Summary
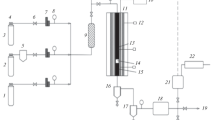
Characterization (BET and TPD) and reaction studies were conducted with activated carbon supported iron catalysts (Fe/AC) used for the Fischer-Tropsch synthesis (FTS). The TPD study showed that there existed interactions between metals and the AC surface. Greater association of Cu and K promoters with the AC surface resulted in stronger promoter to surface interaction, which enhanced the H2 desorption ability of the Cu and K promoted Fe/AC catalyst prepared under vacuum impregnation (VI). Catalytic behavior of a Fe/AC catalyst (VI-15 Fe/2 Cu/2 K/81 AC, in parts per weight) was studied in a 1-liter slurry phase continuous stirred tank reactor. The catalyst presented moderate syngas conversion (44.3-60.6%) and high gaseous selectivity (CH4, 12.8-15.1% and C2-C4, 42.4-46.1%) under 304oC, 3.0 MPa, 1.1 L(STP)/g-cat/h, and H2/CO = 2.0 during 166 h of testing. Detectable hydrocarbons up to C18 were formed on the Fe/AC catalyst.
Similar content being viewed by others
Change history
06 October 2023
A Correction to this paper has been published: https://doi.org/10.1007/s11144-023-02499-5
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Ma, W., Ding, Y., Yang, J. et al. Study of activated carbon supported iron catalysts for the Fischer-Tropsch synthesis. React Kinet Catal Lett 84, 11–19 (2005). https://doi.org/10.1007/s11144-005-0185-6
Issue Date:
DOI: https://doi.org/10.1007/s11144-005-0185-6