Abstract
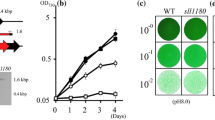
The ATP-binding cassette (ABC) transporter is a multi-subunit membrane protein complex involved in lipid transport and acid stress tolerance in the cyanobacterium Synechocystis sp. PCC 6803. This organism has two sets of three ABC transporter subunits: Slr1045 and Slr1344, Sll0751 and Sll1002, and Sll1001 and Sll1041. We previously found that Slr1045 is essential for survival under acid stress condition (Tahara et al. 2012). In the present study, we examined the participation of other ABC transporter subunits in acid stress tolerance using a deletion mutant series of Synechocystis sp. PCC 6803. Although Slr1344 is highly homologous to Slr1045, Δslr1344 cells were not susceptible to acid stress. Δsll0751 and Δsll1041 cells displayed acid stress sensitivity, whereas Δsll1001/sll1002 double mutant cells grew normally. Under high- and low-temperature stress conditions, the growth rate of Δslr1344 and Δsll1001/sll1002 cells did not differ from WT cells, whereas Δsll0751 and Δsll1041 cells showed significant growth retardation, as previously observed in Δslr1045 cells. Moreover, nile red staining showed more lipid accumulation in Δslr1045, Δsll0751, and Δsll1041 cells than in WT cells. These results suggest that Slr1045, Sll0751, and Sll1041 function together as a lipid transport complex in Synechocystis sp. PCC 6803 and are essential for growth under various stresses.






Similar content being viewed by others
Abbreviations
- ABC:
-
ATP-binding cassette
- PG:
-
Phosphatidylglycerol
- PM:
-
Plasma membrane
- Synechocystis :
-
Synechocystis sp. PCC 6803
- TM:
-
Thylakoid membrane
References
Awai K, Xu C, Tamot B, Benning C (2006) A phosphatidic acid-binding protein of the chloroplast inner envelope membrane involved in lipid trafficking. Proc Natl Acad Sci USA 103:10817–10822
Barrero JM, Rodríguez PL, Quesada V, Piqueras P, Ponce MR, Micol JL (2006) Both abscisic acid (ABA)-dependent and ABA-independent pathways govern the induction of NCED3, AAO3 and ABA1 in response to salt stress. Plant, Cell Environ 29:2000–2008
Chen J, Burke JJ, Xin Z, Xu C, Velten J (2006) Characterization of the Arabidopsis thermosensitive mutant atts02 reveals an important role for galactolipids in thermotolerance. Plant, Cell Environ 29:1437–1448
Gombos Z, Várkonyi Z, Hagio M, Iwaki M, Kovács L, Masamoto K, Itoh S, Wada H (2002) Phosphatidylglycerol requirement for the function of electron acceptor plastoquinone Q(B) in the photosystem II reaction center. Biochemistry 41:3796–3802
Greenspan P, Fowler SD (1985) Spectrofluorometric studies of the lipid probe, nile red. J Lipid Res 26:781–789
Greenspan P, Mayer EP, Fowler SD (1985) Nile red: a selective fluorescent stain for intracellular lipid droplets. J Cell Biol 100:965–973
Hur J, Schlautman MA, Yim S (2004) Effects of organic ligands and pH on the leaching of copper from brake wear debris in model environmental solutions. J Environ Monit 6:89–94
Kanamaru K, Kashiwagi S, Mizuno T (1994) A copper-transporting P-type ATPase found in the thylakoid membrane of the cyanobacterium Synechococcus species PCC7942. Mol Microbiol 13:369–377
Kawaguchi R, Girke T, Bray EA, Bailey-Serres J (2004) Differential mRNA translation contributes to gene regulation under non-stress and dehydration stress conditions in Arabidopsis thaliana. Plant J 38:823–839
Koyama Y, Kaneko Y, Matsuoka S, Matsumoto K, Hara H, Ohta N (2012) Expression and localization of two SecA homologs in the unicellular red alga Cyanidioschyzon merolae. Biosci Biotechnol Biochem 76:417–422
Kruse O, Hankamer B, Konczak C, Gerle C, Morris E, Radunz A, Schmid GH, Barber J (2000) Phosphatidylglycerol is involved in the dimerization of photosystem II. J Biol Chem 275:6509–6514
Liu TW, Wu FH, Wang WH, Chen J, Li ZJ, Dong XJ, Patton J, Pei ZM, Zheng HL (2011) Effects of calcium on seed germination, seedling growth and photosynthesis of six forest tree species under simulated acid rain. Tree Physiol 31:402–413
Lu B, Xu C, Awai K, Jones AD, Benning C (2007) A small ATPase protein of Arabidopsis, TGD3, involved in chloroplast lipid import. J Biol Chem 282:35945–35953
Malinverni JC, Silhavy TJ (2009) An ABC transport system that maintains lipid asymmetry in the gram-negative outer membrane. Proc Natl Acad Sci USA 106:8009–8014
Nitta K, Nagayama K, Danev R, Kaneko Y (2009) Visualization of BrdU-labelled DNA in cyanobacterial cells by Hilbert differential contrast transmission electron microscopy. J Microsc 234:118–123
Ohta H, Shibata Y, Haseyama Y, Yoshino Y, Suzuki T, Kagasawa T, Kamei A, Ikeuchi M, Enami I (2005) Identification of genes expressed in response to acid stress in Synechocystis sp. PCC 6803 using DNA microarrays. Photosynth Res 84:225–230
Phung LT, Ajlani G, Haselkorn R (1994) P-type ATPase from the cyanobacterium Synechococcus 7942 related to the human Menkes and Wilson disease gene products. Proc Natl Acad Sci USA 91:9651–9654
Pinto FL, Thapper A, Sontheim W, Lindblad P (2009) Analysis of current and alternative phenol based RNA extraction methodologies for cyanobacteria. BMC Mol Biol 10:79
Pisareva T, Kwon J, Oh J, Kim S, Ge C, Wieslander A, Choi JS, Norling B (2011) Model for membrane organization and protein sorting in the cyanobacterium Synechocystis sp. PCC 6803 inferred from proteomics and multivariate sequence analyses. J Proteome Res 10:3617–3631
Sakurai I, Mizusawa N, Wada H, Sato N (2007) Digalactosyldiacylglycerol is required for stabilization of the oxygen-evolving complex in photosystem II. Plant Physiol 145:1361–1370
Sato N, Hagio M, Wada H, Tsuzuki M (2000) Requirement of phosphatidylglycerol for photosynthetic function in thylakoid membranes. Proc Natl Acad Sci U S A 97:10655–10660
Siaut M, Cuiné S, Cagnon C, Fessler B, Nguyen M, Carrier P, Beyly A, Beisson F, Triantaphylidès C, Li-Beisson Y, Peltier G (2011) Oil accumulation in the model green alga Chlamydomonas reinhardtii: characterization, variability between common laboratory strains and relationship with starch reserves. BMC Biotechnol 11:7
Sonoda M, Katoh H, Vermaas W, Schmetterer G, Ogawa T (1998) Photosynthetic electron transport involved in PxcA-dependent proton extrusion in Synechocystis sp. strain PCC6803: effect of pxcA inactivation on CO2, HCO3-, and NO3- uptake. J Bacteriol 180:3799–3803
Stanier RY, Kunisawa R, Mandel M, Cohen-Bazire G (1971) Purification and properties of unicellular blue-green algae (order Chroococcales). Bacteriol Rev 35:171–205
Tahara H, Uchiyama J, Yoshihara T, Matsumoto K, Ohta H (2012) Role of Slr 1045 in environmental stress tolerance and lipid transport in the cyanobacterium Synechocystis sp. PCC6803. Biochim Biophys Acta 1817:1360–1366
Takahashi Y, Nakamura M (1999) Functional assignment of the ORF2-iscS-iscU-iscA-hscB-hscA-fdx-ORF3 gene cluster involved in the assembly of Fe-S clusters in Escherichia coli. J Biochem 126:917–926
Tsunekawa K, Shijuku T, Hayashimoto M, Kojima Y, Onai K, Morishita M, Ishiura M, Kuroda T, Nakamura T, Kobayashi H, Sato M, Toyooka K, Matsuoka K, Omata T, Uozumi N (2009) Identification and characterization of the Na+/H+ antiporter NhaS3 from the thylakoid membrane of Synechocystis sp. PCC 6803. J Biol Chem 284:16513–16521
Uchiyama J, Asakura R, Kimura M, Moriyama A, Tahara H, Kobayashi Y, Kubo Y, Yoshihara T, Ohta H (2012) Slr0967 and Sll0939 induced by the SphR response regulator in Synechocystis sp. PCC 6803 are essential for growth under acid stress conditions. Biochim Biophys Acta 1817:1270–1276
Vasil’eva SV, Streltsova DA, Starostina IA, Sanina NA (2013) Nitrogen oxide is involved in the regulation of the Fe-S cluster assembly in proteins and the formation of biofilms by Escherichia coli cells. Izv Akad Nauk Ser Biol 4:398–404
Xu C, Fan J, Riekhof W, Froehlich JE, Benning C (2003) A permease-like protein involved in ER to thylakoid lipid transfer in Arabidopsis. EMBO J 22:2370–2379
Xu C, Fan J, Froehlich JE, Awai K, Benning C (2005) Mutation of the TGD1 chloroplast envelope protein affects phosphatidate metabolism in Arabidopsis. Plant Cell 17:3094–3110
Xu C, Fan J, Cornish AJ, Benning C (2008) Lipid trafficking between the endoplasmic reticulum and the plastid in Arabidopsis requires the extraplastidic TGD4 protein. Plant Cell 20:2190–2204
Yamauchi Y, Kaniya Y, Kaneko Y, Hihara Y (2011) Physiological roles of the cyAbrB transcriptional regulator pair Sll0822 and Sll0359 in Synechocystis sp. strain PCC 6803. J Bacteriol 193:3702–3709
Zhang X, Chen G, Qin C, Wang Y, Wei D (2012) Slr0643, an S2P homologue, is essential for acid acclimation in the cyanobacterium Synechocystis sp. PCC 6803. Microbiology 158:2765–2780
Acknowledgments
This study was supported by the Program for Development of Strategic Research Center in Private Universities, supported by MEXT (S1001020).
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
11120_2015_153_MOESM1_ESM.tif
Multiple sequence alignment of Slr1045, Slr1344, MlaE, and TGD1 protein. Conserved amino acid sequences are shown by black boxes. Asterisks indicate amino acid sequence pairs with the same sequence. Slr1045 and Slr1344 are more similar to TGD1 than to MlaE. Slr1045 has homology to MlaE (38 % identity and 57 % positive), which plays a role in the maintenance of outer membrane lipid asymmetry in E. coli, and TGD1 (53 % identity and 73 % positive), which is a phosphatidic acid transporter in A. thaliana. Slr1344 protein has homology to MlaE (32 % identity and 54 % positive) and TGD1 (50 % identity and 71 % positive). These proteins may have the same function (TIFF 85227 kb)
11120_2015_153_MOESM2_ESM.tif
Construction of slr1344, sll1001/sll1002, sll0751, and sll1041 mutant cells. PCR analyses using chromosomal DNA template for WT and mutant genes: slr1344 (a), sll1002 and sll1001 (b), sll0751 (c), and sll1041 (d). Primers specific for each gene were used to confirm chromosomal DNA disruption in mutants (TIFF 40575 kb)
11120_2015_153_MOESM3_ESM.tif
Histogram of observed bright spots. WT, Δslr1045, Δslr1344, Δsll1001/sll1002, Δsll0751, and Δsll1041 cells were cultivated on BG-11 medium (pH 8.0) for 7 days at 30 °C. Stained cells were then observed by fluorescence microscopy with optical filters. The number of bright spots in cells was counted. (TIFF 20078 kb)
Rights and permissions
About this article
Cite this article
Tahara, H., Matsuhashi, A., Uchiyama, J. et al. Sll0751 and Sll1041 are involved in acid stress tolerance in Synechocystis sp. PCC 6803. Photosynth Res 125, 233–242 (2015). https://doi.org/10.1007/s11120-015-0153-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11120-015-0153-6