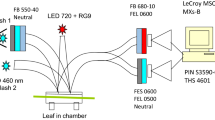
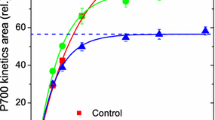
Abstract
Oxygen evolution was measured in sunflower leaves in steady-state and during multiple-turnover pulses (MTP) of different light (630 nm LED plus far-red light) intensity and duration. In parallel, Chl fluorescence yields F 0 (minimum), F s (steady-state), and F m (pulse-saturated), as well as fluorescence induction during MTPs were recorded. Extra O2 evolution was measured in response to a saturating single-turnover Xe flash (STF) applied immediately subsequently to the actinic light in the steady-state and to each MTP. Under the used anaerobic conditions and randomized S-states electron transport per STF was calculated as 4O2 evolution. The STF-induced electron transport (=the number of open PSII) was maximal at the low background light, but decreased with progressing light saturation in steady-state and with the increasing duration of MTP. The quantum yield (effective antenna size) of open PSII centers remained constant when adjacent centers became closed. The photochemical quenching of fluorescence q P = (F m − F s)/(F m − F 0) was proportional with the portion of open PSII centers in the steady-state (variable non-photochemical quenching, NPQ) and with increasing MTP duration (NPQ absent). Comparison of experimental responses to a model based on PSII dimers with well-connected antennae showed no energetic connectivity between PSII antennae in intact leaves, suggesting that in vivo PSII exist as monomers, or dimers with energetically disconnected antennae.











Similar content being viewed by others
Abbreviations
- DCMU :
-
3-(3,4-Dichlorophenyl)-1,1-dimethylurea
- ETR:
-
Electron transport rate
- FI:
-
Fluorescence induction
- FR:
-
Far-red
- F m, F s, F 0 :
-
Fluorescence yield in the light, maximum, steady-state and minimum
- LED:
-
Light-emitting diode
- MTP:
-
Multi-turnover light pulse
- NPQ:
-
Non-photochemical quenching
- PAD:
-
Photon absorption density
- PFD:
-
Photon flux density, incident
- PSII, PSI:
-
Photosystems, II and I
- PQ, PQH2 :
-
Plastoquinone, oxidized and reduced
- QA, QB :
-
PSII acceptors, primary and secondary
- STF:
-
Single-turnover flash
References
Buser CA, Diner BA, Brudvig GW (1992) Photooxidation of cytochrome b559 in oxygen-evolving photosystem II. Biochemistry 31:11449–11459
Den Hollander WTF, Bakker JGC, van Grondelle R (1983) Trapping, loss and annihilation of excitations in a photosynthetic system. I. Theoretical aspects. Biochim Biophys Acta 725:492–507
Diner BA, Rappaport F (2002) Structure, dynamics, and energetics of the primary photochemistry of photosystem II of oxygenix photosynthesis. Annu Rev Plant Biol 53:551–580
Doschek WW, Kok B (1972) Photon trapping in photosystem II of photosynthesis. Biophys J 12:832–838
France LL, Geacintov NE, Breton J, Valkunas L (1992) The dependence of the degrees of sigmoidicities of fluorescence induction curves in spinach chloroplasts on the duration of actinic pulses in pump-probe experiments. Biochim Biophys Acta 1101:105–119
Garab G (1992) Macrodomain organization of complexes in the thylakoid membranes. Structural and regulatory roles. Conclusions from macroscopic and microscopic circular dichroism of chloroplast thylakoid membranes and aggregates of LHCII. In: Murata N (ed) Research in Photosynthesis, vol I. Kluwer, Dordrecht, pp 171–178
Genty B, Briantais JM, Baker NR (1989) The relationship between quantum yield of photosynthetic electron transport and quenching of chlorophyll fluorescence. Biochim Biophys Acta 990:87–92
Hodges M, Moya I (1987) Modification of room-temperature picosecond chlorophyll fluorescence kinetics in photosystem-II-enriched particles by photochemistry. Biochim Biophys Acta 892:42–47
Jahns P, Trissl H-W (1997) Indications for a dimeric organization of the antenna-depleted reaction center core of photosystem II in thylakoids of intermittent light grown pea plants. Biochim Biophys Acta 1318:1–5
Johnson DM, Smith WK, Vogelmann TC, Brodersen CR (2005) Leaf architecture and direction of incident light influence mesophyll fluorescence profiles. Am J Bot 92(9):1425–1431
Joliot A, Joliot P (1964) Étude cinétique de la réaction photochimique libérant l’oxygène au cours de la photosynthése. C R Acad Sci Paris 258:4622–4625
Joliot P, Joliot A (2003) Excitation transfer between photosynthetic units: the 1964 experiment. Photosynth Res 76:241–245
Joliot P, Bennoun P, Joliot A (1973) New evidence supporting energy transfer between photosynthetic units. Biochim Biophys Acta 305:317–328
Joliot P, Lavergne J, Béal D (1992) Plastoquinone compartmentation in chloroplasts. I. Evidence for domains with different rates of photo-reduction. Biochim Biophys Acta 1101:1–12
Kaminskaya O, Shuvalov VA, Renger G (2007) Evidence for a novel quinone-binding site in the photosystem II (PS II) complex that regulates the redox potential of cytochrome b559. Biochemistry 46:1091–1105
Kirchhoff H, Horstmann S, Weis E (2000) Control of the photosynthetic electron transport by PQ diffusion microdomains in thylakoids of higher plants. Biochim Biophys Acta 1459:148–168
Kramer DM, Johnson G, Kiirats O, Edwards GE (2004) New fluorescence parameters for the determination of QA redox state and excitation energy fluxes. Photosynth Res 79:209–218
Laisk A, Oja V (1976) Adaptation of the photosynthetic apparatus to light profile in the leaf. Fiziologija Rastenij. Sov Plant Physiol 23(3):445–451 (in Russian)
Laisk A, Oja V (1998) Dynamic gas exchange of leaf photosynthesis. Measurement and interpretation. CSIRO, Canberra
Laisk A, Oja V (2000a) Alteration of PSII properties with non-photochemical excitation quenching. Phil Trans R Soc Lond B 355:1405–1418
Laisk A, Oja V (2000b) Electron transport through photosystem II in leaves during light pulses: acceptor resistance increases with nonphotochemical excitation quenching. Biochim Biophys Acta 1460:255–267
Laisk A, Oja V, Rasulov B, Eichelmann H, Sumberg A (1997) Quantum yields and rate constants of photochemical and nonphotochemical excitation quenching. Experiment and model. Plant Physiol 115:803–815
Laisk A, Oja V, Rasulov B, Rämma H, Eichelmann H, Kasparova I, Pettai H, Padu E, Vapaavuori E (2002) A computer-operated routine of gas exchange and optical measurements to diagnose photosynthetic apparatus in leaves. Plant Cell Environ 25:923–943
Laisk A, Eichelmann H and Oja V (2012) Oxygen evolution and chlorophyll fluorescence from multiple turnover light pulses: charge recombination in photosystem II in sunflower leaves. Photosynth Res. doi: 10.1007/s11120-012-9751-8
Lavergne J, Trissl H-W (1995) Theory of fluorescence induction in photosystem II: derivation of analytical expressions in a model including exciton-radical-pair equilibrium and restricted energy transfer between photosynthetic units. Biophys J 68:2474–2492
Lazár D (1999) Chlorophyll a fluorescence induction. Biochim Biophys Acta 1412:1–28
Lazár D and Schansker G (2009) Models of chlorophyll a fluorescence transients. In: Laisk A, Nedbal L, Govindjee (eds) Photosynthesis in silico. Understanding complexity from molecules to ecosystems, Springer Science + Business Media, Dordrecht, pp 85–123
Ley AC, Mauzerall DC (1982) Absolute absorption cross-sections for photosystem II and the minimum quantum requirement for photosynthesis in Chlorella vulgaris. Biochim Biophys Acta 680:95–106
Ley AC, Mauzerall DC (1986) The extent of energy transfer among photosystem II reaction centers in chlorella. Biochim Biophys Acta 850:234–248
Melis A, Duysens LNM (1979) Biphasic energy conversion kinetics and absorbance difference spectra of photosystem II of chloroplasts. Evidence for two different photosystem II reaction centers. Photochem Photobiol 29:373–382
Melis A, Homann PH (1975) Kinetic analysis of fluorescence induction in 3-(3,4-dichlorophenyl)-1,1-dimethylurea poisoned chloroplasts. Photochem Photobiol 21:431–437
Melis A, Homann PH (1976) Heterogeneity of the photochemical centers in system II of chloroplasts. Photochem Photobiol 23:343–350
Moya I, Hodges M, Barbet J-C (1986) Modification of room-temperature picosecond chlorophyll fluorescence kinetics in green algae by photosystem II trap closure. FEBS Lett 198:256–262
Oguchi R, Douwstra P, Fujita T, Chow WS, Terashima I (2011) Intra-leaf gradients of photoinhibition induced by different color lights: implications for the dual mechanisms of photoinhibition and for the application of conventional chlorophyll fluorometers. New Phytol 191:146–159
Oja V, Laisk A (2000) Oxygen yield from single turnover flashes in leaves: non-photochemical excitation quenching and the number of active PSII. Biochim Biophys Acta 1460:291–301
Oja V, Eichelmann H, Anijalg A, Rämma H, Laisk A (2010) Equilibrium or disequilibrium? A dual-wavelength investigation of photosystem I donors. Photosynth Res 103:153–166
Oja V, Eichelmann H, Laisk A (2011) Oxygen evolution from single- and multiple-turnover light pulses: temporal kinetics of electron transport through PSII in sunflower leaves. Photosynth Res 110:99–109
Paillotin G (1976) Movement of excitations in the photosynthetic domains of photosystem II. J Theor Biol 58:237–252
Peterson R, Oja V, Laisk A (2001) Chlorophyll fluorescence at 680 and 730 nm and its relationship to photosynthesis. Photosynth Res 70:185–196
Pettai H, Oja V, Freiberg A, Laisk A (2005) Photosynthetic activity of far-red light in green plants. Biochim Biophys Acta 1708:311–321
Pfündel E (1998) Estimating the contribution of photosystem I to total leaf chlorophyll fluorescence. Photosynth Res 56:185–195
Rappaport F, Beal D, Joliot A, Joliot P (2007) On the advantages of using green light to study fluorescence yield changes in leaves. Biochim Biophys Acta 1767:56–65
Schmuk G, Moya I (1994) Time-resolved chlorophyll fluorescence spectra of intact leaves. Remote Sens Environ 47:72–76
Strasser R, Tsimili-Michael M, Srivastava A (2004) Analysis of the chlorophyll a fluorescence transient. In: Papageorgiou GC, Govindjee (eds) Chlorophyll a fluorescence. A signature of photosynthesis, Springer, Dordrecht, pp 321–362
Takahashi T, Inoue-Kashino N, Ozawa S-I, Takahashi Y, Kashino Y (2009) Photosystem II complex in vivo is a monomer. J Biol Chem 284:15598–15606
Vogelmann TC, Evans JR (2002) Profiles of light absorption and chlorophyll within spinach leaves from chlorophyll fluorescence. Plant Cell Environ 25:1313–1323
Vredenberg VJ (2008) Algorithm for analysis of OJDIP fluorescence induction curves in terms of photo- and electrochemical events in photosystems of plant cells. Derivation and application. J Photochem Photobiol B 91:58–65
Vredenberg W, Durchan M, Prášil O (2009) Photochemical and photoelectrochemical quenching of chlorophyll fluorescence in photosystem II. Biochim Biophys Acta 1787:1468–1478
Wendler J, Holzwarth AR (1987) State transitions in the green alga Scenedesmus obliquus probed by time-resolved chlorophyll fluorescence spectroscopy and global data analysis. Biophys J 52:717–728
Whitmarsh J, Pakrasi HB (1996) Form and function of cytochrome b-559. In: Ort DR, Yocum CF (eds) Oxygenic photosynthesis: the light reactions. Kluwer, The Netherlands, pp 249–264
Acknowledgments
This study was supported by Targeted Financing Theme SF0180045s08 from Estonian Ministry of Education and Science and Grants 8283 and 8344 from Estonian Science Foundation. We appreciate the contribution by D. M. Kramer, Michigan State University, during the planning phase of this study. We also thank the two reviewers, whose active participation has resulted in major developments in this study, strongly substantiating the conclusion.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Oja, V., Laisk, A. Photosystem II antennae are not energetically connected: evidence based on flash-induced O2 evolution and chlorophyll fluorescence in sunflower leaves. Photosynth Res 114, 15–28 (2012). https://doi.org/10.1007/s11120-012-9775-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11120-012-9775-0