Abstract
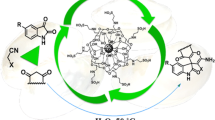
Calcined oyster shell nanoparticles (COS NPs) as a novel heterogeneous nanocatalyst were prepared and fully characterized by X-ray fluorescence analysis, Fourier transform infrared spectroscopy, X-ray powder diffraction, scanning electron microscopy equipped with an energy dispersive spectrometry (SEM-EDS) and transmission electron microscopy. The composition of this catalyst was determined, and it was clearly found that the particle size of COS NPs was about 100–120 nm. The nano-structured solid catalyst as an efficient and eco-friendly catalyst was used for the synthesis of 1,8-dioxo-octahydroxanthenes via the one-pot condensation reaction of 5,5-dimethylcyclohexane-1,3-dione (dimedone) with various aldehydes under solvent-free conditions. This new methodology is cost-effective and offers several advantages such as high yield, excellent purity, short reaction time, high atom economy, simple operation and a convenient workup procedure without the use of any injurious solvents. Moreover, the catalyst can be readily reusable, and considerable catalytic activity could be achieved after the six runs.








Similar content being viewed by others
References
R.S. Varma, Tetrahedron 58, 7 (2002)
K. Chibale, M. Visser, D. van Schalkwyk, P.J. Smith, A. Saravanamuthu, A.H. Fairlamb, Tetrahedron 59, 13 (2003)
Y.M. Shchekotikhin, T.G. Nikolaeva, Chem. Heterocycl. Compd. 42, 1 (2006)
S.A. Hilderbrand, R. Weissleder, Tetrahedron Lett. 48, 25 (2007)
G. Pohlers, J.C. Scaiano, R. Sinta, Chem. Mater. 9, 12 (1997)
C.G. Knight, T. Stephens, Biochem. J. 258, 3 (1989)
R. Giri, J.R. Goodell, C. Xing, A. Benoit, H. Kaur, H. Hiasa, D.M. Ferguson, Biorg. Med. Chem. 18, 4 (2010)
D.M. Parkin, F. Bray, J. Ferlay, P. Pisani, CA Cancer J. Clin. 55, 2 (2005)
S. Hatakeyama, N. Ochi, H. Numata, S. Takano, J. Chem. Soc., Chem. Commun. (1988). doi:10.1039/C39880001202
G. Cingolani, F. Gualtieri, M. Pigini, J. Med. Chem. 12, 3 (1969)
S. Ahadi, H.R. Khavasi, A. Bazgir, Chem. Pharm. Bull. 56, 9 (2008)
C.N. O’Callaghan, T. Brian, M. McMurry, J. Chem. Res., Synop. (1997). doi:10.1039/A607362J
A. Baeyer, Ber. Dtsch. Chem. Ges. 4, 2 (1871)
R.-H. Wang, B. Cui, W.-T. Zhang, G.-F. Han, Synth. Commun. 40, 12 (2010)
H.-Y. Lü, J.-J. Li, Z.-H. Zhang, Appl. Organomet. Chem. 23, 4 (2009)
L. Wu, C. Yang, L. Yang, L. Yang, Phosphorus, Sulfur Silicon Relat. Elem. 185, 4 (2010)
J.-J. Li, X.-Y. Tao, Z.-H. Zhang, Phosphorus, Sulfur Silicon Relat. Elem. 183, 7 (2008)
G.K. Verma, K. Raghuvanshi, R.K. Verma, P. Dwivedi, M.S. Singh, Tetrahedron 67, 20 (2011)
I. Mohammadpoor-Baltork, M. Moghadam, V. Mirkhani, S. Tangestaninejad, H. Reza Tavakoli, Chin. Chem. Lett. 22, 1 (2011)
A. Zare, A.R. Moosavi-Zare, M. Merajoddin, M.A. Zolfigol, T. Hekmat-Zadeh, A. Hasaninejad, A. Khazaei, M. Mokhlesi, V. Khakyzadeh, F. Derakhshan-Panah, M.H. Beyzavi, E. Rostami, A. Arghoon, R. Roohandeh, J. Mol. Liq. 167, 69 (2012)
R.-Z. Wang, L.-F. Zhang, Z.-S. Cui, Synth. Commun. 39, 12 (2009)
B. Das, J. Kashanna, R.A. Kumar, P. Jangili, Synth. Commun. 42, 19 (2012)
L.J.-C. MA Jing-Jun, TANG Ran-Xiao, ZHOU Xin, WU Qiu-Hua, WANG Chun*, ZHANG Ming-Ming, LI Qing Chin, J. Org. Chem. 27, 05 (2007)
A. Khazaei, A. Reza Moosavi-Zare, Z. Mohammadi, A. Zare, V. Khakyzadeh, G. Darvishi, RSC Adv. 3, 5 (2013)
T.S. Jin, J.S. Zhang, A.Q. Wang, T.S. Li, Synth. Commun. 35, 17 (2005)
S. Kantevari, R. Bantu, L. Nagarapu, J. Mol. Catal. A: Chem. 269, 1–2 (2007)
P. Srihari, S.S. Mandal, J.S.S. Reddy, R.S. Rao, J.S. Yadav, Chin. Chem. Lett. 19, 7 (2008)
H.A. Soliman, T.A. Salama, Chin. Chem. Lett. 24, 5 (2013)
Z.-H. Zhang, Y.-H. Liu, Catal. Commun. 9, 8 (2008)
H.R. Shaterian, A. Hosseinian, M. Ghashang, Phosphorus, Sulfur Silicon Relat. Elem. 183, 12 (2008)
M. Seyyedhamzeh, P. Mirzaei, A. Bazgir, Dyes Pigm. 76, 3 (2008)
M. Esmaeilpour, J. Javidi, F. Dehghani, F. Nowroozi Dodeji, New J. Chem. 38, 11 (2014)
G.I. Shakibaei, P. Mirzaei, A. Bazgir, Appl. Catal. A 325, 1 (2007)
B. Das, P. Thirupathi, I. Mahender, V.S. Reddy, Y.K. Rao, J. Mol. Catal. A: Chem. 247, 1–2 (2006)
M. Dabiri, M. Baghbanzadeh, E. Arzroomchilar, Catal. Commun. 9, 5 (2008)
J.-J. Ma, C. Wang, Q.-H. Wu, R.-X. Tang, H.-Y. Liu, Q. Li, Heteroat. Chem. 19, 6 (2008)
S. Rostamizadeh, A.M. Amani, G.H. Mahdavinia, G. Amiri, H. Sepehrian, Ultrason. Sonochem. 17, 2 (2010)
P. Sivaguru, A. Lalitha, Chin. Chem. Lett. 25, 2 (2014)
Z. Wei, C. Xu, B. Li, Bioresour. Technol. 100, 11 (2009)
N. Nakatani, H. Takamori, K. Takeda, H. Sakugawa, Bioresour. Technol. 100, 3 (2009)
L. Yang, A. Zhang, X. Zheng, Energy Fuels 23, 8 (2009)
N. Viriya-empikul, P. Krasae, B. Puttasawat, B. Yoosuk, N. Chollacoop, K. Faungnawakij, Bioresour. Technol. 101, 10 (2010)
P.-L. Boey, G.P. Maniam, S.A. Hamid, D.M.H. Ali, Fuel 90, 7 (2011)
P.-L. Boey, G.P. Maniam, S.A. Hamid, Bioresour. Technol. 100, 24 (2009)
R. Chakraborty, S. Bepari, A. Banerjee, Bioresour. Technol. 102, 3 (2011)
T. Issariyakul, A.K. Dalai, Renew. Sustain. Energy Rev. 31, 446 (2014)
T.-C. Hsu, J. Hazard. Mater. 171, 1–3 (2009)
W.-T. Kuo, H.-Y. Wang, C.-Y. Shu, D.-S. Su, Constr. Build. Mater. 46, 128 (2013)
C.-R. Yang, T.-H. Ko, Y.-C. Lin, S.-Z. Lee, Y.-F. Chang, H.-T. Hsueh, Environ. Chem. Lett. 11, 1 (2013)
N. Tsugawa, T. Okano, R. Higashino, T. Kimura, Y. Oshio, Y. Teraoka, C. Igarashi, I. Ezawa, T. Kobayashi, Biol. Pharm. Bull. 18, 5 (1995)
B. Yoosuk, P. Udomsap, B. Puttasawat, P. Krasae, Bioresour. Technol. 101, 10 (2010)
N. Razavi, B. Akhlaghinia, RSC Adv. 5, 16 (2015)
S.S.E. Ghodsinia, B. Akhlaghinia, RSC Adv. 5, 62 (2015)
M. Zarghani, B. Akhlaghinia, Appl. Organomet. Chem. 29, 10 (2015)
Z. Zarei, B. Akhlaghinia, Chem. Pap. 69, 11 (2015)
M. Zarghani, B. Akhlaghinia, RSC Adv. 5, 107 (2015)
N. Razavi, B. Akhlaghinia, New J. Chem. 40, 1 (2016)
R. Jahanshahi, B. Akhlaghinia, RSC Adv. 5, 126 (2015)
N.Y. Siavashi, B. Akhlaghinia, M. Zarghani, Res. Chem. Intermed. 42, 6 (2016)
E. Karimian, B. Akhlaghinia, S.S.E. Ghodsinia, J. Chem. Sci. 128, 3 (2016)
R. Jahanshahi, B. Akhlaghinia, RSC Adv. 6, 35 (2016)
M. Zarghani, B. Akhlaghinia, RSC Adv. 6, 38 (2016)
M. Zarghani, B. Akhlaghinia, RSC Adv. 6, 45 (2016)
S.S.E. Ghodsinia, B. Akhlaghinia, R. Jahanshahi, RSC Adv. 6, 68 (2016)
M. Zarghani, B. Akhlaghinia, Bull. Chem. Soc. Jpn 89, 10 (2016)
S.M. Masjed, B. Akhlaghinia, M. Zarghani, N. Razavi, Aust. J. Chem. 70, 1 (2017)
Z. Zarei, B. Akhlaghinia, RSC Adv. 6, 108 (2016)
N. Mohammadian, B. Akhlaghinia, Res. Chem. Intermed. (2016). doi:10.1007/s11164-016-2828-9
R. Jahanshahi, B. Akhlaghinia, New J. Chem. 41, 15 (2017)
M.S. Ghasemzadeh, B. Akhlaghinia, Bull. Chem. Soc. Jpn (2017). doi:10.1246/bcsj.20170148
R. Jahanshahi, B. Akhlaghinia, Catal. Lett. (2017). doi:10.1007/s10562-017-2170-x
A. Mohammadinezhad, B. Akhlaghinia, Aust. J. Chem. (2017). doi:10.1071/CH17093
M. Nejatianfar, B. Akhlaghinia, Appl. Organomet. Chem. (2017). doi:10.1002/aoe.4095
P. Das, A. Dutta, A. Bhaumik, C. Mukhopadhyay, Green Chem. 16, 3 (2014)
S.K. Barbara Plavsic, Boris Orel KZLTET 33, 6 (1999)
G. Vidal-Valat, J.P. Vidal, K. Kurki-Suonio, Acta Crystallogr. Sect. A 34, 4 (1978)
M. Kidwai, A. Jain, Appl. Organomet. Chem. 26, 10 (2012)
A. Fallah, M. Tajbakhsh, H. Vahedi, A. Bekhradnia, Res. Chem. Intermed. 43, 1 (2017)
K. Gong, H. Wang, S. Wang, Y. Wang, J. Chen, Chin. J. Catal. 36, 8 (2015)
Acknowledgements
The authors gratefully acknowledge the partial support of this study by Ferdowsi University of Mashhad Research Council (grant no. p/3/43388).
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Mohammadian, N., Akhlaghinia, B. Calcined oyster shell nanoparticles (COS NPs): a new, efficient and reusable catalyst for one-pot rapid preparation of 1,8-dioxo-octahydroxanthenes under solvent-free conditions. Res Chem Intermed 44, 1085–1103 (2018). https://doi.org/10.1007/s11164-017-3153-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11164-017-3153-7