Abstract
Purpose
It is unknown how sepsis survivors conceptualize health-related quality of life (HRQL). We aimed to identify important HRQL domains for this population.
Methods
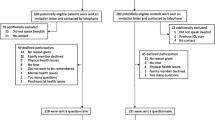
A literature search was performed to inform an interview guide. Open-ended interviews were held with 15 purposefully sampled sepsis survivors. Interview transcripts were analyzed by interpretative phenomenological analysis to allow themes to develop organically. Resulting codes were reviewed by an independent expert. The preliminary list of domains was rated in a two-round Delphi consensus procedure with therapists and survivors.
Results
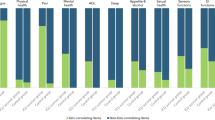
Eleven domains emerged as critically important: Psychological impairment, Fatigue, Physical impairment, Coping with daily life, Return to normal living, Ability to walk, Cognitive impairment, Self-perception, Control over one’s life, Family support, and Delivery of health care. Sepsis survivors want a “normal life,” to walk again, and to regain control without cognitive impairment. Family support is essential to overcome sepsis aftermaths.
Conclusions
Survivors described many HRQL domains which are not captured by the QoL instruments that have traditionally been used to study ICU survivorship (i.e., SF-36 and EQ-5D). Future studies of QoL in ICU survivors should consider using both a traditional instrument so that results are comparable to previous research, as well as a more holistic QoL measurement instrument like the WHOQOL-BREF.

Similar content being viewed by others
Abbreviations
- ARDS:
-
Acute respiratory distress syndrome
- EQ-5D:
-
EuroQoL-5D
- HRQL:
-
Health-related quality of life
- ICU:
-
Intensive care unit
- IPA:
-
Interpretative phenomenological analysis
- PROM:
-
Patient-reported outcome measurement
- RNL:
-
Return to normal living
- SF-36:
-
Short Form 36 Health Survey
- WHOQOL-BREF:
-
Short version of the World Health Organization Quality of Life assessment (WHOQOL-100)
References
Fleischmann, C., Scherag, A., Adhikari, N. K., Hartog, C. S., Tsaganos, T., Schlattmann, P., et al. (2016). Assessment of global incidence and mortality of hospital-treated sepsis. Current estimates and limitations. American Journal of Respiratory and Critical Care Medicine, 193(3), 259–272. https://doi.org/10.1164/rccm.201504-0781OC.
Prescott, H. C., & Angus, D. C. (2018). Enhancing recovery from sepsis: A review. JAMA, 319(1), 62–75. https://doi.org/10.1001/jama.2017.17687.
Iwashyna, T. J., Ely, E. W., Smith, D. M., & Langa, K. M. (2010). Long-term cognitive impairment and functional disability among survivors of severe sepsis. JAMA, 304(16), 1787–1794. https://doi.org/10.1001/jama.2010.1553.
Hall, M. J., Williams, S. N., DeFrances, C. J., & Golosinskiy, A. (2011). Inpatient care for septicemia or sepsis: A challenge for patients and hospitals. NCHS Data Brief, 62, 1–8.
Halpern, N. A., Goldman, D. A., Tan, K. S., & Pastores, S. M. (2016). Trends in critical care beds and use among population groups and Medicare and Medicaid beneficiaries in the United States: 2000–2010. Critical Care Medicine, 44(8), 1490–1499. https://doi.org/10.1097/CCM.0000000000001722.
Seymour, C. W., Gesten, F., Prescott, H. C., Friedrich, M. E., Iwashyna, T. J., Phillips, G. S., et al. (2017). Time to treatment and mortality during mandated emergency care for sepsis. The New England Journal of Medicine, 376(23), 2235–2244. https://doi.org/10.1056/NEJMoa1703058.
Iwashyna, T. J. (2010). Survivorship will be the defining challenge of critical care in the 21st century. Annals of Internal Medicine, 153(3), 204–205. https://doi.org/10.7326/0003-4819-153-3-201008030-00013.
Angus, D. C., Carlet, J., & Brussels Roundtable, P. (2003). Surviving intensive care: A report from the 2002 Brussels Roundtable. Intensive Care Medicine, 29(3), 368–377. https://doi.org/10.1007/s00134-002-1624-8.
Oeyen, S. G., Vandijck, D. M., Benoit, D. D., Annemans, L., & Decruyenaere, J. M. (2010). Quality of life after intensive care: A systematic review of the literature. Critical Care Medicine, 38(12), 2386–2400. https://doi.org/10.1097/CCM.0b013e3181f3dec5.
Schmidt, K., Worrack, S., Von Korff, M., Davydow, D., Brunkhorst, F., Ehlert, U., et al. (2016). Effect of a primary care management intervention on mental health-related quality of life among survivors of sepsis: A randomized clinical trial. JAMA, 315(24), 2703–2711. https://doi.org/10.1001/jama.2016.7207.
Turnbull, A. E., Rabiee, A., Davis, W. E., Nasser, M. F., Venna, V. R., Lolitha, R., et al. (2016). Outcome measurement in ICU survivorship research from 1970 to 2013: A scoping review of 425 publications. Critical Care Medicine, 44(7), 1267–1277. https://doi.org/10.1097/CCM.0000000000001651.
Cuthbertson, B. H., Elders, A., Hall, S., Taylor, J., MacLennan, G., Mackirdy, F., et al. (2013). Mortality and quality of life in the five years after severe sepsis. Critical Care, 17(2), R70. https://doi.org/10.1186/cc12616.
Needham, D. M., Sepulveda, K. A., Dinglas, V. D., Chessare, C. M., Friedman, L. A., & Bingham, C. O., 3rd, et al. (2017). Core outcome measures for clinical research in acute respiratory failure survivors. An international modified Delphi consensus study. American Journal of Respiratory and Critical Care Medicine, 196(9), 1122–1130, https://doi.org/10.1164/rccm.201702-0372OC.
Ware, J. E., Jr., & Sherbourne, C. D. (1992). The MOS 36-item short-form health survey (SF-36). I. Conceptual framework and item selection. Medical Care, 30(6), 473–483.
EuroQol Group. (1990). EuroQol—A new facility for the measurement of health-related quality of life. Health Policy, 16(3), 199–208.
Carr, A. J., & Higginson, I. J. (2001). Are quality of life measures patient centred? BMJ, 322(7298), 1357–1360.
Ramsay, P. (2011). Health-related quality of life: Implications for critical care interventional studies and why we need to collaborate with patients. Current Opinion in Critical Care, 17(5), 510–514. https://doi.org/10.1097/MCC.0b013e32834a4bd4.
Kirkham, J. J., Gorst, S., Altman, D. G., Blazeby, J. M., Clarke, M., Devane, D., et al. (2016). Core outcome Set-STAndards for reporting: The COS-STAR Statement. PLoS Medicine, 13(10), e1002148. https://doi.org/10.1371/journal.pmed.1002148.
Food and Drug Administration (FDA). Patient-reported outcome measures: Use in medical product development to support labeling claims (2009). http://www.fda.gov/downloads/Drugs/Guidances/UCM193282.pdf.
Hofhuis, J. G., van Stel, H. F., Schrijvers, A. J., Rommes, J. H., & Spronk, P. E. (2015). ICU survivors show no decline in health-related quality of life after 5 years. Intensive Care Medicine, 41(3), 495–504. https://doi.org/10.1007/s00134-015-3669-5.
Novick, G. (2008). Is there a bias against telephone interviews in qualitative research? Research in Nursing & Health, 31(4), 391–398. https://doi.org/10.1002/nur.20259.
Sturges, J. E., & Hanrahan, K. J. (2004). Comparing telephone and face-to-face qualitative interviewing: A research note. Qualitative Research, 4(1), 107–118. https://doi.org/10.1177/1468794104041110.
Smith, J. A. (1996). Beyond the divide between cognition and discourse: Using interpretative phenomenological analysis in health psychology. Psychology & Health, 11(2), 261–271. https://doi.org/10.1080/08870449608400256.
Reid, K., Flowers, P., & Larkin, M. (2005). Exploring lived experience. Psychologist, 18(1), 20–23.
Smith, J. A., & Osborn, M. (2007). Interpretative phenomenological analysis. In J. A. Smith (Ed.), Qualitative psychology: A practical guide to research methods (pp. 53–80). Thousand Oaks: SAGE Publications.
Guest, G., Bunce, A., & Johnson, L. (2006). How many interviews are enough?: An experiment with data saturation and variability. Field Methods, 18(1), 59–82. https://doi.org/10.1177/1525822x05279903.
Kerr, C., Nixon, A., & Wild, D. (2010). Assessing and demonstrating data saturation in qualitative inquiry supporting patient-reported outcomes research. Expert Review of Pharmacoeconomics & Outcomes Research, 10(3), 269–281. https://doi.org/10.1586/erp.10.30.
Hasson, F., Keeney, S., & McKenna, H. (2000). Research guidelines for the Delphi survey technique. Journal of Advanced Nursing, 32(4), 1008–1015. https://doi.org/10.1046/j.1365-2648.2000.t01-1-01567.x.
Lim, W. C., Black, N., Lamping, D., Rowan, K., & Mays, N. (2016). Conceptualizing and measuring health-related quality of life in critical care. Journal of Critical Care, 31(1), 183–193. https://doi.org/10.1016/j.jcrc.2015.10.020.
Singer, J. P., Chen, J., Katz, P. P., Blanc, P. D., Kagawa-Singer, M., & Stewart, A. L. (2015). Defining novel health-related quality of life domains in lung transplantation: A qualitative analysis. Quality of Life Research, 24(6), 1521–1533. https://doi.org/10.1007/s11136-014-0875-5.
Eakin, M. N., Patel, Y., Mendez-Tellez, P., Dinglas, V. D., Needham, D. M., & Turnbull, A. E. (2017). Patients’ outcomes after acute respiratory failure: A qualitative study with the PROMIS framework. American Journal of Critical Care, 26(6), 456–465. https://doi.org/10.4037/ajcc2017834.
Skevington, S. M., Lotfy, M., O’Connell, K. A., & Group, W. (2004). The World Health Organization’s WHOQOL-BREF quality of life assessment: Psychometric properties and results of the international field trial. A report from the WHOQOL group. Quality of Life Research, 13(2), 299–310.
Blackwood, B., Marshall, J., & Rose, L. (2015). Progress on core outcome sets for critical care research. Current Opinion in Critical Care, 21(5), 439–444.
Turnbull, A. E., Sepulveda, K. A., Dinglas, V. D., Chessare, C. M., Bingham, C. O., 3rd, & Needham, D. M. (2017). Core domains for clinical research in acute respiratory failure survivors: An international modified Delphi consensus study. Critical Care Medicine, 45(6), 1001–1010. https://doi.org/10.1097/CCM.0000000000002435.
Needham, D. M., Dowdy, D. W., Mendez-Tellez, P. A., Herridge, M. S., & Pronovost, P. J. (2005). Studying outcomes of intensive care unit survivors: Measuring exposures and outcomes. Intensive Care Medicine, 31(9), 1153–1160. https://doi.org/10.1007/s00134-005-2656-7.
Czerwonka, A. I., Herridge, M. S., Chan, L., Chu, L. M., Matte, A., & Cameron, J. I. (2015). Changing support needs of survivors of complex critical illness and their family caregivers across the care continuum: A qualitative pilot study of Towards RECOVER. Journal of Critical Care, 30(2), 242–249. https://doi.org/10.1016/j.jcrc.2014.10.017.
Morse, J. M. (2000). Determining sample size. Qualitative Health Research, 10(1), 3–5. https://doi.org/10.1177/104973200129118183.
Lasch, K. E., Marquis, P., Vigneux, M., Abetz, L., Arnould, B., Bayliss, M., et al. (2010). PRO development: Rigorous qualitative research as the crucial foundation. Quality of Life Research, 19(8), 1087–1096. https://doi.org/10.1007/s11136-010-9677-6.
Acknowledgements
We thank all interview partners and experts who participated in the Delphi process. We thank the two student assistants.
Funding
Christian König and Bastian Matt received a scholarship from the Center of Sepsis Control & Care (CSCC). The CSCC is supported by the German Ministry of Education and Research (Grant No. 01 E0 1002). Christiane S. Hartog was partly funded by the Mid-German Sepsis Cohort which receives funding from the CSCC and the Rudolf Presl GmbH, Kreischa. The funding source had no influence on study design, collection, analysis, and interpretation of data.
Author information
Authors and Affiliations
Contributions
CK and CSH planned the study and drafted the manuscript. CK conducted the study and gathered data. CK, BM, and CSH analyzed and interpreted the data. AET and AK revised the work for important intellectual content. All authors gave final approval of the version to be published.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
The study was approved by the ethics committee of Jena University Hospital (IRB No. 4392-04/15). All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Electronic supplementary material
Rights and permissions
About this article
Cite this article
König, C., Matt, B., Kortgen, A. et al. What matters most to sepsis survivors: a qualitative analysis to identify specific health-related quality of life domains. Qual Life Res 28, 637–647 (2019). https://doi.org/10.1007/s11136-018-2028-8
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11136-018-2028-8