Abstract
Background and Aim
Models predicting climate change suggest that the frequency of occurrence of combined biotic and abiotic stresses is likely to increase in the future. Plant growth promoting bacteria increase the defensive response of plants against these stresses. The main objective of this study was to identify, in the available collection of bacteria isolated from the peanut rhizosphere or phyllosphere, those whose inoculation protects against the effects of both high temperature (35 °C for 6 days) and Sclerotium rolfsii occurring simultaneously through induced systemic tolerance (IST).
Methods
The trials were carried out in pots containing a peanut plant growing under optimal conditions or under combined biotic and abiotic stress (high temperatures and Sclerotium rolfsii). Ninenative bacterial strains were tested to mitigate stress caused by such combined stressors by IST. Finally, systemic protection was confirmed by quantifying biochemical markers of plant defense.
Results
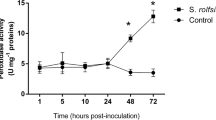
The native strain Paenibacillus sp. A224 was able to reverse the deleterious effect of both stresses on the growth and health of peanut plants. Furthermore, peroxidase activity and phenolic compound content were induced earlier in plants inoculated with Paenibacillus sp. A224 than in uninoculated ones exposed to both stresses, in concordance with their higher tolerance to S. rolfsii and high temperature.
Conclusion
The results indicate that inoculation of Paenibacillus sp. A224 could be a useful strategy to increase peanut tolerance against biotic and abiotic stresses that occur simultaneously, through a mechanism that involves the induction of systemic defense responses.






Similar content being viewed by others
References
Ahmad B, Raina A, Khan S (2019) Impact of biotic and abiotic stresses on plants, and their responses. Disease resistance in crop plants. Springer, Cham, pp 1–19
Ainsworth EA, Gillespie KM (2007) Estimation of total phenolic content and other oxidation substrates in plant tissues using folin–ciocalteu reagent. Nat Protoc 2:875–877. https://doi.org/10.1038/nprot.2007.102
Alderete LGS, Talano MA, Ibáñez SG, Purro S, Agostini E, Milrad SR, Medina MI (2009) Establishment of transgenic tobacco hairy roots expressing basic peroxidases and its application for phenol removal. JBiotechnol 139:273–279. https://doi.org/10.1016/j.jbiotec.2008.11.008
Arnon DI (1949) Copper enzymes in isolated chloroplasts. Polyphenoloxidase in Beta vulgaris. Plant Physiol 24:1. https://doi.org/10.1104/pp.24.1.1
Bates B, Kundzewicz Z, Wu S (2008) Climate change and water. Intergovernmental Panel on Climate Change Secretariat. Geneva pp 210
BCCBA, Bolsa de cereales de Córdoba (2022) https://www.bccba.org.ar/
Ben Rejeb I, Pastor V, Mauch-Mani B (2014) Plant responses to simultaneous biotic and abiotic stress: molecular mechanisms. Plants 3:458–475. https://doi.org/10.3390/plants3040458
Berg G (2009) Plant–microbe interactions promoting plant growth and health: perspectives for controlled use of microorganisms in agriculture. Appl MicrobiolBiotechnol 84:11–18. https://doi.org/10.1007/s00253-009-2092-7
Bradford M (1976) A rapid and sensitive method for the quantification of microgram quantities of protein utilizing the principle of protein dye binding. Annal Biochem 72:248–254. https://doi.org/10.1016/0003-2697(76)90527-3
Chen T, Duan L, Zhou B, Yu H, Zhu H, Cao Y, Zhang Z (2017) Interplay of pathogen-induced defense responses and symbiotic establishment in Medicago truncatula FrontMicrobiol 8:973. https://doi.org/10.3389/fmicb.2017.00973
Colodete CM, Ruas KF, de Oliveira Barbirato J, Barroso ALP, Dobbss LB (2015) Biochemistry characterization of proteins defense against oxidative stress in plants and their biosynthetic pathways of secondary metabolites. Natureza on line 13:195–204
Cox FR (1979) Effect of temperature treatment on peanut vegetative and fruit growth. Peanut Sci 6:14–17. https://doi.org/10.3146/i0095-3679-6-1-4
Dart PJ, Mercer FV (1965) The effect of growth temperature, level of ammonium nitrate, and light intensity on the growth and nodulation of cowpea (Vigna sinensis Endl. Ex Hassk.). Aust J Agric Res 16:321–345. https://doi.org/10.1071/AR9650321
De Gara L, de Pinto MC, Tommasi F (2003) The antioxidant systems vis-à-vis reactive oxygen species during plant–pathogen interaction. Plant PhysiolBiochem 41:863–870. https://doi.org/10.1016/S0981-9428(03)00135-9
Figueredo MS, Ibáñez F, Rodríguez J, Fabra A (2018) Simultaneous inoculation with beneficial and pathogenic microorganisms modifies peanut plant responses triggered by each microorganism. Plant Soil 433:353–361. https://doi.org/10.1007/s11104-018-3846-8
Figueredo MS, Tonelli ML, Ibáñez F, Morla F, Cerioni G, Tordable M, Fabra A (2017) Induced systemic resistance and symbiotic performance of peanut plants challenged with fungal pathogens and co-inoculated with the biocontrol agent Bacillus sp. CHEP5 and Bradyrhizobium sp. SEMIA6144. Microbiol Res 197:65–73. https://doi.org/10.1016/j.micres.2017.01.002
Frings JFJ (1976) The Rhizobiumpea symbiosis as affected by high temperatures. Wageningen University and Research, Wegeningen, Netherlands
Garret KA, Dendy SP, Frank EE, Rouse MN, Travers SE (2006) Climate change effects on plant disease: genomes to ecosystems. Annu Rev Phytopathol 44:489–509. https://doi.org/10.1146/annurev.phyto.44.070505.143420
Gupta C, Dubey R, Maheshwari D (2002) Plant growth enhancement and suppression of Macrophominaphaseolina causing charcoal rot of peanut by fluorescent Pseudomonas. Biol Fert Soils 36:399–405. https://doi.org/10.1007/s00374-002-0486-0
Hasanuzzaman M, Nahar K, Alam MM, Roychowdhury R, Fujita M (2013) Physiological, biochemical, and molecular mechanisms of heat stress tolerance in plants. Int J Mol Sci 14:9643–9684. https://doi.org/10.3390/ijms14059643
Heath RL, Packer L (1968) Photoperoxidation in isolated chloroplasts: I. Kinetics and stoichiometry of fatty acid peroxidation. Arch Biochem Biophys 125:189–198. https://doi.org/10.1016/0003-9861(68)90654-1
Hernandez-Armenta R, Wien HC, Eaglesham ARJ (1989) Maximum temperature for nitrogen fixation in common bean. Crop Sci 29:1260–1265. https://doi.org/10.2135/cropsci1989.0011183X002900050034x
Hoagland DR, Arnon DI (1950) The water-culture method for growing plants without soil. Circular. California agricultural experiment station, 347(2nd edit), pp 32
Hungria M, Franco AA (1993) Effects of high temperature on nodulation and nitrogen fixation by Phaseolus vulgaris L. Plant Soil 149:95–102. https://doi.org/10.1007/BF00010766
Hungria M, Vargas MA (2000) Environmental factors affecting N2 fixation in grain legumes in the tropics, with an emphasis on Brazil. Field Crops Res 65:151–164. https://doi.org/10.1016/S0378-4290(99)00084-2
Jespersen D (2020) Heat shock induced stress tolerance in plants: physiological, biochemical, and molecular mechanisms of acquired tolerance. In: Hossain MA, Liu F, David J, Burritt D (eds) Priming-mediated stress and cross-stress tolerance in crop plants, 1st edn. Academic Press Elsevier Inc, London, pp 161–174
Ketring DL (1985) Evaluation of peanut genotypes for membrane thermostability. Peanut Sci 12:28–32. https://doi.org/10.3146/pnut.12.1.0007
Landy MWGH, Warren GH, Rosenmanm SB, Colio LG (1948) Bacillomycin: an antibiotic from Bacillus subtilis active against pathogenic fungi. Proc Soc Exp Biol Med 67:539–541. https://doi.org/10.3181/00379727-67-16367
Lehmann S, Serrano M, L’Haridon F, Tjamos SE, Metraux JP (2015) Reactive oxygen species and plant resistance to fungal pathogens. Phytochem 112:54–62. https://doi.org/10.1016/j.phytochem.2014.08.027
Lopez-Delacalle M, Silva CJ, Mestre TC, Martinez V, Blanco-UlateB, Rivero RM (2021) Synchronization of proline, ascorbate and oxidative stress pathways under the combination of salinity and heat in tomato plants. Environ Exp Bot 183:104351. https://doi.org/10.1016/j.envexpbot.2020.104351
Lopez-Gomez M, Sandal N, Stougaard J, Boller T (2012) Interplay of flg22-induced defence responses and nodulation in Lotus japonicus J Exp Bot 63:393–401. https://doi.org/10.1093/jxb/err291
Lugtenberg B, Kamilova F (2009) Plant-growth-promoting rhizobacteria. AnnuRevMicrobiol 63:541–556. https://doi.org/10.1146/annurev.micro.62.081307.162918
March G, Marinelli A (2005) In: Enfermedades del maní en Argentina. Bigli a Impresores, Córdoba, pp 142
Marfo KO, Denwar NN, Adu-Dapaah HK, AsafoAgyei B, Marfo KA, Adjei J, Haleegoah J (1999) Groundnutproduction in Ghana. In: Proceedings of a national workshop on groundmised groundnut aflatoxins, Kumasi Ghana, pp 11–16
Marinelli A, March G, Oddino C (2017) Enfermedades fúngicas del maní. In: El cultivo del Maníen Córdoba. ISBN 978-987-42-3736-1
McKinney KB (1938) Physical characteristics on the foliage of beans and tomatoes that tend to control some small insect pests. J Econ Entomol 31. https://doi.org/10.1093/jee/31.5.630
Meyer DR, Anderson AJ (1959) Temperature and symbiotic nitrogen fixation. Nature 183:61–61. https://doi.org/10.1038/183061a0
Pankhurst CE, Sprent JI (1976) Effects of temperature and oxygen tension on the nitrogenase and respiratory activities of turgid and water-stressed soybean and french bean root nodules. J Exp Bot 27:1–9. https://doi.org/10.1093/jxb/27.1.1-a
Peck S, Mittler R (2020) Plant signaling in biotic and abiotic stress. J Exp Bot 71:1649–1651. https://doi.org/10.1093/jxb/eraa051
Piha MI, Munns DN (1987) Sensitivity of the common bean (Phaseolus vulgaris L.) symbiosis to high soil temperature. Plant Soil 98:183–194. https://doi.org/10.1007/BF02374822
Pörtner HO, Roberts DC, Adams H, Adler C, Aldunce P, Ali E, Birkmann J (2022) Climate change 2022: impacts, adaptation and vulnerability. IPCC Sixth Assessment Report
Prášil I, Zámečník J (1998) The use of a conductivity measurement method for assessing freezing injury: I. Influence of leakage time, segment number, size and shape in a sample on evaluation of the degree of injury. Environ Exp Bot 40:1–10. https://doi.org/10.1016/S0098-8472(98)00010-0
Preininger C, Sauer U, Bejarano A, Berninger T (2018) Concepts and applications of foliar spray for microbial inoculants. App Microbiol Biotechnol 102:7265–7282. https://doi.org/10.1007/s00253-018-9173-4
Ramegowda V, Senthil-Kumar M (2015) The interactive effects of simultaneous biotic and abiotic stresses on plants: mechanistic understanding from drought and pathogen combination. J Plant Physiol 176:47–54. https://doi.org/10.1016/j.jplph.2014.11.008
Ramegowda V, Senthil-Kumar M, Ishiga Y, Kaundal A, Udayakumar M, Mysore KS (2013) Drought stress acclimation imparts tolerance to Sclerotinia sclerotiorum and Pseudomonas syringae in Nicotiana benthamiana Int J Mol Sci 14:9497–9513. https://doi.org/10.3390/ijms14059497
Rivero RM, Mittler R, Blumwald E, Zandalinas SI (2022) Developing climate-resilient crops: improving plant tolerance to stress combination. Plant J 109:373–389. https://doi.org/10.1111/tpj.15483
Sarkar J, Chakraborty B, Chakraborty U (2018) Plant growth promoting rhizobacteria protect wheat plants against temperature stress through antioxidant signalling and reducing chloroplast and membrane injury. J Plant Growth Regul 37:1396–1412. https://doi.org/10.1007/s00344-018-9789-8
Shaikh SS, Sayyed RZ (2015) Role of plant growth-promoting rhizobacteria and their formulation in biocontrol of plant diseases. Plant microbes symbiosis: Applied facets. Springer, New Delhi, pp 337–351. https://doi.org/10.1007/978-81-322-2068-8_18
Sharma P, Jha AB, Dubey RS, Pessarakli M (2012) Reactive oxygen species, oxidative damage, and antioxidative defense mechanism in plants under stressful conditions. J Bot. https://doi.org/10.1155/2012/217037
Solomon S, Manning M, Marquis M, Qin D (2007) Climate change 2007-the physical science basis: Working group I contribution to the fourth assessment report of the IPCC (Vol. 4). Cambridge University Press, Cambridge
Somasegaran P, Hoben HJ (1994) Quantifying the growth of rhizobia. Handbook for rhizobia. Springer, New York, pp 47–57
Sousa B, Rodrigues F, Soares C, Martins M, Azenha M, Lino-Neto T, Santos C, Cunha A, Fidalgo F (2022) Impact of combined heat and salt stresses on tomato plants-insights into nutrient uptake and redox homeostasis. Antioxidants 11:478. https://doi.org/10.3390/antiox11030478
Talwar HS, Yanagihara S, Yajima M, Hayashi T (1999) Physiological basis for heat tolerance during flowering and pod setting stages in groundnut (Arachis hypogaea L.). JIRCAS Working Report 14:47–65
Timmusk S, Seisenbaeva G, Behers L (2018) Titania (TiO2) nanoparticles enhance the performance of growth-promoting rhizobacteria. Sci Rep 8:617. https://doi.org/10.1038/s41598-017-18939-x
Tonelli ML, Taurian T, Ibáñez F, Angelini J, Fabra A (2010) Selection and in vitro characterization of biocontrol agents with potential to protect peanut plants against fungal pathogens. J Plant Pathol 73–82. http://www.jstor.org/stable/41998770
Van Loon LC, Bakker PAHM, Pieterse CMJ (1998) Systemic resistance induced by rhizosphere bacteria. Annu Rev Phytopathol 36:453–483. https://doi.org/10.1146/annurev.phyto.36.1.453
Vara Prasad PV, Craufurd PQ, Summerfield RJ (1999) Sensitivity of peanut to timing of heat stress during reproductive development. Crop Sci 39:1352–1357. https://doi.org/10.2135/cropsci1999.3951352x
Vara Prasad PV, Craufurd PQ, Summerfield RJ (1999) Fruit number in relation to pollen production and viability in groundnut exposed to short episodes of heat stress. Ann Bot 84:381–386. https://doi.org/10.1006/anbo.1999.0926
Vara Prasad PV, Craufurd PQ, Summerfield RJ (2000) Effect of high air and soil temperature on dry matter production, pod yield and yield components of groundnut. Plant Soil 222:231–239. https://doi.org/10.1023/A:1004793220787
Vara Prasad PV, Craufurd PQ, Summerfield RJ (2001) Response of groundnuts dependent on symbiotic and inorganic nitrogen to high air and soil temperatures. J Plant Nutr 24:623–637. https://doi.org/10.1081/PLN-100103657
Vincent J (1970) In: a manual for the practical study of root nodule bacteria. International Biological Programme Handbook nº 15. Blackwell Scientific Publications Ltd, Oxford, pp 73–97
Wahid A, Gelani S, Ashraf M, Foolad MR (2007) Heat tolerance in plants: an overview. Environ Exp Bot 61:199–223. https://doi.org/10.1016/j.envexpbot.2007.05.011
Walters M (2015) The plant innate immune system. Endocytobiosis Cell Res 26:8–12
Wheeler TR, Chatzialioglou A, Craufurd PQ, Ellis RH, Summerfield RJ (1997) Dry matter partitioning in groundnut exposed to high temperature stress. Crop Sci 37:1507–1513. https://doi.org/10.2135/cropsci1997.0011183X003700050016x
Williams JH, Boote KJ (1995) Physiology and modeling-predicting the unpredictable legume. In: Advances in peanut science. Am Peanut Res Educ Soc. Stillwater pp 301–353
Wise RR, Olson AJ, Schrader SM, Sharkey TD (2004) Electron transport is the functional limitation of photosynthesis in field-grown Pima cotton plants at high temperature. Plant Cell Environ 27:717–724. https://doi.org/10.1111/j.1365-3040.2004.01171.x
Wood IMW (1968) The effect of temperature at early flowering on the growth and development of peanuts (Arachis hypogaea). Aust J Agric Res 19:241–251. https://doi.org/10.1071/AR9680241
Xalxo R, Yadu B, Chandra J, Chandrakar V, Keshavkant S (2020) Alteration in carbohydrate metabolism modulates thermotolerance of plant under heat stress. In: Wani SH, Kumar V (eds) Heat stress tolerance in plants: physiological, molecular and genetic perspectives, 1st edn. Wiley, Hoboken, pp 77–115
Yang J, Kloepper JW, Ryu CM (2009) Rhizosphere bacteria help plants tolerate abiotic stress. Trends Plant Sci 14:1–4. https://doi.org/10.1016/j.tplants.2008.10.004
Yu Y, Gui Y, Li Z, Jiang C, Guo J, Niu D (2022) Induced systemic resistance for improving plant immunity by beneficial microbes. Plants 11:386. https://doi.org/10.3390/plants11030386
Zandalinas SI, Fritschi FB, Mittler R (2020) Signal transduction networks during stress combination. J Exp Bot 71:1734–1741. https://doi.org/10.1093/jxb/erz486
Zhou J, Wang J, Zheng Z, Fan B, Yu JQ, Chen Z (2015) Characterization of the promoter and extended C-terminal domain of Arabidopsis WRKY33 and functional analysis of tomato WRKY33 homologues in plant stress responses. J Exp Bot 66:4567–4583. https://doi.org/10.1093/jxb/erv221
Acknowledgements
María Soledad Figueredo, María Laura Tonelli and Adriana Fabra are members of the Research Career from CONICET.
Funding
Thisstudywasfinanciallysupportedby Ministerio de Ciencia y Tecnología de la provincia de Córdoba, Fundación Maní Argentino and Agencia Nacional de Promoción Científica y Tecnológica (ANPCyT).
Author information
Authors and Affiliations
Contributions
AF, MSF and MLTconceived and designed the experiments, and wrote the manuscript.
MSF and TA performed the experiments.
Corresponding author
Additional information
Responsible Editor: Stéphane Compant.
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Below is the link to the electronic supplementary material.
ESM 1
(DOCX 855 KB)
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Figueredo, M.S., Álamo, T., Tonelli, M.L. et al. The native strain Paenibacillus sp. A224 induces systemic tolerance and mitigates stresses caused in peanut plants by high temperatures and the pathogen Sclerotium rolfsii. Plant Soil 486, 375–390 (2023). https://doi.org/10.1007/s11104-023-05876-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11104-023-05876-0