Abstract
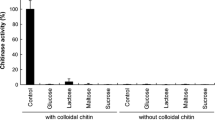
Microbial chitinase has received great attention due to its medical, biological, and agricultural applications. In this study, over 50 bacterial strains were isolated from Taiwanese soils using medium containing squid pen powder (SPP) as the sole source of carbon/nitrogen. Based on chitinolytic activity, Streptomyces thermocarboxydus TKU045 was selected for further study. Optimized culture conditions revealed that S. thermocarboxydus TKU045 could produce the highest chitinase activity (52.985 U/mL) when cultured in a medium containing 1% (w/v) SPP at 45 °C for 36 h. Characterized TKU045 chitinase showed novel properties with a smallest molecular weight (12.8 kDa by sodium dodecyl sulfate–polyacrylamide gel electrophoresis analysis) and more acidic optimal pH (pH 4) than those of other Streptomyces chitinases. A combination of high-performance liquid chromatography and matrix-assisted laser desorption ionization time of flight mass spectrometer data revealed that chitin oligomers (COS) obtained from the hydrolysis of colloidal chitin by TKU045 chitinase comprise oligomers with multiple degrees of polymerization (DP) varying from 1 to 7. The COS with low DP exhibited enhanced 2,2-diphenyl-1-picrylhydrazyl radical scavenging capability and promoted the growth of Lactobacillus lactis. Taken together, the COS obtained by hydrolyzing colloidal chitin with TKU045 chitinase could have the potential to be used in medicine or nutraceuticals due to its active anti-oxidant and prebiotic contents.







Similar content being viewed by others
References
S.L. Wang, T.W. Liang, Res. Chem. Interm. 43, 3445 (2017)
G. Akca, A. Özdemir, Z.G. Öner, S. Şenel, Res. Chem. Interm. 44, 4811 (2018)
F. Ding, H. Li, Y. Du, X. Shi, Res. Chem. Interm. 44, 4827 (2018)
M.M. Jaworska, A. Górak, Res. Chem. Interm. 44, 4841 (2018)
L.C. Tsai, M.L. Tsai, K.Y. Lu, F.L. Mi, Res. Chem. Interm. 44, 4855 (2018)
A. Mohandas, W. Sun, T.R. Nimal, S.A. Shankarappa, N.S. Hwang, R. Jayakumar, Res. Chem. Interm. 44, 4873 (2018)
P. Hiranpattanakul, T. Jongjitpissamai, S. Aungwerojanawit, W. Tachaboonyakiat, Res. Chem. Interm. 44, 4913 (2018)
D. Kotatha, K. Morishima, S. Uchida, M. Ogino, M. Ishikawa, T. Furuike, H. Tamura, Res. Chem. Interm. 44, 4971 (2018)
T.W. Liang, W.T. Chen, Z.H. Lin, Y.H. Kuo, A.D. Nguyen, P.S. Pan, S.L. Wang, Int. J. Mol. Sci. 17, 1302 (2016)
S. Sinha, S. Chand, P. Tripathi, Appl. Biochem. Biotechnol. 180, 883 (2016)
Q. Xiong, Y. Wei, H. Xie, Z. Feng, Y. Gan, C. Wang, M. Liu, F. Bai, F. Xie, G. Shao, Vaccine 32, 3445 (2014)
C. Villiers, M. Chevallet, H. Diemer, R. Couderc, H. Freitas, A. Van Dorsselaer, P.N. Marche, T. Rabilloud, Mol. Cell. Proteomics 8, 1252 (2009)
D.V. Gerasimenko, I.D. Avdienko, G.E. Bannikova, OYu. Zueva, V.P. Varlamov, Appl. Biochem. Micro. 40, 253 (2004)
S. Sinha, P. Tripathi, S. Chand, Appl. Biochem. Biotechnol. 167, 1029 (2012)
P.J. Park, J.Y. Je, S.K. Kim, Carbohydr. Polym. 55, 17 (2004)
W.J. Jung, R.D. Park, Mar Drugs 12, 5328 (2014)
M. Kuddus, I.Z. Ahmad, J. Gen. Eng. Biotechnol. 11, 39 (2013)
S.L. Wang, T.W. Liang, Y.H. Yen, Carbohydr. Polym. 84, 732 (2011)
S.L. Wang, H.T. Yu, M.H. Tsai, C.T. Doan, V.B. Nguyen, V.C. Do, A.D. Nguyen, Res. Chem. Interm. 44, 4903 (2018)
C.L. Wang, J.W. Su, T.W. Liang, A.D. Nguyen, S.L. Wang, Res. Chem. Interm. 40, 2237 (2014)
T.W. Liang, Y.Y. Chen, P.S. Pan, S.L. Wang, Int. J. Biol. Macromol. 63, 8 (2014)
T.W. Liang, T.Y. Shieh, S.L. Wang, Bioproc. Biosyst. Eng. 37, 1201 (2014)
S.L. Wang, C.P. Liu, T.W. Liang, Carbohydr. Polym. 90, 1305 (2012)
T.W. Liang, J.L. Hsieh, S.L. Wang, Carbohydr. Res. 362, 38 (2012)
S.L. Wang, P.C. Wu, T.W. Liang, Carbohydr. Res. 344, 979 (2009)
K.J. Kim, Y.J. Yang, J.G. Kim, J. Biochem. Mol. Biol. 36, 185 (2003)
Y. Han, B. Yang, F. Zhang, X. Miao, Z. Li, Mar. Biotechnol. 11, 132 (2009)
G.C. Pradeep, H.Y. Yoo, Y.H. Choi, J.C. Yoo, Appl. Biochem. Biotechnol. 175, 372 (2015)
M. Rabeeth, A. Anitha, G. Srikanth, Pak. J. Biol. Sci. 14, 788 (2011)
A. Nagpure, P.K. Gupta, J. Basic Microbiol. 53, 429 (2013)
M. Gangwar, V. Singh, A.K. Pandey, C.K. Tripathi, Indian J. Exp. Biol. 54, 64 (2016)
C.T. Doan, T.N. Tran, V.B. Nguyen, A.D. Nguyen, S.L. Wang, Mar. Drugs 16, 83 (2018)
T.W. Liang, S.C. Tseng, S.L. Wang, Mar. Drugs 14, 40 (2016)
S.L. Wang, C.W. Yang, T.W. Liang, C.L. Wang, Carbohydr. Polym. 78, 205 (2009)
S.L. Wang, C.L. Lin, T.W. Liang, K.C. Liu, Y.H. Kuo, Bioresour. Technol. 100, 316 (2009)
A.D. Nguyen, C.C. Huang, T.W. Liang, V.B. Nguyen, P.S. Pan, S.L. Wang, Carbohydr. Polym. 108, 331 (2014)
N. Shekhar, D. Bhattacharya, D. Kumar, R.L. Gupta, Can. J. Microbiol. 52, 805 (2006)
P. Mander, S.S. Cho, Y.H. Cho, S. Panthi, Y.S. Cho, H.M. Kim, J.C. Yoo, Arch. Pharm. Res. 39, 878 (2016)
N. Karthik, P. Binod, A. Pandey, Bioresour. Technol. 188, 195 (2015)
M.A. Rahman, Y.H. Choi, G.C. Pradeep, J.C. Yoo, Arch. Pharmac. Res. 37, 1522 (2014)
G.J. Joo, Biotechnol. Lett. 27, 1483 (2005)
G. Mukherjee, S.K. Sen, Curr. Microbiol. 53, 265 (2006)
S.L. Wang, T.Y. Huang, C.Y. Wang, T.W. Liang, Y.H. Yen, Y. Sakata, Bioresour. Technol. 99, 5436 (2008)
S.L. Wang, J.Y. Liou, T.W. Liang, K.C. Liu, Process Biochem. 44, 854 (2009)
S.L. Wang, D.Y. Kao, C.L. Wang, Y.H. Yen, M.K. Chern, Y.H. Chen, Enzyme Microb. Technol. 39, 724 (2006)
V.B. Nguyen, A.D. Nguyen, S.L. Wang, Mar. Drugs 15, 274 (2017)
S.L. Wang, H.T. Li, L.J. Zhang, Z.H. Lin, Y.H. Kuo, Mar. Drugs 14, 183 (2016)
T.W. Liang, S.C. Tseng, S.L. Wang, Mar. Drugs 14, 40 (2016)
T.W. Liang, C.C. Wu, W.T. Cheng, Y.C. Chen, C.L. Wang, I.L. Wang, S.L. Wang, Appl. Biochem. Biotechnol. 172, 933 (2014)
C.L. Wang, T.H. Huang, T.W. Liang, S.L. Wang, New Biotechnol. 28, 559 (2011)
S.L. Wang, C.Y. Wang, Y.H. Yen, T.W. Liang, S.Y. Chen, C.H. Chen, Process Biochem. 47, 1684 (2012)
S.L. Wang, W.H. Hsu, T.W. Liang, Carbohydr. Res. 345, 880 (2010)
S.L. Wang, W.T. Chang, Appl. Environ. Microbiol. 63, 380 (1997)
T.W. Liang, Y.J. Chen, Y.H. Yen, S.L. Wang, Process Biochem. 42, 527 (2007)
K. Kim, N. Rajapakse, Carbohydr. Polym. 62, 357 (2005)
H.W. Lee, Y.S. Parkb, J.S. Jungb, W.S. Shinb, Anaerobe 8, 319 (2002)
Acknowledgment
This work was supported in part by a grant from the Ministry of Science and Technology, Taiwan (MOST 106-2320-B-032-001-MY3).
Author information
Authors and Affiliations
Contributions
Conceived the study: S-LW. Designed and performed the study: S-LW, TNT. Contributed reagents/materials/analysis tools: S-LW. Analyzed data: TNT, CTD, VBN, ADN. Wrote the paper: S-LW, TNT.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Tran, T.N., Doan, C.T., Nguyen, V.B. et al. The isolation of chitinase from Streptomyces thermocarboxydus and its application in the preparation of chitin oligomers. Res Chem Intermed 45, 727–742 (2019). https://doi.org/10.1007/s11164-018-3639-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11164-018-3639-y