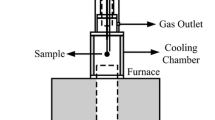
The paper examines the physicochemical processes that occur in the treatment of iron-containing metallurgical waste with the purpose of producing hydrogen with iron vapor method and subsequent processing the iron agglomerates. Iron oxides and reduced metal show high activity in the initial oxidation and reduction cycles. It is shown that the optimal oxidation–reduction temperature should be no more than 1000°C to prevent the intensive sintering of iron particles and decrease the activity in the reactive volume. The reduction process is most intensive in gaseous reducing medium.
Similar content being viewed by others
References
V. S. Zenkov, V. A. Lavrenko, and M. A. Kabanets, “A unit for studying heterogeneous interaction of materials with dissociated gases,” Zh. Fiz. Khim., 48, No. 5, 1300–1302 (1974).
V. S. Zenkov, “Influence of the reduction conditions for γ-Fe2O3 on the kinetics, phase composition, and particle size of reaction products,” in: Important Problems in Materials Technology [in Russian], Kiev (1996), pp. 117–132.
V. S. Zenkov, “Adsorption processes on getters of gaseous products of reduction of metal oxides,” in: Present-Day Achievements in Physical Materials Technology [in Russian], Kiev (1995), pp. 42–56.
S. T. Rostovtsev, Theory of Metallurgical Processes [in Russian], Metallurgizdat, Moscow (1956), pp. 117–132.
N. A. Vasyutinskii, Thermodynamics and Kinetics of Metal Reduction Processes [in Russian], Nauka, Moscow (1972), p. 375.
R. Ripan and I. Ceteanu, Inorganic Chemistry [Russian translation], Mir, Moscow (1972).
Author information
Authors and Affiliations
Corresponding author
Additional information
Translated from Poroshkovaya Metallurgiya, Vol. 47, No. 11–12 (464), pp. 138–152, 2008.
Rights and permissions
About this article
Cite this article
Zenkov, V.S., Pasichnyi, V.V. & Red’ko, V.P. Reduction of iron-containing metallurgical waste to obtain hydrogen with iron vapor method. Powder Metall Met Ceram 47, 733–742 (2008). https://doi.org/10.1007/s11106-009-9068-0
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11106-009-9068-0