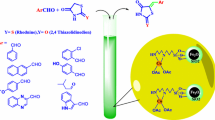
Abstract
A novel nanomagnetic catalyst with Cl[DABCO-NO2]C(NO2)3 tags was designed, synthesized and fully characterized by several techniques such as Fourier transform infrared spectroscopy, energy-dispersive X-ray spectroscopy, elemental mapping analysis, thermogravimetric analysis, derivative thermal gravimetric, powder X-ray diffraction patterns, field scanning electron microscopy, high resolution transmission electron microscopy, physical adsorption and desorption of N2 isotherms (BET) and vibrating sample magnetometer. The synthesized nanomagnetic particles were used as an efficient and recyclable catalyst for the one-pot three component condensation reaction of 3-methyl-1-phenyl-1H-pyrazol-5-amine or 3-methyl-1H-pyrazol-5-amine, aryl aldehydes and malononitrile for the synthesis of pyrazolo[3,4-b]-pyridine derivatives under neat conditions.

















Similar content being viewed by others
References
O. Deutschmann, H. Knözinger, K. Kochloefl, T. Turek, Heterogeneous Catalysis and Solid Catalysts (Wiley, Weinheim, 2009)
M.A. Zolfigol, T. Azadbakht, V. Khakyzadeh, R. Nejatyami, D. Perrin, RSC Adv. 4, 40036 (2014)
M.A. Zolfigol, V. Khakyzadeh, A.R. Moosavi-Zare, A. Rostami, A. Zare, N. Iranpoor, M.H. Beyzavi, R. Luque, Green Chem. 15, 2132 (2013)
H. Sharghi, A. Khoshnood, M.M. Doroodmand, R. Khalifeh, J. Iran. Chem. Soc. 9, 231 (2012)
H. Sharghi, A. Khoshnood, R. Khalifeh, M.M. Doroodmand, Mol. Divers. 19, 481 (2015)
S.B. Sapkal, K.F. Shelke, B.B. Shingate, M.S. Shingare, Tetrahedron Lett. 50, 1754 (2009)
M. Fallah-Mehrjardi, Mini. Rev. Org. Chem. 14, 122 (2017)
M. Mokhtary, J. Iran. Chem. Soc. 13, 1827 (2016)
V. Polshettiwar, R. Luque, A. Fihri, H. Zhu, M. Bouhrara, J.M. Basset, Chem. Rev. 111, 3036 (2011)
B. Karimi, F. Mansouri, H.M. Mirzaei, ChemCatChem 7, 1736 (2015)
D. Zhang, C. Zhou, Z. Sun, L.Z. Wu, C.H. Tung, T. Zhang, Nanoscale 4, 6244 (2012)
M.B. Gawande, P.S. Branco, R.S. Varma, Chem. Soc. Rev. 42, 3371 (2013)
S. Hu, Y. Guan, Y. Wang, H. Han, Appl. Energ. 88, 2685 (2011)
A. Khazaei, M.A. Zolfigol, A.R. Moosavi-Zare, J. Afsar, A. Zare, V. Khakyzadeh, M.H. Beyzavi, Chin. J. Catal. 34, 1936 (2013)
M.A. Zolfigol, M. Yarie, RSC Adv. 5, 103617 (2015)
H. Sharghi, A. Khoshnood, M.M. Doroodmand, R. Khalifeh, J. Heterocycl. Chem. 53, 164 (2016)
E. Juaristi, G. Cuevas, Tetrahedron 48, 5019 (1992)
S.A. Glover, Tetrahedron 54, 7229 (1998)
E. Juaristi, Y. Bandala, Adv. Heterocycl. Chem. 105, 189 (2012)
C.M. Filloux, Angew. Chem. Int. Ed. 54, 8880 (2015)
M. Yarie, Iran. J. Catal. 7, 85 (2017)
G. Hamasaka, H. Tsuji, Y. Uozumi, Synlett 26, 2037 (2015)
T. He, R. Shi, Y. Gong, G. Jiang, M. Liu, S. Qian, Z. Wang, Synlett 27, 1864 (2016)
C.-B. Bai, N.-X. Wang, Y. Xing, X.-W. Lan, Synlett 28, 402 (2017)
N.-X. Wang, J. Zhao, Synlett 18, 2785 (2007)
L.F. Pedrosa, W.P. de Macedo, A.C.R. Furtado, G.P. Guedes, J.C. Borges, J.A.L.C. Resende, M.G.F. Vaz, A.M.R. Bernardino, M.C. de Souza, ARKIVOC iv, 38 (2014)
V. Kumar, K. Kaur, G.K. Gupta, A.K. Sharma, Eur. J. Med. Chem. 69, 735 (2013)
D.-Q. Shi, H. Yao, J.-W. Shi, Synth. Commun. 38, 1662 (2008)
I.H. Eissa, A.M. El-Naggar, M.A. El-Hashash, Bioorg. Chem. 67, 43 (2016)
T.I. El-Emary, J. Chin. Chem. Soc. 54, 507 (2007)
X. Zhang, X. Li, X. Fan, X. Wang, J. Wang, G. Qu, Aust. J. Chem. 62, 382 (2009)
Y.L. Chen, Chem. Abstr. 124, 232447 (1995)
J. Quiroga, S. Cruz, B. Insuasty, R. Abonia, J. Heterocycl. Chem. 38, 53 (2001)
B. Zhao, Y. Li, P. Xu, Y. Dai, C. Luo, Y. Sun, J. Ai, M. Geng, W. Duan, ACS Med. Chem. Lett. 7, 629 (2016)
M.D. Hill, H. Fang, J.M. Brown, T. Molski, A. Easton, X. Han, R. Miller, M. Hill-Drzewi, L. Gallagher, M. Matchett, M. Gulianello, A. Balakrishnan, R.L. Bertekap, K.S. Santone, V.J. Whiterock, X. Zhuo, J.J. Bronson, J.E. Macor, A.P. Degnan, ACS Med. Chem. Lett. 7, 1082 (2016)
M.A. Zolfigol, A. Khazaei, S. Alaie, S. Baghery, F. Maleki, Y. Bayat, A. Asghari, RSC Adv. 6, 58667 (2016)
M.A. Zolfigol, H. Gholami, V. Khakyzadeh, Principles of Organic Synthesis with a New Approach, 3rd edn. (Bu-Ali Sina University Publishers, Hamedan, 2014), p. 26
S.J. Angyal, K. James, Aust. J. Chem. 23, 1209 (1970)
J.M. Erhardt, J.D. Wuest, J. Am. Chem. Soc. 102, 6363 (1980)
T.J. Atkins, J. Am. Chem. Soc. 102, 6364 (1980)
J.M. Erhardt, E.R. Grover, J.D. Wuest, J. Am. Chem. Soc. 102, 6365 (1980)
M.A. Zolfigol, F. Afsharnadery, S. Baghery, S. Salehzadeh, F. Maleki, RSC Adv. 5, 75555 (2015)
M.A. Zolfigol, M. Safaiee, F. Afsharnadery, N. Bahrami-Nejad, S. Baghery, S. Salehzadeh, F. Maleki, RSC Adv. 5, 100546 (2015)
A.R. Moosavi-Zare, M.A. Zolfigol, Z. Rezanejad, Can. J. Chem. 94, 626 (2016)
M.A. Zolfigol, M. Kiafar, M. Yarie, A. Taherpour, M. Saeidi-Rad, RSC Adv. 6, 50100 (2016)
M.A. Zolfigol, M. Yarie, Appl. Organometal. Chem. 31, 3598 (2017)
M.A. Zolfigol, M. Kiafar, M. Yarie, A. Taherpour, T. Fellowes, A.N. Hancok, A. Yari, J. Mol. Struct. 1137, 674 (2017)
M. Kiafar, M.A. Zolfigol, M. Yarie, A. Taherpour, RSC Adv. 6, 102280 (2016)
M.A. Zolfigol, M. Safaiee, B. Ebrahimghasri, S. Baghery, S. Alaie, M. Kiafar, A. Taherpour, Y. Bayat, A. Asgari, J. Iran. Chem. Soc. 14, 1839 (2017)
S. Baghery, M.A. Zolfigol, F. Maleki, New J. Chem. 41, 9276 (2017)
N. Koukabi, E. Kolvari, A. Khazaei, M.A. Zolfigol, B.S. Shaghasemi, H.R. Khavasi, Chem. Commun. 47, 9230 (2011)
N. Koukabi, E. Kolvari, M.A. Zolfigol, A. Khazaei, B.S. Shaghasemi, B. Fasahati, Adv. Synth. Catal. 354, 2001 (2012)
T. Azadbakht, M.A. Zolfigol, R. Azadbakht, V. Khakizadeh, D. Perrin, New J. Chem. 39, 439 (2015)
M. Safaiee, M.A. Zolfigol, F. Afsharnadery, S. Baghery, RSC Adv. 5, 102340 (2015)
M. Yarie, M.A. Zolfigol, Y. Bayat, A. Asgari, D.A. Alonso, A. Khoshnood, RSC Adv. 6, 82842 (2016)
L. Mohammadi, M.A. Zolfigol, M. Ebrahiminia, K.P. Roberts, S. Ansari, T. Azadbakht, S.R. Hussaini, Catal. Commun. 102, 44 (2017)
M. Rajabi-Salek, M.A. Zolfigol, M. Zarei, Res. Chem. Intermed (2018)
S. Moradi, M.A. Zolfigol, M. Zarei, D.A. Alonso, A. Khoshnood, A. Tajally, Appl. Organomet. Chem. (2018).
M.A. Zolfigol, R. Ayazi-Nasrabadi, S. Baghery, V. Khakyzadeh, S. Azizian, J. Mol. Catal. A Chem. 418–419, 54 (2016)
M.A. Zolfigol, A.R. Moosavi-Zare, M. Zarei, A. Zare, E. Noroozizadeh, R. Karamian, M. Asadbegy, RSC Adv. 6, 62460 (2016)
Acknowledgements
We thank Bu-Ali Sina University, Iran National Science Foundation (INSF) (Grant Number: 95831207), National Elites Foundation, University of Alicante (VIGROB-173, UAUSTI16-03), and the Spanish Ministerio de Economía y Competitividad (CTQ2015-66624-P) for financial support to our research groups.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Afsar, J., Zolfigol, M.A., Khazaei, A. et al. Synthesis and application of a novel nanomagnetic catalyst with Cl[DABCO-NO2]C(NO2)3 tags in the preparation of pyrazolo[3,4-b]pyridines via anomeric based oxidation. Res Chem Intermed 44, 7595–7618 (2018). https://doi.org/10.1007/s11164-018-3576-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11164-018-3576-9