Abstract
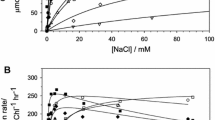
Extraction of Ca2+ from the O2-evolving complex (OEC) of photosystem II (PSII) membranes with 2 M NaCl in the light (PSII(–Ca/NaCl)) results in 90% inhibition of the O2-evolution reaction. However, electron transfer from the donor to acceptor side of PSII, measured as the reduction of the exogenous acceptor 2,6-dichlorophenolindophenol (DCIP) under continuous light, is inhibited by only 30%. Thus, calcium extraction from the OEC inhibits the synthesis of molecular O2 but not the oxidation of a substrate we term X, the source of electrons for DCIP reduction. The presence of electron transfer across PSII(–Ca/NaCl) membranes was demonstrated using fluorescence induction kinetics, a method that does not require an artificial acceptor. The calcium chelator, EGTA (5 mM), when added to PSII(–Ca/NaCl) membranes, does not affect the inhibition of O2 evolution by NaCl but does inhibit DCIP reduction up to 92% (the reason why electron transport in Ca2+-depleted materials has not been noticed before). Another chelator, sodium citrate (citrate/low pH method of calcium extraction), also inhibits both O2 evolution and DCIP reduction. The role of all buffer components (including bicarbonate and sucrose) as possible sources of electrons for PSII(–Ca/NaCl) membranes was investigated, but only the absence of chloride anions strongly inhibited the rate of DCIP reduction. Substitution of other anions for chloride indicates that Cl− serves its well-known role as an OEC cofactor, but it is not substrate X. Multiple turnover flash experiments have shown a period of four oscillations of the fluorescence yield (both the maximum level, F max, and the fluorescence level measured 50 s after an actinic flash in the presence of DCMU) in native PSII membranes, reflecting the normal function of the OEC, but the absence of oscillations in PSII(–Ca/NaCl) samples. Thus, PSII(–Ca/NaCl) samples do not evolve O2 but do transfer electrons from the donor to acceptor sides and exhibit a disrupted S-state cycle. We explain these results as follows. In Ca2+-depleted PSII membranes, obtained without chelators, the oxidation of the OEC stops after the absorption of three quanta of light (from the S1 state), which should convert the native OEC to the S4 state. An one-electron oxidation of the water molecule bound to the Mn cluster then occurs (the second substrate water molecule is absent due to the absence of calcium), and the OEC returns to the S3 state. The appearance of a sub-cycle within the S-state cycle between S3-like and S4-like states supplies electrons (substrate X is postulated to be OH−), explains the absence of O2 production, and results in the absence of a period of four oscillation of the normal functional parameters, such as the fluorescence yield or the EPR signal from S2. Chloride anions probably keep the redox potential of the Mn cluster low enough for its oxidation by Y •Z .







Similar content being viewed by others
Abbreviations
- Chl:
-
Chlorophyll
- DCMU:
-
3-(3,4-Dichlorophenyl)-1,1-dimethylurea
- DCIP:
-
2,6-Dichlorophenolindophenol
- EGTA:
-
Ethylene glycol-bis(β-aminoethyl ether)-N,N,N′,N′-tetraacetic acid
- EDTA:
-
Ethylenediaminetetraacetic acid
- F o :
-
Minimal fluorescence of “open” reaction centers in dark-adapted samples
- F max :
-
Maximum fluorescence yield
- FIK:
-
Fluorescence induction kinetics
- FYD:
-
Pulse-probe fluorescence yield decay kinetics
- MES:
-
2-(N-morpholino)ethanesulfonic acid
- OEC:
-
Oxygen-evolving complex
- PSII:
-
Photosystem II
- PSII(–Mn):
-
Mn-depleted photosystem II
- PSII(–Ca/NaCl):
-
Photosystem II with Ca2+ extracted by treatment with 2 M NaCl
- PSII(–Ca/NaCl + 50 μM EGTA):
-
Photosystem II with Ca2+ extracted by treatment with 2 M NaCl in the presence of 50 μM EGTA
- PSII(–Ca/NaCl + 5 mM EGTA):
-
Photosystem II with Ca2+ extracted by treatment with 2 M NaCl in the presence of 5 mM EGTA
- PSII(–Ca/NaCl + 5 mM EGTA→ –EGTA):
-
Photosystem II with Ca2+ extracted by treatment with 2 M NaCl in the presence of 5 mM EGTA and subsequent washing free of EGTA
- PSII(–Ca/pH 3.0):
-
Photosystem II with Ca2+ extracted by treatment with citrate buffer (pH 3.0)
- YZ :
-
Tyrosine 161 on the D1 protein in spinach, the first electron donor to P680+ in PSII
References
Ananyev G, Wydrzynski T, Renger G, Klimov V (1992) Transeint peroxide formation by the manganese-containing, redox-active donor side of photosystem II upon inhibition of O2 evolution with lauroylchloline chloride. Biochim Biophys Acta 1100:303–311
Andréasson L-E, Vass I, Styring S (1995) Ca2+ depletion modifies the electron transfer on both donor and acceptor sides in photosystem II from spinach. Biochim Biophys Acta 1230:155–164. doi:10.1016/0005-2728(95)00047-M
Armstrong JM (1964) The molar extinction coefficient of 2,6-dichlorophenol indophenol. Biochim Biophys Acta 86:194–197
Arnon DJ (1949) Copper enzymes in isolated chloroplast: polyphenol oxidase in Beta vulgaris. Plant Physiol 24:1–15
Baumgarten M, Philo JS, Dismukes GC (1990) Mechanism of photoinhibition of photosynthetic water oxidation by chloride depletion and fluoride substitution: oxidation of a protein residue. Biochemistry 29:10814–10822. doi:10.1021/bi00500a014
Blough NV, Sauer K (1984) The effects of mono- and divalent salts on the O2-evolution activity and low temperature multiline EPR spectrum of photosystem II preparations from spinach. Biochim Biophys Acta 767:377–381. doi:10.1016/0005-2728(84)90208-1
Blubaugh DJ, Cheniae GM (1990) Kinetics of photoinhibition in hydroxylamine-extracted photosystem II membranes: relevance to photoactivation and sites of electron donation. Biochemistry 29:5109–5118. doi:10.1021/bi00473a016
Boussac A, Rutherford AW (1988) Nature of the inhibition of the oxygen-evolving enzyme of photosystem II induced by sodium chloride washing and reversed by the addition of calcium(2+) or strontium(2+). Biochemistry 27:3476–3483. doi:10.1021/bi00409a052
Boussac A, Rutherford AW (1992) The involvement of Ca2+ in the Ca2+-effect on Photosystem-II oxygen evolution. Photosynth Res 32:207–209. doi:10.1007/BF00034796
Boussac A, Rutherford AW (1994) Electron transfer events in chloride-depleted photosystem II. J Biol Chem 269:12462–12467
Boussac A, Maison-Peteri B, Vernotte C, Etienne AL (1985) The charge accumulation mechanism in NaCl-washed and in Ca2+-reactivated Photosystem-II particles. Biochim Biophys Acta 808:225–230. doi:10.1016/0005-2728(85)90003-9
Boussac A, Zimmermann J-L, Rutherford AW (1989) EPR signals from modified charge accumulation states of the oxygen evolving enzyme in Ca2+-deficient photosystem II. Biochemistry 28:8984–8989. doi:10.1021/bi00449a005
Boussac A, Zimmermann JL, Rutherford AW (1990a) Factors influencing the formation of modified S2 EPR signal and the S3 EPR signal in Ca(2+)-depleted photosystem II. FEBS Lett 277:69–74. doi:10.1016/0014-5793(90)80811-V
Boussac A, Zimmermann JL, Rutherford AW, Lavergne J (1990b) Histidine oxidation in the oxygen-evolving photosystem-II enzyme. Nature 347:303–306. doi:10.1038/347303a0
Boussac A, Sètif P, Rutherford AW (1992) Inhibition of tyrosine Z photooxidation after formation of the S3-state in calcium-depleted and chloride-depleted photosystem-II. Biochemistry 31:1224–1234. doi:10.1021/bi00119a036
Bryson DJ, Doctor N, Johnson R, Baranov S, Haddy A (2005) Characteristics of iodide activation and inhibition of oxygen evolution by photosystem II. Biochemistry 44:7354–7360. doi:10.1021/bi047475d
Cammarata KV, Cheniae GM (1987) Studies on 17,24 kD depleted photosystem II membranes I. Evidences for high and low affinity calcium sites in 17,24 kD depleted PSII membranes from wheat versus spinach. Plant Physiol 84:587–595
Chu H-A, Nguyen AP, Debus RJ (1995) Amino acid residues that influence the binding of manganese or calcium to photosystem II. 1. The lumenal interhelical domains of the D1 polypeptide. Biochemistry 34:5839–5858. doi:10.1021/bi00017a016
Debus RJ (1992) The manganese and calcium ions of photosynthetic oxygen evolution. Biochim Biophys Acta 1102:269–352. doi:10.1016/0005-2728(92)90133-M
Ghanotakis DF, Babcock GT, Yocum CF (1984a) Calcium reconstitutes high rates of oxygen evolution in polypeptide depleted photosystem II preparations. FEBS Lett 167:127–130. doi:10.1016/0014-5793(84)80846-7
Ghanotakis DF, Babckock GT, Yocum CF (1984b) Water-soluble 17 and 23 kDa polypeptides restore oxygen evolution activity by creating a high-affinity binding site for Ca2+ the oxidizing side of photosystem II. FEBS Lett 170:169–173. doi:10.1016/0014-5793(84)81393-9
Ghirardi ML, Lutton TW, Seibert M (1996) Interactions between diphenylcarbazide, zinc, cobalt, and manganese on the oxidizing side of photosystem II. Biochemistry 35:1820–1828. doi:10.1021/bi951657d
Haddy A (2007) EPR spectroscopy of the manganese clusters of photosystem II. Photosynth Res 92:357–368. doi:10.1007/s11120-007-9194-9
Hallahan BJ, Nugent JHA, Warden JT, Evans MCW (1992) Investigation of the origin of the “S3” EPR signal from the oxygen-evolving complex of photosystem 2: the role of tyrosine Z. Biochemistry 31:4562–4573. doi:10.1021/bi00134a005
Halverson KM, Barry BA (2003) Sucrose and glycerol effects on photosystem II. Biophys J 85:1317–1325
Hasegawa K, Kimura Y, Ono T (2002) Chloride cofactor in the photosynthetic oxygen-evolving complex studied by Fourier transform infrared spectroscopy. Biochemistry 41:13839–13850. doi:10.1021/bi026595n
Haumann M, Junge W (1999) Evidence for empaired hydrogen-bonding of tyrosine Yz in calcium-depleted photosystem II. Biochim Biophys Acta 1411:121–133. doi:10.1016/S0005-2728(99)00045-6
Hendry G, Wydrzynski T (2003) 18O isotope exchange measurements reveal that calcium is involved in the binding of one substrate-water molecule to the oxygen-evolving complex in photosystem II. Biochemistry 42:6209–6217. doi:10.1021/bi034279i
Homann PH (1988) Chloride relations of photosystem II membrane preparations depleted of, and resupplied with, their 17 and 23 kDa extrinsic polypeptides. Photosynth Res 15:205–220. doi:10.1007/BF00047353
Homann PH (1993) Thermoluminescence properties of the S2-state in chloride-depleted water oxidizing complexes after reconstituting treatments with various monovalent anions. Photosynth Res 38:395–400. doi:10.1007/BF00046766
Jajoo A, Bharti S, Kawamori A (2005) EPR characteristics of chloride-depleted photosystem II membranes in the presence of other anions. Photochem Photobiol Sci 4:459–462. doi:10.1039/b414849e
Lazar D (1999) Chlorophyll a fluorescence induction. Biochim Biophys Acta 1412:1–28. doi:10.1016/S0005-2728(99)00047-X
Lindberg K, Andréasson LE (1996) A one-site, two-state model for the binding of anions in photosystem II. Biochemistry 35:14259–14267. doi:10.1021/bi961244s
Loll B, Kern J, Saenger W, Zouni A, Biesiadka J (2005) Towards complete cofactor arrangement in the 3.0 A resolution structure of photosystem II. Nature 438:1040–1044. doi:10.1038/nature04224
McEvoy JP, Brudvig GW (2006) Water-splitting chemistry of photosystem II. Chem Rev 106:4455–4482. doi:10.1021/cr0204294
McEvoy JP, Garcon JA, Batista VS, Brudvig GW (2005) Mechanism of photosynthetic water splitting. Photochem Photobiol Sci 4:940–949. doi:10.1039/b506755c
Metz JG, Nixon PJ, Rögner M, Brudwig GW, Diner BA (1989) Directed alteration of the D1 polypeptide of photosystem II: evidence that tyrosine-161 is the redox component, Z, connecting the oxygen-evolving complex to the primary electron donor, P680. Biochemistry 28:6960–6969. doi:10.1021/bi00443a028
Miqyass M, van Gorkom HJ, Yocum CF (2007) The PSII calcium site revisited. Photosynth Res 92:275–287. doi:10.1007/s11120-006-9124-2
Miyao M, Murata N (1984) Calcium ions can be substituted for the 24-kDa polypeptide in photosynthetic oxygen evolution. FEBS Lett 168:118–120. doi:10.1016/0014-5793(84)80218-5
Miyao M, Murata N (1985) The Cl− effect on photosynthetic oxygen evolution: interaction of Cl− with 18-kDa, 24-kDa and 33-kDa proteins. FEBS Lett 180:303–308. doi:10.1016/0014-5793(85)81091-7
Nakatani HY (1984) Photosynthetic oxygen evolution does not require the participation of polypeptides of 16 and 24 kilodaltons. Biochem Biophys Res Commun 120:299–304. doi:10.1016/0006-291X(84)91448-7
Nelson N, Yocum CF (2006) Structure and function of photosystems I and II. Annu Rev Plant Biol 57:521–565. doi:10.1146/annurev.arplant.57.032905.105350
Olesen K, Andréasson LE (2003) The function of the chloride ion in photosynthetic oxygen evolution. Biochemistry 42:2025–2035. doi:10.1021/bi026175y
Ono T, Inoue Y (1988) Discrete extraction of the Ca atom functional for O2 evolution in higher plant photosystem II by a simple low pH treatment. FEBS Lett 227:147–152. doi:10.1016/0014-5793(88)80886-X
Ono T, Inoue Y (1989) Removal of Ca by pH 3.0 treatment inhibits S2 to S3 transition in photosynthetic oxygen evolution system. Biochim Biophys Acta 973:443–449. doi:10.1016/S0005-2728(89)80386-X
Ono T, Inoue Y (1990) Abnormal redox reactions in photosynthetic O2-evolving centers in NaCl/EDTA-washed PS II A dark-stable EPR multiline signal and an unknown positive charge accumulator. Biochim Biophys Acta 1020:269–277. doi:10.1016/0005-2728(90)90157-Y
Ono T, Zimmermann JL, Inoue Y, Rutherford AW (1986) EPR evidence for a modified S-state transition in chloride-depleted photosystem II. Biochim Biophys Acta 851:193–201. doi:10.1016/0005-2728(86)90125-8
Ono T, Nakayama H, Gleiter H, Inoue I, Kawamori A (1987) Modification of the properties of S2 state in photosynthetic O2-evolving center by replacement of chloride with other anions. Arch Biochem Biophys 256:618–624. doi:10.1016/0003-9861(87)90619-9
Popelková H, Yocum CF (2007) Current status of the role of Cl− ion in the oxygen-evolving complex. Photosynth Res 93:111–121. doi:10.1007/s11120-006-9121-5
Porra RJ, Tompson WA, Kriedemann PE (1989) Determination of accurate extinction coefficients and simultaneous-equations for assaying chlorophyll-a and chlorophyll-b extracted with 4 different solvents—verification of the concentration of chlorophyll standards by atomic-absorption spectroscopy. Biochim Biophys Acta 975:384–394. doi:10.1016/S0005-2728(89)80347-0
Pospišil P, Dau H (2000) Chlorophyll fluorescence transients of photosystem II membrane particles as a tool for studying photosynthetic oxygen evolution. Photosynth Res 65:41–52. doi:10.1023/A:1006469809812
Preston C, Seibert M (1991) The carboxyl modifier 1-ethyl-3-[3-(dimethylamino) propyl]carbodiimide (EDC) inhibits half of the high-affinity manganese-binding site in photosystem II membrane fragments. Biochemistry 30:9615–9624. doi:10.1021/bi00104a008
Rashid A, Homann PH (1992) Properties of iodide-activated photosynthetic water-oxidizing complexes. Biochim Biophys Acta 1101:303–310. doi:10.1016/0005-2728(92)90086-H
Sandusky PO, Yocum CF (1983) The mechanism of amine inhibition of the photosynthetic oxygen evolving complex. Amines displace functional chloride from a ligand site on manganese. FEBS Lett 162:339–343. doi:10.1016/0014-5793(83)80784-4
Sandusky PO, Yocum CF (1984) The chloride requirement for photosynthetic oxygen evolution. Analysis of the effects of chloride and other anions on amine inhibition of the oxygen-evolving complex. Biochim Biophys Acta 766:603–611. doi:10.1016/0005-2728(84)90121-X
Seibert M, Lavorel J (1983) Oxygen-evolution patterns from spinach photosystem II preparations. Biochim Biophys Acta 723:160–168. doi:10.1016/0005-2728(83)90115-9
Semin BK, Seibert M (2005) Iron bound to the high-affinity Mn-binding site of the oxygen-evolving complex shifts the pK of a component controlling electron transport via YZ. Biochemistry 43:6772–6782. doi:10.1021/bi036047p
Semin BK, Seibert M (2006) Flash-induced blocking of the high-affinity manganese-binding site in photosystem II by iron cations: dependence on the dark interval between flashes and binary oscillations of fluorescence yield. J Phys Chem B 110:25532–25542. doi:10.1021/jp0652796
Semin BK, Ghirardi ML, Seibert M (2002) Blocking of electron donation by Mn(II) to Y •Z following incubation of Mn-depleted photosystem II membranes with Fe(II) in the light. Biochemistry 41:5854–5864. doi:10.1021/bi0200054
Semin BK, Davletshina LN, Bulychev AA, Ivanov II, Seibert M, Rubin AB (2007) Effect of calcium chelators on the formation and oxidation of the slowly relaxing reduced plastoquinone pool in calcium-depleted PSII membranes. Investigation of the F0 yield. Biochemistry (Mosc) 72:1205–1215. doi:10.1134/S0006297907110065
Sivaraja M, Tso J, Dismukes GC (1989) A calcium-specific site influences the structure and activity of the manganese cluster responsible for photosynthetic water oxidation. Biochemistry 28:9459–9464. doi:10.1021/bi00450a032
Strasser BJ (1997) Donor side capacity of photosystem II probed by chlorophyll a fluorescence transients. Photosynth Res 52:147–155. doi:10.1023/A:1005896029778
Styring S, Rutherford AW (1988) Deactivation kinetics and temperature dependence of the S-state transitions in the oxygen-evolving system of photosystem II measured by EPR spectroscopy. Biochim Biophys Acta 933:378–387. doi:10.1016/0005-2728(88)90046-1
van Vliet P, Rutherford AW (1996) Properties of the chloride-depleted oxygen-evolving complex of photosystem II studied by electron paramagnetic resonance. Biochemistry 35:1829–1839. doi:10.1021/bi9514471
van Vliet P, Boussac A, Rutherford AW (1994) Chloride-depletion effects in the calcium-deficient oxygen-evolving complex of photosystem II. Biochemistry 33:12998–13004. doi:10.1021/bi00248a008
Vrettos JS, Limburg J, Brudvig GW (2001) Mechanism of photosynthetic water oxidation: combining biophysical studies of photosystem II with inorganic model chemistry. Biochim Biophys Acta 1503:229–245. doi:10.1016/S0005-2728(00)00214-0
Wincencjusz H, van Gorkom HJ, Yocum CF (1997) The photosynthetic oxygen evolving complex requires chloride for its redox state S2 → S3 and S3 → S0 transitions but not for S0 → S1 or S1 → S2 transitions. Biochemistry 36:3663–3670. doi:10.1021/bi9626719
Wydrzynski T, Huggins BJ, Jursinic PA (1985) Uncoupling of detectable O2 evolution from the apparent S-state transitions in photosystem II by lauroylcholine chloride: possible implications in the photosynthetic water-splitting mechanism. Biochim Biophys Acta 809:125–136. doi:10.1016/0005-2728(85)90175-6
Yano J, Kern J, Sauer K, Latimer J, Pushkar Y, Biesiadka J et al (2006) Where water is oxidized to dioxygen: structure of the photosynthetic Mn4Ca cluster. Science 314:821–825. doi:10.1126/science.1128186
Yocum CF (1991) Calcium activation photosynthetic of water oxidation. Biochim Biophys Acta 105:1–15
Acknowledgments
This study was supported by the Russian Foundation for Basic Research, Project Numbers 08-04-00490a (BS, LD) and 08-04-00354 (AR), and by the Office of Basic Energy Sciences, Office of Science, U.S. Department of Energy (MS). BKS appreciates support from the Division of Energy Biosciences, Office of Science, U.S. Department of Energy while at NREL. We thank Professor R. Debus for many helpful suggestions.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Semin, B.K., Davletshina, L.N., Ivanov, I.I. et al. Decoupling of the processes of molecular oxygen synthesis and electron transport in Ca2+-depleted PSII membranes. Photosynth Res 98, 235–249 (2008). https://doi.org/10.1007/s11120-008-9347-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11120-008-9347-5