Abstract
Background
The factorial instability of the CIVIQ-20 social dimension in different populations has necessitated the development of a new stable questionnaire to interpret results from international studies.
Objective
Construction of a stable and psychometrically validated questionnaire from CIVIQ-20.
Methods and major findings
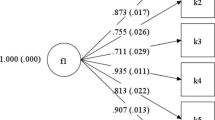
A prospective, international study was used to construct a stable CIVIQ scale and to validate its psychometric properties. An iterative process was implemented to eliminate the more unstable items (six), and the social and physical dimensions were combined. The resulting instrument comprised 14 items, split into three dimensions (pain, physical, and psychological), and was named CIVIQ-14. The stability of the CIVIQ-14 factorial structure was confirmed in Polish, Czech, Spanish, and French populations using principal component analysis and multitrait/multimethod analysis. Psychometric assessment demonstrated that CIVIQ-14 was reliable (intra-class coefficient >0.8; weighted kappa >0.8), valid (correlation coefficients between dimension scores and clinical severity scores between 0.3 and 0.6), and sensitive (effect sizes >0.6 for psychological dimension; >0.8 for the other dimensions).
Conclusion
CIVIQ-14 is a reliable, valid, and sensitive instrument applicable to international studies of patients with chronic venous disease.
Similar content being viewed by others
References
Vasquez, M. A., & Munschauer, C. E. (2008). Venous clinical severity score and quality of life assessment tools: Application to vein practice. Phlebology, 23, 259–275.
Smith, J. J., Guest, M. G., Greenhalgh, R. M., & Davies, A. H. (2000). Measuring the quality of life in patients with venous ulcers. Journal of Vascular Surgery, 31(4), 642–649.
Hareendran, A., Doll, H., Wild, D. J., Moffatt, C. J., Musgrove, E., Wheatley, C., et al. (2007). The venous leg ulcer quality of life (VLU-QoL) questionnaire: Development and psychometric validation. Wound Repair Regen, 15(4), 465–473.
Garratt, A. M., Macdonald, L. M., Ruta, D. A., Russell, I. T., Buckingham, J. K., & Krukowski, Z. H. (1993). Towards measurement of outcome for patients with varicose veins. Quality Health Care, 2(1), 5–10.
Lamping, D. L., Schroter, S., Kurz, X., Kahn, S. R., & Abenhaim, L. (2003). Evaluation of outcomes in chronic venous disorders of the leg: Development of a scientifically rigorous, patient-reported measure of symptoms and quality of life. Journal of Vascular Surgery, 37(2), 410–419.
Guex, J. J., Zimmet, S. E., Boussetta, S., Nguyen, C., & Taieb, C. (2007). Construction and validation of a patient-reported outcome dedicated to chronic venous disorders: SQOR-V (specific quality of life and outcome response - venous). Journal des Maladies Vasculaires, 32, 135–147.
Launois, R., Reboul-Marty, J., & Henry, B. (1996). Construction and validation of a quality of life questionnaire in chronic lower limb venous insufficiency (CIVIQ). Quality of Life Research, 5, 539–554.
Launois, R., Mansilha, A., & Jantet, G. (2010). International psychometric validation of the chronic venous disease quality of life questionnaire CIVIQ-20. European Journal of Vascular and Endovascular Surgery, 40, 783–789.
Padberg, F. (2003). Regarding “Evaluation of outcomes in chronic venous disorders of the leg: Development of a scientifically rigorous, patient-reported measure of symptoms and quality of life”. Journal of Vascular Surgery, 37(4), 911–912.
Guidance for Industry. (2010). Patient-reported outcome measures: Use in medical product development to support labeling claims. http://www.fda.gov/downloads/Drugs/GuidanceComplianceRegulatoryinformation/Guidances/UCM193282.pdf. Accessed Dec 2010.
Jantet, G. (2002). Chronic venous insufficiency: worldwide results of the RELIEF study. Reflux assEssment and quaLity of lIfe improvEment with micronized Flavonoids. Angiology, 53, 245–256.
Launois, R. Data on file, REES France Paris.
Chassany, O., Le-Jeunne, P., Duracinsky, M., Schwalm, M. S., & Mathieu, M. (2006). Discrepancies between patient-reported outcomes and clinician-reported outcomes in chronic venous disease, irritable bowel syndrome, and peripheral arterial occlusive disease. Value Health, 9, 39–46.
Localio, A. R., Berlin, J. A., Ten Have, T. R., & Kimmel, S. E. (2001). Adjustments for center in multicenter studies: An overview. Annals of Internal Medicine, 135, 112–123.
Methods of Monte-Carlo and of Re-Sampling (Jack-Knife Bootstrap) [in French]. (2006). In G. Saporta (Ed.), Probabilités, analyse des données et statistique (2nd edn., pp. 371–386). Paris, France: Editions Technip.
Campbell, D. T., & Fiske, D. W. (1959). Convergent and discriminant validation by the multitrait-multimethod matrix. Psychological Bulletin, 56, 81–105.
Deyo, R. A., Diehr, P., & Patrick, D. L. (1991). Reproducibility and responsiveness of health status measures. Statistics and strategies for evaluation. Controlled Clinical Trials, 12(4 Suppl), 142S–158S.
Kirshner, B., & Guyatt, G. (1985). A methodological framework for assessing health indices. Journal of Chronic Diseases, 38, 27–36.
Guyatt, G., Feeny, D., & Patrick, D. (1993). Measuring health-related quality of life. Annals of Internal Medicine, 118, 622–629.
Norman, G. (1989). Issues in the use of change scores in randomised trial. Journal of Clinical Epidemiology, 42, 1097–1105.
Biemans, A. A., van der Velden, S. K., Bruijninckx, C. M., Buth, J., & Nijsten, T. (2011). Validation of the chronic venous insufficiency quality of life questionnaire in Dutch patients treated for varicose veins. European Journal of Vascular and Endovascular Surgery. doi:10.1016/ejves.2011.04.007.
Guyatt, G. H., Bombardier, C., & Tugwell, P. X. (1986). Measuring disease specific quality of life in clinical trials. CMAJ, 134, 889–895.
Alegre, P., Puskas, A., Giurcaneanu, C., Andercou, A., & Jantet, G. (2010). Absenteeism and impaired quality of life in chronic venous disease in patients in Romania. ISPOR 13th Annual European congress research abstracts. Value in Health, 13, A239–A250.
Sarvananthan, T., Sheperd, A. C., & Davies, A. (2010). Who benefits the most from varicose vein intervention: Can we predict treatment outcomes? Phlebology, 25, 1–2.
Acknowledgments
We thank Les Laboratoires Servier for their financial support of the present analysis and for the decision to submit the present manuscript for publication.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Launois, R., Le Moine, J.G., Lozano, F.S. et al. Construction and international validation of CIVIQ-14 (a short form of CIVIQ-20), a new questionnaire with a stable factorial structure. Qual Life Res 21, 1051–1058 (2012). https://doi.org/10.1007/s11136-011-0008-3
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11136-011-0008-3