Abstract
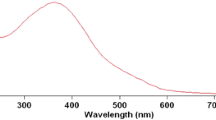
The kinetics of propanol oxidation by quinquivalent vanadium in the presence of H2SO4 has been followed by UV-spectrophotometer at 30 °C temperature. It has been found that sodium dodecyl sulfate (SDS) micelles produce a large catalytic effect on this oxidation process. The pseudo-first-order rate constants for the reaction increases with SDS concentrations. The kinetic results have been explained by considering the preferential partitioning of reactants between the micellar and aqueous phase and also local the concentration effect. The oxidized product propionaldehyde is identified by 2,4-DNP test and 1H-NMR spectral measurement. The formation of aggregate in reaction condition was confirmed by measuring the CMC value of SDS in aqueous medium and in the presence of propanol by conductivity measurements. The change in the size of aggregate, from DLS measurement, confirms the localization of reactant species towards the aggregate, which is the consequence of rate enhancement.






Similar content being viewed by others

References
R.A. Basson, H.J. van der Linde, J. Chem. Soc. A. 662–665 (1968)
R.A. Basson, H.J. van der Linde, Chem. Commun. (Lond.) 91–92 (1967)
A.R. Day, A. Eisner, J. Phys. Chem. 36, 1912–1915 (1932)
J. Schnaidt, M. Heinen, Z. Jusys, R.J. Behm, Electrochim. Acta 104, 505–517 (2013)
P. Ocon, C. Alonso, R. Celdran, J.G. Velasco, J. Electroanal. Chem. Interfacial Electrochem. 206, 179–196 (1986)
A. Ghosh, R. Saha, B. Saha, J. Ind. Eng. Chem. 20, 345–350 (2014)
A. Ghosh, R. Saha, K. Mukherjee, S.K. Ghosh, S.S. Bhattacharyya, S. Laskar, B. Saha, Int. J. Chem. Kinet. 45, 175–179 (2013)
V.V. Namboodiri, V. Polshettiwar, R.S. Varma, Tetrahedron Lett. 48, 8839–8842 (2007)
S. Rani, B.R. Bhat, Tetrahedron Lett. 51, 6403–6405 (2010)
M.S. Thompson, T.J. Meyer, J. Am. Chem. Soc. 104, 4106–4115 (1982)
A. Ghosh, R. Saha, K. Mukherjee, P. Sar, S.K. Ghosh, S. Malik, S.S. Bhattacharyya, B. Saha, J. Mol. Liq. 190, 81–86 (2014)
A. Ghosh, R. Saha, P. Sar, B. Saha, J. Mol. Liq. 186, 122–130 (2013)
M.A. Malika, S.A. AL-Thabaitib, Z. Khan, Eng. Asp. 337, 9–14 (2009)
S. Dash, S. Patel, B.K. Mishra, Tetrahedron 65, 707–739 (2009)
B. Saha, C. Orvig, Coord. Chem. Rev. 254, 2959–2972 (2010)
T. Hirao, Chem. Rev. 97, 2707–2724 (1997)
D.C. Crans, J.J. Smee, E. Gaidamauskas, L. Yang, Chem. Rev. 104, 849–902 (2004)
E.O. Odebunmi, A.S. Ogunlaja, S.O. Owalude, J. Chil. Chem. Soc. 55, 293–297 (2010)
P.C. Wilkins, M.D. Johnson, A.A. Holder, D.C. Crans, Inorg. Chem. 45, 1471–1479 (2006)
S. Malik, B. Jain, S. Ghosh, Asian J. Exp. Sci. 21, 87–92 (2007)
P. Sar, A. Ghosh, R. Saha, B. Saha, Res. Chem. Intermed. (2014). doi:10.1007/s11164-014-1635-4
P. Sar, A. Ghosh, D. Ghosh, B. Saha, Res. Chem. Intermed. (2014). doi:10.1007/s11164-014-1682-x
S.K. Ghosh, A. Basu, K.K. Paul, B. Saha, Mol. Phys. 107, 615–619 (2009)
S.K. Ghosh, R. Saha, A. Ghosh, K. Mukherjee, B. Saha, Tenside Surf. Det. 49, 296–299 (2012)
B. Saha, K.M. Chowdhury, J. Mandal, J. Solut. Chem. 37, 1321–1328 (2008)
B. Saha, S. Sarkar, K.M. Chowdhury, Int. J. Chem. Kinet. 40, 282–286 (2008)
B. Saha, Inorg. React. Mech. 6, 287–291 (2008)
K.R. Lange (ed.), Surfactants: A Practical Handbook (Hanser Publishers, Munich, 1999)
M.J. Schwuger, K. Stickdorn, R. Schomacker, Chem. Rev. 95, 849–864 (1995)
Kabir-ud-Din, M.S. Ali, Z. Khan, Colloid Polym. Sci. 284, 627–633 (2006)
C.A. Bunton, Arkivoc. 7, 490–504 (2011)
B. Kumar, M.L. Satnami, K.K. Ghosh, K. Kuca, J. Phys. Org. Chem. 25, 864–871 (2012)
M.A. Malik, F. Nabi, Z. Khan, J. Dispers Sci Technol. 29, 1396–1400 (2008)
M. Arias, L.G. Rı´O, J.C. Mejuto, P.R. Dafonte, J.S. Gaandara, J. Agric. Food Chem. 53, 7172–7178 (2005)
F. Feigl, Spot Tests in Organic Analysis, vol. 342 (Elsevier, Amsterdam, 1956), p. 121
T.S. Smith, V.L. Pecoraro, Inorg. Chem. 41, 6754–6760 (2002)
Acknowledgments
We are grateful to the UGC and CSIR, New Delhi, for providing financial help in the form of a research grant and fellowship. The authors would also like to recognize IACS, Kolkata, India, for providing SEM and DLS measurements.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Sar, P., Ghosh, A. & Saha, B. The influence of SDS micelle on the oxidative transformation of propanol to propionaldehyde by quinquivalent vanadium in aqueous medium at room temperature. Res Chem Intermed 41, 7775–7784 (2015). https://doi.org/10.1007/s11164-014-1858-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11164-014-1858-4