adv. Mater. 18, 2073 (2006)
M.J. Bojdys, J. Jeromenok, A. Thomas, M. Antonietti, Adv. Mater. 22, 2202 (2010)
R. Wang, K. Wang, Z. Wang, H. Song, H. Wang, S. Ji, J. Power Sources 297, 295 (2015)
X.-H. Li, M. Antonietti, Chem. Soc. Rev. 42, 6593 (2013)
X. Chen, Y.S. Jun, Chem. Mater 21, 150 (2009)
S.Y. Cho, Y.S. Yun, S. Lee, D. Jang, K.-Y. Park, J.K. Kim, B.H. Kim, K. Kang, D.L. Kaplan, H.-J. Jin, Nat. Commun. 6, 1 (2015)
P. Zhang, Y. Gong, H. Li, Z. Chen, Y. Wang, Nat. Commun. 4, 1 (2013)
P.E. Ingham, J. Appl. Polym. Sci. 15, 3025 (1971)
M.-M. Titirici, M. Antonietti, Chem. Soc. Rev. 39, 103 (2010)
H.-S. Kim, S.H. Yoon, S.-M. Kwon, H.-J. Jin, Biomacromol 10, 82 (2009)
R. Nazarov, H.-J. Jin, D.L. Kaplan, Biomacromol 5, 718 (2004)
C. Fu, Z. Shao, and V. Fritz, Chem. Commun. 45, 6515 (2009)
H.-W. Liang, S. Brüller, R. Dong, J. Zhang, X. Feng, K. Müllen, Nat. Commun. 6, 1 (2015)
H.H. Kim, M.K. Kim, K.H. Lee, Y.H. Park, I.C. Um, Int. J. Biol. Macromol. 79, 943 (2015)
D. Lardizábal-G, Y. Verde-Gómez, I. Alonso-Lemus, A. Aguilar-Elguezabal, Int. J. Hydrogen Energy 40, 17300 (2015)
Z. Heidarinejad, M.H. Dehghani, M. Heidari, G. Javedan, I. Ali, M. Sillanpää, Environ. Chem. Lett. 18, 393 (2020)
H. Tounsadi, A. Khalidi, M. Farnane, M. Abdennouri, N. Barka, Process Saf. Environ. Prot. 102, 710 (2016)
Z. Hezarkhani, A. Shaabani, Appl. Organomet. Chem. 31, e3624 (2017)
Y. Zhu, W. Sun, J. Luo, W. Chen, T. Cao, L. Zheng, J. Dong, J. Zhang, M. Zhang, Y. Han, Nat. Commun. 9, 1 (2018)
J. Wang, S. Kaskel, J. Mater. Chem. 22, 23710 (2012)
M. Ma, Q. Zhang, D. Yin, J. Dou, H. Zhang, H. Xu, Catal. Commun. 17, 168 (2012)
M.A. Bodaghifard, J. Organomet. Chem. 886, 57 (2019)
Q. Du, W. Zhang, H. Ma, J. Zheng, B. Zhou, Y. Li, Tetrahedron 68, 3577 (2012)
A. Marandi, N. Koukabi, Colloids Surf. A Physicochem. Eng. Asp. 621, 126597 (2021)
A. Marandi, E. Nasiri, N. Koukabi, F. Seidi, Int. J. Biol. Macromol. 190, 61 (2021)
C.W. Lim, I.S. Lee, Nano Today 5, 412 (2010)
P. Akbarzadeh, N. Koukabi, Appl. Organomet. Chem. 34, e5395 (2020)
P. Akbarzadeh, N. Koukabi, Appl. Organomet. Chem. 35, e6039 (2021)
A. Marandi, N. Koukabi, M.A. Zolfigol, Res. Chem. Intermed. 47, 3145 (2021)
P. Akbarzadeh, N. Koukabi, E. Kolvari, Mol. Divers. 24(2), 319 (2019)
P. Akbarzadeh, N. Koukabi, E. Kolvari, Res. Chem. Intermed. 45, 1009 (2019)
M. Esmaeilpour, A.R. Sardarian, H. Firouzabadi, Appl. Organomet. Chem. 32, e4300 (2018)
M. Abdollahi-Alibeik, A. Moaddeli, New J. Chem. 39, 2116 (2015)
M.M. Heravi, A. Fazeli, H.A. Oskooie, Y.S. Beheshtiha, H. Valizadeh, Synlett 23, 2927 (2012)
M. Sridhar, K.K.R. Mallu, R. Jillella, K.R. Godala, C.R. Beeram, N. Chinthala, Synthesis (Stuttg). 45, 507 (2013)
S. Behrouz, J. Saudi Chem. Soc. 21, 220 (2017)
J. Sun, Z. Zhang, J. Ji, M. Dou, F. Wang, Appl. Surf. Sci. 405, 372 (2017)
T. Altun, E. Pehlivan, Food Chem. 132, 693 (2012)
V. Sahu, S. Grover, B. Tulachan, M. Sharma, G. Srivastava, M. Roy, M. Saxena, N. Sethy, K. Bhargava, D. Philip, Electrochim. Acta 160, 244 (2015)
K.M. Khan, I. Fatima, S.M. Saad, M. Taha, W. Voelter, Tetrahedron Lett. 57, 523 (2016)
B. Mitra, S. Mukherjee, G.C. Pariyar, P. Ghosh, Tetrahedron Lett. 59, 1385 (2018)
U.B. Patil, K.R. Kumthekar, J.M. Nagarkar, Tetrahedron Lett. 53, 3706 (2012)
S.D. Guggilapu, S.K. Prajapti, A. Nagarsenkar, K.K. Gupta, B.N. Babu, Synlett 27, 1241 (2016)
Acknowledgements
The authors sincerely acknowledge the Research Council of Semnan University for supporting this work.
Funding
Semnan University, Grant Recipient: Dr. Nadiya Koukabi, Associated Professor.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
All authors are aware of the submission and agree to its publication and have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Tahmasbi, M., Akbarzadeh, P. & Koukabi, N. Magnetic nitrogen-doped carbon derived from silk cocoon biomass: a promising and sustainable support for copper. Res Chem Intermed 48, 1383–1401 (2022). https://doi.org/10.1007/s11164-021-04623-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11164-021-04623-3