Abstract
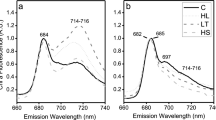
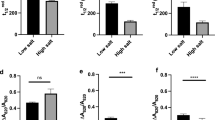
Chlamydomonas raudensis Ettl UWO241, a natural variant of C. raudensis, is deficient in state transitions. Its habitat, the deepest layer of Lake Bonney in Antarctica, features low irradiance, low temperature, and high salinity. Although psychrophily and low-light acclimation of this green alga has been described, very little information is available on the effect of salinity. Here, we demonstrate that this psychrophile is halotolerant, not halophilic, and it shows energy redistribution between photosystem I and II based on energy spillover under low-salt conditions. Furthermore, we revealed that C. raudensis exhibits higher non-photochemical quenching in comparison with the mesophile Chlamydomonas reinhardtii, when grown with low-salt, which is due to the lower proton conductivity across the thylakoid membrane. Significance of the C. raudensis UWO241 traits found in the low salinity culture are implicated with their natural habitats, including the high salinity and extremely stable light environments.







Similar content being viewed by others
Abbreviations
- F m :
-
Maximal yield of fluorescence.
- Fm′:
-
Quenched maximal yield of fluorescence
- LHC:
-
Light-harvesting complexes
- NPQ:
-
Non-photochemical quenching
- PQ:
-
Plastoquinone
- PSI and PSII:
-
Photosystem I and II
References
Allen JF, Bennett J, Steinback KE, Arntzen CJ (1981) Chloroplast protein phosphorylation couples plastoquinone redox state to distribution of excitation energy between photosystems. Nature 291:25–29
Avenson TJ, Cruz JA, Kramer DM (2004) Modulation of energy-dependent quenching of excitons in antennae of higher plants. Proc Natl Acad Sci USA 101:5530–5535
Bulté L, Wollman F-A (1992) Evidence for a selective destabilization of an integral membrane protein, the cytochrome b 6/f complex, during gametogenesis in Chlamydomonas reinhardtii. Eur J Biochem 204:327–336
Casper-Lindley C, Björkman O (1996) Nigericin insensitive post-illumination reduction in fluorescence yield in Dunaliella tertiolecta (chlorophyte). Photosynth Res 50:209–222
Cruz JA, Avenson TJ, Kanazawa A, Takizawa K, Edwards GE, Kramer DM (2005) Plasticity in light reactions of photosynthesis for energy production and photoprotection. J Exp Bot 56:395–406
Delosme R, Olive J, Wollman F-A (1996) Changes in light energy distribution upon state transitions: an in vivo photoacoustic study of the wild type and photosynthesis mutants from Chlamydomonas reinhardtii. Biochim Biophys Acta 1273:150–158
Depège N, Bellafiore S, Rochaix J-D (2003) Role of chloroplast protein kinase Stt7 in LHCII phosphorylation and state transition in Chlamydomonas. Science 299:1572–1575
Finazzi G, Rappaport F, Furia A, Fleischmann M, Rochaix J-D, Zito F, Forti G (2002) Involvement of state transitions in the switch between linear and cyclic electron flow in Chlamydomonas reinhardtii. EMBO Rep 3:280–285
Gans P, Rebeille F (1990) Control in the dark of the plastoquinone redox state by mitochondrial activity in Chlamydomonas reinhardtii. Biochim Biophys Acta 1015:150–155
Genty B, Briantais J-M, Baker NR (1989) The relationship between the quantum yield of photosynthetic electron transport and quenching of chlorophyll fluorescence. Biochim Biophys Acta 990:87–92
Gudynaite-Savitch L, Gretes M, Morgan-Kiss RM, Savitch LV, Simmonds J, Kohalmi SE, Huner NP (2006) Cytochrome f from the Antarctic psychrophile, Chlamydomonas raudensis UWO 241: structure, sequence, and complementation in the mesophile, Chlamydomonas reinhardtii. Mol Genet Genomics 275:387–398
Gudynaite-Savitch L, Loiselay C, Savitch LV, Simmonds J, Kohalmi SE, Choquet Y, Huner NP (2007) The small domain of cytochrome f from the psychrophile Chlamydomonas raudensis UWO 241 modulates the apparent molecular mass and decreases the accumulation of cytochrome f in the mesophile Chlamydomonas reinhardtii. Biochem Cell Biol 85:616–627
Iwai M, Kato N, Minagawa J (2007) Distinct physiological responses to a high light and low CO2 environment revealed by fluorescence quenching in photoautotrophically grown Chlamydomonas reinhardtii. Photosynth Res 94:307–314
Kanazawa A, Kramer DM (2002) In vivo modulation of nonphotochemical exciton quenching (NPQ) by regulation of the chloroplast ATP synthase. Proc Natl Acad Sci USA 99:12789–12794
Kitajima M, Butler WL (1975) Quenching of chlorophyll fluorescence and primary photochemistry in chloroplasts by dibromothymoquinone. Biochim Biophys Acta 376:105–115
Kramer DM, Avenson TJ, Edwards GE (2004a) Dynamic flexibility in the light reactions of photosynthesis governed by both electron and proton transfer reactions. Trends Plant Sci 9:349–357
Kramer DM, Johnson G, Kiirats O, Edwards GE (2004b) New fluorescence parameters for the determination of QA redox state and excitation energy fluxes. Photosynth Res 79:209–218
Krause GH, Weis E (1991) Chlorophyll fluorescence and photosynthesis: the basics. Annu Rev Plant Physiol Plant Mol Biol 42:313–349
Lizotte MP, Priscu JC (1994) Natural fluorescence and quantum yields in vertically stationary phytoplankton from perennially ice-covered lakes. Limnol Oceanogr 39:1399–1410
Morgan RM, Ivanov AG, Priscu JC, Maxwell DP, Huner NPA (1998) Structure and composition of the photochemical apparatus of the antarctic green alga, Chlamydomonas subcaudata. Photosynth Res 56:303–314
Morgan-Kiss R, Ivanov AG, Williams J, Mobashsher K, Huner NP (2002a) Differential thermal effects on the energy distribution between photosystem II and photosystem I in thylakoid membranes of a psychrophilic and a mesophilic alga. Biochim Biophys Acta 1561:251–265
Morgan-Kiss RM, Ivanov AG, Huner NP (2002b) The Antarctic psychrophile, Chlamydomonas subcaudata, is deficient in state I-state II transitions. Planta 214:435–445
Morgan-Kiss RM, Ivanov AG, Pocock T, Krol M, Gudynaite-Savitch L, Huner NPA (2005) The antarctic psychrophile, Chlamydomonas raudensis Ettl (UWO241) (Chlorophyceae, Chlorophyta), exhibits a limited capacity to photoacclimate to red light. J Phycol 41:791–800
Morgan-Kiss RM, Priscu JC, Pocock T, Gudynaite-Savitch L, Huner NP (2006) Adaptation and acclimation of photosynthetic microorganisms to permanently cold environments. Microbiol Mol Biol Rev 70:222–252
Neale PJ, Priscu JC (1995) The photosynthetic apparatus of phytoplankton from a perennially ice-covered antarctic lake: acclimation to an extreme shade environment. Plant Cell Physiol 36:253–263
Pocock T, Lachance M-A, Proschold T, Priscu JC, Kim SS, Huner NPA (2004) Identification of a psychrophilic green alga from Lake Bonney Antarctica: Chlamydomonas raudensis Ettl. (UWO241) Chlorophyceae. J Phycol 40:1138–1148
Sacksteder CA, Kramer DM (2000) Dark-interval relaxation kinetics (DIRK) of absorbance changes as a quantitative probe of steady-state electron transfer. Photosynth Res 66:145–158
Sacksteder CA, Jacoby ME, Kramer DM (2001) A portable, non-focusing optics spectrophotometer (NoFOSpec) for measurements of steady-state absorbance changes in intact plants. Photosynth Res 70:231–240
Stein JR (1973) Culture methods and growth measurements. In: Stein JR (ed) Handbook of phycological methods. Cambridge University Press, Cambridge, UK, p 448
Sueoka N (1960) Mitotic replication of deoxyribonucleic acid in Chlamydomonas reinhardti. Proc Natl Acad Sci USA 46:83–91
Szyszka B, Ivanov AG, Huner NP (2007) Psychrophily is associated with differential energy partitioning, photosystem stoichiometry and polypeptide phosphorylation in Chlamydomonas raudensis. Biochim Biophys Acta 1767:789–800
Takahashi H, Iwai M, Takahashi Y, Minagawa J (2006) Identification of the mobile light-harvesting complex II polypeptides for state transitions in Chlamydomonas reinhardtii. Proc Natl Acad Sci USA 103:477–482
Takizawa K, Cruz JA, Kanazawa A, Kramer DM (2007) The thylakoid proton motive force in vivo. Quantitative, non-invasive probes, energetics, and regulatory consequences of light-induced pmf. Biochim Biophys Acta 1767:1233–1244
Takizawa K, Kanazawa A, Kramer DM (2008) Depletion of stromal Pi induces high ‘energy-dependent’ antenna exciton quenching (qE) by decreasing proton conductivity at CFO-CF1 ATP synthase. Plant Cell Environ 31:235–243
Weis E, Berry JA (1987) Quantum efficiency of photosystem II in relation to ‘energy’-dependent quenching of chlorophyll fluorescence. Biochim Biophys Acta 894:198–208
Witt HT, Zickler A (1973) Electrical evidence for the field indicating absorption change in bioenergetic membranes. FEBS Lett 37:307–310
Wollman F-A (2001) State transitions reveal the dynamics and flexibility of the photosynthetic apparatus. EMBO J 20:3623–3630
Zito F, Finazzi G, Delosme R, Nitschke W, Picot D, Wollman F-A (1999) The Qo site of cytochrome b 6 f complexes controls the activation of the LHCII kinase. EMBO J 18:2961–2969
Acknowledgments
This work was supported in part by Grants-in-Aid for Scientific Research to J.M. from the Ministry of Education, Culture, Sports, Science and Technology (No. 18GS0318) and to N.P.A.H. from the Natural Sciences and Engineering Research Council of Canada.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Takizawa, K., Takahashi, S., Hüner, N.P.A. et al. Salinity affects the photoacclimation of Chlamydomonas raudensis Ettl UWO241. Photosynth Res 99, 195–203 (2009). https://doi.org/10.1007/s11120-008-9397-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11120-008-9397-8