Abstract
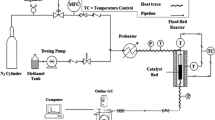
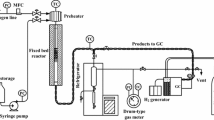
SAPO-34 molecular sieves with different crystal morphologies were synthesized under microwave conditions. The type of organic templates, crystallization time and temperature were optimized to control the synthesis. These SAPO-34 samples were characterized by XRD, SEM, EDX, TG, nitrogen adsorption–desorption, NH3-TPD and solid state MAS NMR techniques. It was found that the crystallization conditions greatly affected the crystal morphology, textural data, Si incorporation and acidity of SAPO-34. Pure SAPO-34 with uniform sphere particles of 20 nm was obtained after crystallization at 165 °C for 0.75 h under microwave conditions using TEAOH as the template. Raising the temperature to 220 °C and prolonging the crystallization time to 2 h led to the formation of nano sheet-like SAPO-34 with a BET surface area of 593 m2/g. NH3-TPD profiles showed that low crystallization temperature and short crystallization time resulted in fewer weak acid sites and strong acid sites, while higher crystallization temperature and longer crystallization time increased the amount of both acid sites, which may benefit the methanol to olefins (MTO) reaction. The nano sheet-like SAPO-34 exhibited 100 % methanol conversion for 380 min during the MTO process, an ethylene selectivity of 51.77 % and a C =2 –C =4 selectivity of 90.20 %. The result was much better than that of the sample with a larger cubic morphology of 1.5–2.5 μm synthesized under hydrothermal conditions, on which the 100 % methanol conversion could only be kept for 212 min as well as an ethylene selectivity of 49.84 % and a C =2 –C =4 selectivity of 86.81 %. The unique morphology of the nano sheet-like SAPO-34 decreased its coke formation rate, which might be responsible for its improved catalytic performance.







Similar content being viewed by others
References
Wilson ST, Lok BM, Messina CA, Cannan TR, Flanigen EM (1982) J Am Chem Soc 104:1146–1147
Lok BM, Messina CA, Patton RL, Gajek RT, Cannan TR, Flanigen EM (1984) US patent:4,400,781
Salmasi M, Fatemi S, Najafabadi AT (2011) J Ind Eng Chem 17:755–761
Davis ME (2002) Nature 417:813–821
Yu J, Xu R (2006) Chem Soc Rev 35:593–604
Vora BV, Marker TL, Barger PT, Nilsen HR, Kvisle S, Fuglerud T (1997) Stud Surf Sci Catal 107:87–98
Liang J, Li HY, Zhao S, Guo WG, Wang RH, Ying ML (1990) Appl Catal 64:31–40
Ye L, Cao F, Ying W, Fang D, Sun Q (2010) J Porous Mater 18:225–232
Nishiyama N, Kawaguchi M, Hirota Y, Van Vu D, Egashira Y, Ueyama K (2009) Appl Catal A Gen 362:193–199
Jun JW, Lee JS, Seok HY, Chang JS, Hwang JS, Jhung SH (2011) Bull Korean Chem Soc 32:1957–1964
Lee KY, Chae HJ, Jeong SY, Seo G (2009) Appl Catal A Gen 369:60–66
Chen D, Moljord K, Fuglerud T, Holmen A (1999) Microporous Mesoporous Mater 29:191–203
Álvaro-Muñoz T, Márquez-Álvarez C, Sastre E (2012) Catal Today 179:27–34
Dargahi M, Kazemian H, Soltanieh M, Rohani S, Hosseinpour M (2011) Particuology 9:452–457
Yao J, Wang H, Ringer SP, Chan KY, Zhang L, Xu N (2005) Microporous Mesoporous Mater 85:267–272
Hirota Y, Murata K, Tanaka S, Nishiyama N, Egashira Y, Ueyama K (2010) Mater Chem Phys 123:507–509
Askari S, Halladj R (2012) Ultrason Sonochem 19:554–559
Lin S, Li J, Sharma RP, Yu J, Xu R (2010) Top Catal 53:1304–1310
Venna SR, Carreon MA (2009) J Mater Chem 19:3138–3140
Tan J, Liu Z, Bao X, Liu X, Han X, He C, Zhai R (2002) Microporous Mesoporous Mater 53:97–108
Liu G, Tian P, Li J, Zhang D, Zhou F, Liu Z (2008) Microporous Mesoporous Mater 111:143–149
Cheng M, Tan D, Liu X, Han X, Bao X, Lin L (2001) Microporous Mesoporous Mater 42:307–316
van Niekerk MJ, Fletcher JCQ, O’Connor CT (1996) Appl Catal A Gen 138:135–145
Travalloni L, Gomes ACL, Gaspar AB, da Silva MAP (2008) Catal Today 133–135:406–412
Tan J, Liu Z, Bao X, Liu X, Han X (2002) Microporous Mesoporous Mater 53:97–108
Chen T, Wouters BH, Grobet PJ (1999) J Phys Chem B 103:6179–6184
Zibrowius B, Löffler E, Hunger M (1992) Zeolites 12:167–174
Ashtekar S, Chilukuri SVV, Chakrabarty DK (1994) J Phys Chem 98:4878–4883
Ojo AF, Dwyer J, Dewing J, O’Malley PJ, Nabhan A (1992) J Chem Soc Faraday Trans 88:105–112
Wei Y, Zhang D, He Y, Xu L, Yang Y, Su BL, Liu Z (2007) Catal Lett 114:30–35
Beale AM, O’Brien MG, Kasunič M, Golobič A, Sanchez–Sanchez M, Lobo AJW, Lewis DW, Wragg DS, Nikitenko S, Bras W, Weckhuysen BM (2011) J Phys Chem C 115:6331–6340
Aguayo AT, Gayubo AG, Atutxa A, Olazar M, Bilbao J (1999) J Chem Technol Biotechnol 74:1082–1088
Hajiashrafi T, Kharat AN (2013) Reac Kinet Mech Cat 108:417–432
Wang P, Lv A, Hu J, Xu J, Lu G (2012) Microporous Mesoporous Mater 152:178–184
Zhang J, Zhang H, Yang X, Huang Z, Cao W (2011) J Nat Gas Chem 20:266–270
Chen D, Rebo HK, Moljord K, Holmen A (1997) Ind Eng Chem Res 36:3473–3479
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Wu, L., Liu, Z., Qiu, M. et al. Morphology control of SAPO-34 by microwave synthesis and their performance in the methanol to olefins reaction. Reac Kinet Mech Cat 111, 319–334 (2014). https://doi.org/10.1007/s11144-013-0639-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11144-013-0639-1